8.8.2: Table of Electronegativities
- Page ID
- 49740
This table displays the Linus Pauling derivation of electronegativities. Fluorine, the most electronegative element, has arbitrarily been given a value of 4.0. Every other element's electronegativity has been scaled accordingly.
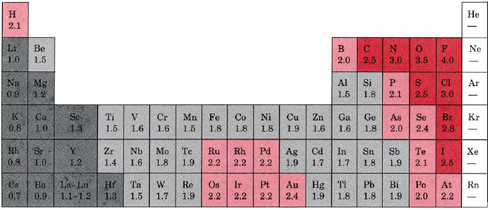
Elements with electronegativities of 2.5 or more are all nonmetals in the top right-hand comer of the periodic table. These have been color-coded dark red. By contrast, elements with negativities of 1.3 or less are all metals on the lower left of the table. These elements have been coded in dark gray. They are often referred to as the most electropositive elements, and they are the metals which invariably form binary ionic compounds. Between these two extremes we notice that most of the remaining metals (largely transition metals) have electronegativities between 1.4 and 1.9 (light gray), while most of the remaining nonmetals have electronegativities between 2.0 and 2.4 (light red). Another feature worth noting is the very large differences in electronegativities in the top right-hand comer of the table. Fluorine, with an electronegativity of 4, is by far the most electronegative element. At 3.5 oxygen is a distant second, while chlorine and nitrogen are tied for third place at 3.0.


