9.3: Reactions of Alkynes - Addition of HX and X₂
- Last updated
- Save as PDF
- Page ID
- 227108
Learning Objective
- predict the products and specify the reagents for the Electrophilic Addition Reactions (EARs) of alkynes with HX and X2
Addition by Electrophilic Reagents
Since the most common chemical transformation of a carbon-carbon double bond is an addition reaction, we might expect the same to be true for carbon-carbon triple bonds. Indeed, most of the alkene addition reactions also take place with alkynes with similar regio- and stereoselectivity.
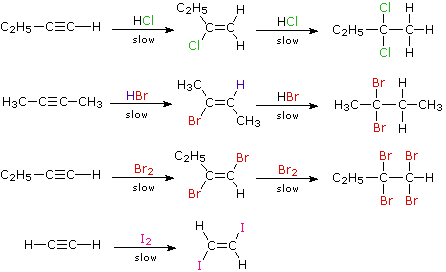
When the addition reactions of electrophilic reagents, such as strong Brønsted acids and halogens, to alkynes are studied we find a curious paradox. The reactions are even more exothermic than the additions to alkenes, and yet the rate of addition to alkynes is slower by a factor of 100 to 1000 than addition to equivalently substituted alkenes. The reaction of one equivalent of bromine with 1-penten-4-yne, for example, gave 4,5-dibromo-1-pentyne as the chief product.
HC≡C-CH2-CH=CH2 + Br2 → HC≡C-CH2-CHBrCH2Br
Although these electrophilic additions to alkynes are sluggish, they do take place and generally display Markovnikov Rule regioselectivity and anti-stereoselectivity. One problem, of course, is that the products of these additions are themselves substituted alkenes and can therefore undergo further addition. Because of their high electronegativity, halogen substituents on a double bond act to reduce its nucleophilicity, and thereby decrease the rate of electrophilic addition reactions. Consequently, there is a delicate balance as to whether the product of an initial addition to an alkyne will suffer further addition to a saturated product. Although the initial alkene products can often be isolated and identified, they are commonly present in mixtures of products and may not be obtained in high yield. The following reactions illustrate many of these features. In the last example, 1,2-diodoethene does not suffer further addition inasmuch as vicinal-diiodoalkanes are relatively unstable.
As a rule, electrophilic addition reactions to alkenes and alkynes proceed by initial formation of a pi-complex, in which the electrophile accepts electrons from and becomes weakly bonded to the multiple bond. Such complexes are formed reversibly and may then reorganize to a reactive intermediate in a slower, rate-determining step. Reactions with alkynes are more sensitive to solvent changes and catalytic influences than are equivalent alkenes.
Why are the reactions of alkynes with electrophilic reagents more sluggish than the corresponding reactions of alkenes? After all, addition reactions to alkynes are generally more exothermic than additions to alkenes, and there would seem to be a higher π-electron density about the triple bond ( two π-bonds versus one ). Two factors are significant in explaining this apparent paradox. First, although there are more π-electrons associated with the triple bond, the sp-hybridized carbons exert a strong attraction for these π-electrons, which are consequently bound more tightly to the functional group than are the π-electrons of a double bond. This is seen in the ionization potentials of ethylene and acetylene.
| Acetylene | HC≡CH + Energy → [HC≡CH •(+) + e(–) | ΔH = +264 kcal/mole | |
|---|---|---|---|
| Ethylene | H2C=CH2 + Energy → [H2C=CH2] •(+) + e(–) | ΔH = +244 kcal/mole | |
| Ethane | H3C–CH3 + Energy → [H3C–CH3] •(+) + e(–) | ΔH = +296 kcal/mole |
As defined by the preceding equations, an ionization potential is the minimum energy required to remove an electron from a molecule of a compound. Since pi-electrons are less tightly held than sigma-electrons, we expect the ionization potentials of ethylene and acetylene to be lower than that of ethane, as is the case. Gas-phase proton affinities show the same order, with ethylene being more basic than acetylene, and ethane being less basic than either. Since the initial interaction between an electrophile and an alkene or alkyne is the formation of a pi-complex, in which the electrophile accepts electrons from and becomes weakly bonded to the multiple bond, the relatively slower reactions of alkynes becomes understandable.
A second factor is presumed to be the stability of the carbocation intermediate generated by sigma-bonding of a proton or other electrophile to one of the triple bond carbon atoms. This intermediate has its positive charge localized on an unsaturated carbon, and such vinyl cations are less stable than their saturated analogs. Indeed, we can modify our earlier ordering of carbocation stability to include these vinyl cations in the manner shown below. It is possible that vinyl cations stabilized by conjugation with an aryl substituent are intermediates in HX addition to alkynes of the type Ar-C≡C-R, but such intermediates are not formed in all alkyne addition reactions.
|
Application of the Hammond postulate indicates that the activation energy for the generation of a vinyl cation intermediate would be higher than that for a lower energy intermediate. This is illustrated for alkenes versus alkynes by the following energy diagrams.
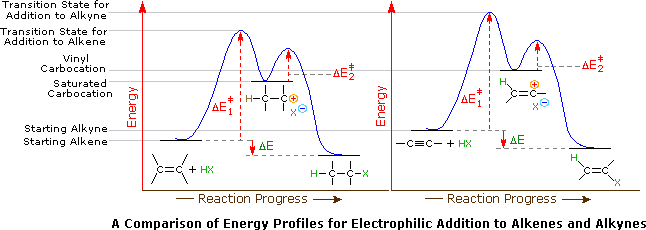
Despite these differences, electrophilic additions to alkynes have emerged as exceptionally useful synthetic transforms.
Addition of Hydrogen Halide to an Alkyne
Summary: Reactivity order of hydrogen halides: HI > HB r> HCl > HF.
Follows Markovnikov’s rule:
- Hydrogen adds to the carbon with the greatest number of hydrogens, the halogen adds to the carbon with fewest hydrogens.
- Protination occurs on the more stable carbocation. With the addition of HX, haloalkenes form.
- With the addition of excess HX, you get anti addition forming a geminal dihaloalkane.
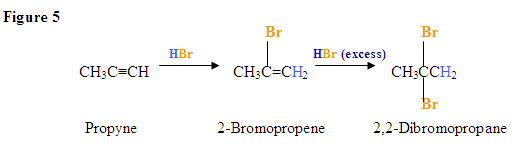
Addition of a HX to an Internal Alkyne
As shown in Figure 2 below, the \(\pi\) electrons react with the hydrogen of the HBr and because the alkyne carbons are equivalent it does not matter which carbon adds the hydrogen. Once the hydrogen is covalently bonded to one of the carbons, the bromide will react with the carbocation intermediate to form a vinyl halide as shown in the example of forming 2-bromobutene from 2-butyne reacting with HBr. The reaction below assumes a 1:1 mole ratio of the alkyne and HBr.
.bmp?revision=1&size=bestfit&width=442&height=160)
Now, what happens if there is excess HBr?
Addition due to excess HX yields a geminal dihaloalkane
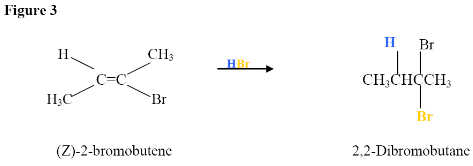
Here, the electrophilic addition proceeds with the same steps used to achieve the product in Addition of a HX to an Internal Alkyne. The \(\pi\) electrons react with the hydrogen (shown in blue) adding it to the carbon on the left because the lone pair electrons of the bromine can help stabilize the carbocation intermediate that reacts with the bromide ions to form a geminal dihalide.
Addition of HX to Terminal Alkyne
For terminal alkynes, the carbon atoms sharing the triple bond are not equivalent. The addition of HX to terminal alkynes occurs in a Markovnikov-manner in which the halide attaches to the most substituted carbon. The pi electrons react with the hydrogen and it bonds to the terminal carbon. The bromide reacts with the resulting carbocation intermediate to form the vinyl halide. The overall reaction and mechanism are shown below.
Addition due to excess HBr present
Similar to the addition of excess HBr to internal alkynes, both halides will add to the same carbon to form a geminal dihalide.
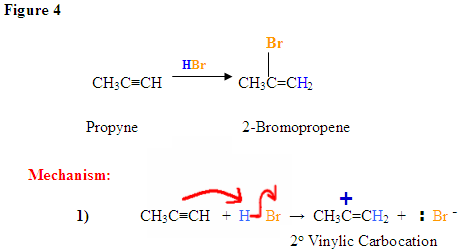
HBr Addition With Radical Mechanism
Most hydrogen halide reactions with terminal alkynes occur in a Markovnikov-manner in which the halide attaches to the most substituted carbon since it is the most positively polarized. However, there are two specific reactions among alkynes where anti-Markovnikov reactions take place: the radical addition of HBr and Hydroboration Oxidation reactions. For alkynes, an anti-Markovnikov addition takes place for terminal alkynes.
The Br of the Hydrogen Bromide (H-Br) attaches to the less substituted 1-carbon of the terminal alkyne shown below in an anti-Markovnikov manner while the Hydrogen proton attaches to the second carbon. As mentioned above, the first carbon is the less substituted carbon since it has fewer bonds attached to carbons and other substituents. The H-Br reagent must also be reacted with heat or some other radicial initiator such as a peroxide in order for this reaction to proceed in this manner. This presence of the radical or heat leads to the anti-Markovnikov addition since it produces the most stable reaction.
The product of a terminal alkyne that is reacted with a peroxide (or light) and H-Br is a 1-bromoalkene.
Regioselectivity: The Bromine can attach in a syn or anti manner which means the resulting alkene can be both cis and trans. Syn addition is when both Hydrogens attach to the same face or side of the double bond (i.e. cis) while the anti addition is when they attach on opposite sides of the bond (trans).
.jpg?revision=1&size=bestfit&width=504&height=271)
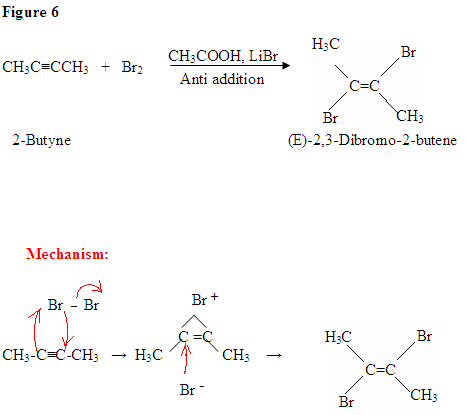
Halogenation of Alkynes
The additon of X2 to alkynes is analogous to the addition of X2 to alkenes. The halogen molecule becomes polarized by the approach of the nucleophilic alkyne. The pi electrons of the alkyne react with the bromine to form a carbon-bromine bond and cyclic halonium ion with halide as the leaving group. The formation of the cyclic halonium ion requires anti-addition of the nucleophilic halide to produce a vicinal dihalide alkene as shown in the reaction below.
Exercise
- Draw the structure, and give the IUPAC name, of the product formed in each of the reactions listed below.
- $\ce{\sf{CH3-C#C-CH3->[\displaystyle{\textrm{1 equiv}}][\displaystyle{\textrm{HCl}}]}}$
- $\ce{\sf{CH3-C#C-CH3->[\displaystyle{\textrm{excess}}][\displaystyle{\textrm{HCl}}]}}$
- $\ce{\sf{CH3-C#C-CH3->[\displaystyle{\textrm{1 equiv}}][\displaystyle{\textrm{Br}_2}]}}$
- $\ce{\sf{CH3-C#C-CH3->[\displaystyle{\textrm{excess}}][\displaystyle{\textrm{Br}_2}]}}$
- $\ce{\sf{CH3CH2-C#C-H->[\displaystyle{\textrm{1 equiv}}][\displaystyle{\textrm{HCl}}]}}$
- $\ce{\sf{CH3CH2-C#C-H->[\displaystyle{\textrm{excess}}][\displaystyle{\textrm{HCl}}]}}$
- $\ce{\sf{CH3CH2CH2-C#C-H->[\displaystyle{\textrm{1 equiv}}][\displaystyle{\textrm{Br}_2}]}}$
- $\ce{\sf{CH3CH2CH2-C#C-H->[\displaystyle{\textrm{excess}}][\displaystyle{\textrm{Br}_2}]}}$
- Answer
-
-
(Z)-2-chloro-2-butene
-
2,2-dichlorobutane
-
(E)-2,3-dibromo-2-butene
-
2,2,3,3-tetrabromobutane
-
2-chloro-1-butene
-
2,2-dichlorobutane
-
(E)-1,2-dibromo-1-pentene
-
1,1,2,2-tetrabromopentane
-
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
Organic Chemistry With a Biological Emphasis by Tim Soderberg (University of Minnesota, Morris)
Jim Clark (Chemguide.co.uk)

