Solutions for Extra Problems for Exam II
- Page ID
- 11295
Problems are found here.
Answers (D-F)
D1. (c) D2. (a) D3. (c) D4. (b) D5. (b) D6. (c) D7. (e) D8. (d) D9. (b) D10. (d) D11. (c)
E1. (e) E2. (c) E3. (d) E4. (c) E5. (b) E6. (a) E7. (b) E8. (b) E9. (d) E10. (d) E11. (e) E12. (c) E13. (b) E14. (b) E15. (e) E16. (c) E17. (a)
F1. (d) F2. (d) F3. (d) F4. (b) F5. (b) F6. (b) F7. (d) F8. (c) F9. (d) F10. (d) F11. (a) F12. (b) F13. (a) F14. (b) F15. (a) F16. (b) F17. (a) F18. (b) F19. (b) F20. (c)
Solutions (G)
G1
(a)
| Formula of Complex | Rewritten Formula (showing the coordinating ligands) |
| Pt(NH3)6Cl4 | [Pt(NH3)6]Cl4 |
| Pt(NH3)5Cl4 | [Pt(NH3)5Cl]Cl3 |
| Pt(NH3)4Cl4 | [Pt(NH3)4Cl2]Cl2 |
| Pt(NH3)3Cl4 | [Pt(NH3)3Cl3]Cl |
| Pt(NH3)2Cl4 | [Pt(NH3)2Cl4] |
(b) and (c):
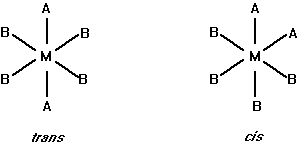
with 4 Cl- as counterions, hexaammineplatinum(IV) chloride
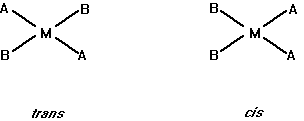
with 3 Cl- as counterions, pentaamminechloroplatinum(IV) chloride
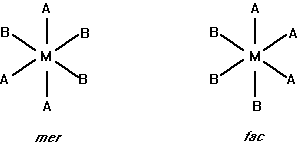
with 2 Cl- as counterions, tetraamminedichloroplatinum(IV) chloride
with 1 Cl- as counterion, triamminetrichloroplatinum(IV) chloride
diamminetetrachloroplatinum(IV)
G2
In Molecular Orbital Theory, the crystal field splitting is related to the difference between the energy of the t2g and eg orbitals. The t2g orbitals are essentially nonbonding and are composed of the dxz, dyz and dxy orbitals. The CO molecule contains empty antibonding p orbitals. These antibonding orbitals have the same symmetry as the t2g orbitals and since they are empty, these additional overlap (see figure below) will lead to a lowering in energy of the t2g orbitals:
The t2g orbitals lower in energy but the eg orbitals (since they do not overlap with the p orbital) remain the same in energy, thus, leading to an increase in the energy separation between the t2g and eg orbitals.
G3
With the two point charges along the +z and -z axis.
___ dz2
___ dxz ___ dyz
___ dxy ___ dx2-y2
G4
(a) Both Br- and SCN- have a charge of negative 1, thus, for the molecule to be neutral, Pt needs a +3 charge. However, this will not be correct since the molecule is diamagnetic. Pt(III) has an odd number of electrons so Pt cannot be diamagnetic if it has a charge of +3. Thus, we will need at least 2 Pt's per molecule. This automatically agrees with the observation that two complex ions are produced. One Pt will be +4, the other +2 (average is then 3). The molecular formula is then Pt2Br2(en)2(SCN)4.
(b) and (c)
In the molecular formula above, from the number of ligands, one can count the number of coordination available for the 2 Pt's. Remember, en is bidentate so each one counts twice. 2 (from the 2Br) + 4 (from 2 en) + 4 (from 4SCN) = 10. With 2 Pt's, this may be 5 for each Pt. A coordination number of 5, however, is not popular among Pt complexes. The more likely solution is that one Pt has a coordination of 4 (therefore, square planar) amd the other Pt having a coordination of 6 (octahedral). To solve this problem further, we need to apply Crystal Field Theory. A coordination number of 4 and a square planar geometry (due to the nature of the splittings of the d orbitals in a square planar field) will be preferred by a d8 central atom. A coordination number of 6 and an octahedral geometry will be preferred by a d6 central atom (provided that the ligands are strong field ligands). Pt(II) is d8 and, thus, will be square planar, Pt(IV) is d6 and, thus, will be octahedral. Pt(II) will be in the complex anion and Pt(IV) will be in the complex cation.
Cation: [Pt(en)2(SCN)2]2+
Anion: [Pt(Br)2(SCN)2]2- (cis- and trans- isomers possible)
(d)
G5
G6
When a solvent is bound as a ligand to a transition metal ion, it loses a great degree of freedom. The degree of randomness or entropy depends heavily on the number of free molecules. When a chelating agent binds to a metal ion, it liberates more than one ligand thereby increasing the number of free molecules and, consequently, the entropy of the system.
G7
(a) Draw all the geometric isomers for an MA2B4 complex.
For complexes that have six ligands, we will assume octahedral geometry.
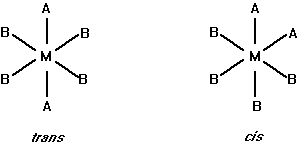
(b) Draw all the geometric isomers for an MA2B2 complex in a planar arrangement. For a tetrahedral arrangement, are geometric isomers possible?
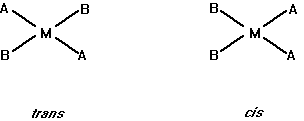
Geometric isomers are not possible in a tetrahedral arrangement because all of the corners of a tetrahedron are adjacent to one another.
(c) Draw all the geometric isomers for an MA3B3 complex.
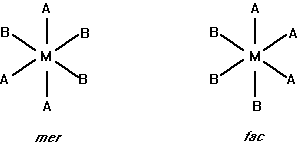
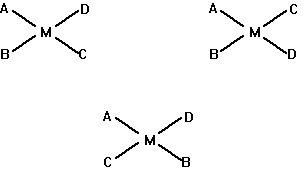
(d) Draw all the geometric isomers for an MABCD complex in a planar arrangement. Are optical isomers possible?

Optical isomers are not possible for square planar complexes because any planar compound will have a mirror plane containing all the atoms and a disymmetric molecule cannot have a mirror plane of symmetry.
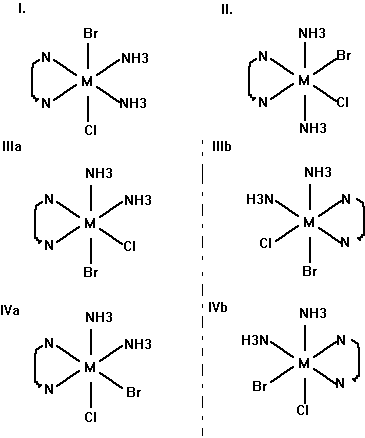
(e) Draw all the geometric isomers of [Cr(en)(NH3)2BrCl]+. Which of these isomers also has an optical isomer? Draw the various isomers.

I, II, IIIa and IVa are geometric isomers; I and II have mirror planes, thus, they do not have optical isomers. IIIa/IIIb and IVa/IVb are the pairs of optical isomers.
G8
H2O is a weak-field ligand, thus, in all the hexaaqua octahedral species of the above ions, the metal is in a high-spin state. Mn3+ is d4, Fe3+ is d5, Co3+ is d6. Only Fe3+ has all its d orbitals half-filled which is a relatively stable electronic configuration. Thus, the reduction potential of the Fe(III) ion is less than its neighbors in the periodic table.
G9
One advantage a tetrahedral arrangement has over a square planar one is space. For this reason, most first row transition metals prefer tetrahedral while the second and third-row transition metals prefer square planar. Square planar is preferred because of the higher splitting of the d orbitals which is advantageous when the metal ion does not have filled d orbitals. The Crystal Field Stabilization Energy (CFSE) derived from the splitting of the d orbital energies is greater for square planar arrangement. In a tetrahedral arrangement, no pair of ligands is pointing at one specific d orbital. Thus, for first-row atoms such as Ni2+ (a d8 species), tetrahedral arrangement will be preferred if the ligands are large and weak-field. If the ligands are small (more rod-like) and are strong-field, the planar arrangement will be preferred. The chloride ion is large and it is a weak-field ligand so tetrachloronickelate(II) is tetrahedral. The cyanide ion is small and it is a strong-field ligand so tetracyanonickelate(II) will probably be square planar.
G10
The process is:
(A) 2H+(aq) + Cu(s) + 4NH3(aq) --> H2(g) + [Cu(NH3)4]2+
E = E0 - RT/nF (ln Q)
Q = 1; E = E0 = 0.08 V
The above process (A) can be written as (I+II+III):
I. 2H+(aq) + 2e- --> H2(g)
II. Cu(s) --> Cu2+(aq) + 2e-
III. Cu2+(aq) + 4NH3(aq) --> [Cu(NH3)4]2+(aq)
The reduction potentials relate to each other:
EA = EI + EII + EIII
EA is known from the experiment, 0.08 V.
EI is 0.00 V (hydrogen reference)
EII is -0.337 V (the negative of the standard reduction potential of Cu(II))
Thus, EIII should be 0.417 V.
E0 = (0.0592/2) log Kf
log Kf = 2 (0.417)/0.0592
Kf = 1.2 x 1014
http://bouman.chem.georgetown.edu/S9...ob10/sol10.htm
www.chem.tamu.edu/class/fyp/mcquest/ch25.html

