6.7: Ionization of Transition and Inner Transition Elements
- Page ID
- 49350
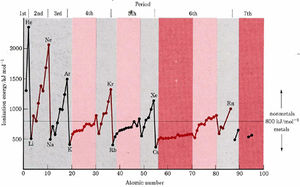
On the graph of ionization energies, it can be seen that ionization energies increase much more slowly across the transition and inner transition elements than for the representative elements. For example, the ionization energy of the representative-element boron is 800 kJ mol–1. Five elements later we find neon, whose ionization energy is 2080 kJ mol–1, an increase of 160 percent. In the fourth period, the transition-element scandium has an ionization energy of 631 kJ mol–1. Five elements later we find iron at 759 kJ mol–1, an increase of only 20 percent. All the lanthanoids have ionization energies from 500 to 600 kJ mol–1, and the actinoids are all between 580 and 680 kJ mol–1.
These similarities among the transition and especially the inner transition elements illustrate statements made about electron configurations and the periodic table. The distinguishing electron for a transition element enters a d subshell in the next-to-outermost shell, while for an inner transition element it usually enters an f subshell in the third-from-outermost shell.
Thus the distinction between an element and the one preceding it in the periodic table is much smaller than among the representative elements. Furthermore, experimental measurements show that for transition and inner transition elements the electrons lost when ionization occurs are not the last ones which were added to build up the atomic electron configuration. Instead, electrons are usually removed first from the subshell having the largest principal quantum number.
Example 6.7.1 : Electron Configuration
Determine the electron configuration of the Fe3+ ion.
Solution
Since the charge on the ion is +3, three electrons must have been removed from a neutral iron atom (Fe). The electron configuration of Fe is
Fe:...1s22s22p63s23p63d64s2 or [Ar]3d64s2
We now remove electrons successively from subshells having the largest principal quantum number:
Fe+ : [Ar]3d64s1 one 4s electron removed
Fe2+: [Ar]3d6 a second 4s electron removed
Fe3+: [Ar]3d5 since no electrons are left in the n = 4 shell, one 3d electron is removed
The behavior described in the previous paragraph and the example may be better understood by comparing the 3d and 4s shells, as in the following figure.
_and_4s_(color)_Electron_Clouds.jpg?revision=1&size=bestfit&width=429&height=319)
Electrons in the subshell having the largest principal quantum number (4s in the example and the figure above) are, on the average, farther from the nucleus, and they are first to be removed. The first ionization energy of iron is not much larger than that of scandium because in each case a 4s electron is being removed. The iron atom has five more protons in the nucleus, but it also has five more 3d electrons which spend most of their time between the nucleus and the 4s electrons. The screening effect of such 3d electrons causes the effective nuclear charge to increase very slowly from one transition element to the next. The attraction for 4s electrons, and hence ionization energy, also increases very slowly.
Metals
Macroscopic properties such as high thermal and electric conductivity, malleability, and ductility were mentioned in a brief introduction to the elements as characteristics of metals. In addition, most metals have low ionization energies, usually below 800 kJ mol–1. In other words, a metal consists of atoms, each of which has at least one loosely held electron.
When such atoms pack close together in a solid metal, the loosely held electrons are relatively free to move from one atom to another. If excess electrons are forced into one end of a metal wire, it is relatively easy for electrons to flow out of the other end. Thus an electric current may be carried through the wire, and the high conductivity of all metals may be understood.
More detailed microscopic interpretations of metallic properties are given for Metals later, but for the time being we are primarily interested in the location of metallic elements in the periodic table. Ionization energies are smallest near the bottom and on the left of the periodic table, and so this is where metals are found. Moreover, ionization energies increase slowly from one transition element to the next and hardly at all across the inner transition elements. Therefore all transition and inner transition elements are metals.
In periodic groups IIIA, IVA, and VA elements near the top of the table have large ionization energies and little metallic character. Ionization energies decrease as one moves downward, however. For example, Al is quite metallic, although the element above it, B, is not. A heavy “stairstep” line is usually drawn on the periodic table to separates the nonmetals (above and to the right) from the metals. Elements such as B, Si, Ge, As, Sb, and Te, which are adjacent to the stairstep, have intermediate properties and are called semimetals. This same class is also referred to as metalloids.


