8.16.1: Biology- Ketosis
- Page ID
- 50827
Prevalent biomolecules processed by the human body every day are carbohydrates, fatty acids, and amino acids, and the ingestion of ethanol in alcoholic beverages is, in moderation, somewhat harmless. These seemingly perfectly edible compounds undergo chemical changes in the body that produce molecules that are not so biologically friendly especially in large amounts. The removal of an amine group from an amino acid helps with the decomposition of fatty acids into acetone, acetoacetic acid, and beta-hydroxybutyric acid[1]. Acetone (and the other two molecules to a lesser extent) are members of the ketone family.
Ethanol is easily oxidized to acetaldehyde, one of the most important aldehydes. Ketones and aldehydes are large families of organic compounds that have the 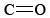 functional group, called a carbonyl group.
functional group, called a carbonyl group.
|
From left to right: ethanol, acetaldehyde, and acetone. |
Rotate the jmol structures to see the similarities and differences between each model. Just as ethanol is oxidized to acetaldehyde, methanol is oxidized to formaldehyde. The loss of the hydrogen atom connected to methanol's oxygen atom forces it to double bond with the adjacent carbon atom, creating the carbonyl group. A ketone such as acetone shares this characteristic, except that the carbonyl group is attached to two carbons.
EXAMPLE
Examine what type of alcohol methanol is and how it oxidizes to formaldehyde. Using this information, what type of alcohol do you think would oxidize to form a ketone?
Answer
Methanol is a primary alcohol, with its hydroxyl group at the end of a carbon chain. Because the carbonyl group of a ketone is bonded to a carbon atom connected to two other carbon atoms, we should deduce that secondary alcohols oxidize to produce ketones.
EXAMPLE
Using what you know about hydrogen bonding, qualitatively compare the melting and boiling points of aldehydes and ketones to their alcohol derivatives.
Answer
Strong hydrogen bonding occurs between alcohols because of their hydroxyl (OH) groups. When these alcohols are oxidized, the hydrogen atom bonded to the oxygen atom is lost in order to form the double bond and carbonyl group. Therefore, aldehydes and ketones have weaker lower intermolecular forces and thus lower melting and boiling points compared to similarly sized alcohols. The carbonyl group is rather polar, however, since the difference between the electronegativities of carbon (2.5) and oxygen (3.5) is rather large, and there are usually no other dipoles in an aldehyde or ketone molecule to cancel the effect of C==O.
Therefore the boiling points of aldehydes and ketones are intermediate between those of alkanes or ethers on the one hand and alcohols on the other. Acetaldehyde, CH3CH2CHO, boils at 20.8°C midway between propane (–42°C) and ethanol (78.5°C). The boiling points of propanal and acetone are compared with other organic compounds in the table of the boiling points of comparable organic compounds which shows the same trend.
Nomenclature and Structure
The endings al and one signify aldehyde and ketone, respectively. Compounds with lower molecular weights have common names, such as formaldehyde, but standard IUPAC nomenclature can also be applied.
Aldehydes are named just as the alcohols from which they are oxidized, just replacing the "ol" with "al". For example, the primary alcohol butanol would oxidize to butanal. The names of ketones depend on where the carbonyl group is located in the chain. When counting the carbon atoms in the longest carbon backbone, the number of the carbon included in the carbonyl group is inserted between the base name and the suffix "one". An organic molecule with five carbon atoms with the carbonyl group consisting of an oxygen atom double bonded to the third carbon would be termed pentan-3-one. Alternatively, the digit can be placed in front, giving 3-pentanone.
The general formula for an aldehyde is 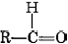 , while for a ketone it is
, while for a ketone it is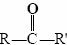 . Note that every ketone is isomeric with at least one aldehyde. Acetone, for example, has the same molecular formula (C3H6O) as propanal.
. Note that every ketone is isomeric with at least one aldehyde. Acetone, for example, has the same molecular formula (C3H6O) as propanal.
EXAMPLE Name the organic molecules shown below.
a)
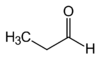
b)
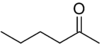
Answer
a) Propanal
b) 2-Hexanone or Hexan-2-one
Detecting Aldehydes and Ketones
Chemical tests may be performed to determine the presence of a carbonyl group. Immunochemically, it can be found by adding 2,4-dinitrophenylhydrazine to test tubes containing 2-propanol, an alcohol, 2-propanone (acetone), a ketone; and propionic acid, a carboxylic acid. 2,4-dinitrophenylhydrazine only reacts with the carbonyl group of 2-propanone, forming an orange precipitate.
The reaction that occurs is:
Another test can distinguish between aldehydes and ketones, using Tollens' reagent, which is an aqueous solution of silver nitrate, sodium hydroxide, and a little ammonia. If an aldehyde, in this case, glucose, is added to the solution, the Ag+ is reduced by the aldehyde, and the aldehyde is oxidized into a carboxylic acid. This produces silver metal, which coats the flask and creates the mirror. Ketones are not as easily oxidized, so only aldehydes produce the silver mirror.
The equation for the reaction in the video is:
Biological Impacts
The simplest aldehydes and ketones are the most commercially important ones. Formaldehyde, the simplest aldehyde, is one of the most widely used biological preservatives; you have probably seen a jar of it containing a random animal organ. It kills most bacteria and fungi, but its true value lies in its size. Many preservatives are chosen because they react with the covalent bonds in chemical tissue, hardening it. Because formaldehyde is smaller compared to other common preservatives, it can penetrate the outer layer of organic tissue and react away more easily.
The speed of this reaction is especially important when the tissue is in an environment in which formaldehyde itself is produced by a chemical reaction that is not especially product-favored. For example, when formaldehyde is in an equilibrium reaction with methanediol or amines, the equilibrium constant is not favored towards formaldehyde. However, as the aldehyde reacts away with organic matter, the equilibrium shifts, creating more formaldehyde to partially replenish the used product[2]. This is consistent with Le Chatelier's Principle.
Acetone, the simplest ketone, is very miscible with water and is common in the body. As mentioned above, large ketones are produced naturally in the liver through the decomposition of fatty acids. These molecules will lose most of their carbonyl groups in a process called decarboxylation, where the carbonyl breaks away to make carbon dioxide. Acetone is the end product and excreted through breath and urine. When the metabolism is unable to quickly flush out ketones, the body enters a state of ketosis. This often happens when not enough carbohydrates are available in the bloodstream.
Does this deprivation of carbs sound familiar? When ketosis is induced, the body relies more of stores of fat for energy. Forced exclusion of carbohydrates from one's diet in order to lose - yes, this is the Atkins diet, chemically dependent on ketosis. While this process is normal, carbohydrate exclusion can lead to prolonged states of ketosis that induces dehydration and stresses the liver. In extreme diabetic or alcoholic cases, the uncontrolled production of ketones, especially acetoacetic acid and beta-hydroxybutyric acid, leads to ketoacidosis. In this state, the pH of a person's bloodstream drops below 7.2, and in some cases may be fatal[3]. Ketoacidosis can be identified by a person's breath; the high levels of acetone exhaled can be detected as having a slightly fruity scent, though some say the smell is more like nail polish remover.
References
- ↑ Ketone bodies: a review of physiology, pathophysiology and application of monitoring to diabetes [www.ncbi.nlm.nih.gov]
- ↑ Goldstein Fixation Time Handout SFAI Fall Lecture [www.appliedimmuno.org]
- ↑ Alcoholic ketoacidosis [www.nlm.nih.gov]


