Carbanions
- Page ID
- 16932
A carbanion is an anion in which carbon has an unshared pair of electrons and bears a negative charge usually with three substituents for a total of eight valence electrons.[1] The carbanion exists in a trigonal pyramidal geometry. Formally, a carbanion is the conjugate base of a carbon acid.
\[\ce{R_3C-H + B^- \rightarrow R_3C^- + H-B}\]
where B stands for the base. A carbanion is one of several reactive intermediates in organic chemistry.
Theory
A carbanion is a nucleophile, which stability and reactivity determined by several factors:
- The inductive effect. Electronegative atoms adjacent to the charge will stabilize the charge;
- Hybridization of the charge-bearing atom. The greater the s-character of the charge-bearing atom, the more stable the anion;
- The extent of conjugation of the anion. Resonance effects can stabilize the anion. This is especially true when the anion is stabilized as a result of aromaticity.
A carbanion is a reactive intermediate and is encountered in organic chemistry for instance in the E1cB elimination reaction and in organometallic chemistry in for instance a Grignard reaction or in alkyl lithium chemistry. Stable carbanions do however exist. In 1984 Olmstead presented the lithium crown ether salt of the triphenylmethyl carbanion from triphenylmethane, n-butyllithium and 12-crown-4 at low temperatures:[2]
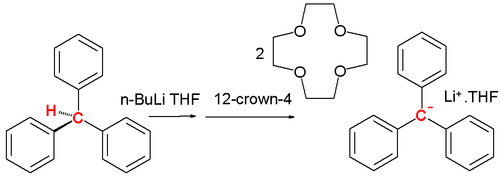
Adding n-butyllithium to triphenylmethane in THF at low temperatures followed by 12-crown-4 results in a red solution and the salt complex precipitates at −20 °C. The central C-C bond lengths are 145 pm with the phenyl ring propelled at an average angle of 31.2°. This propeller shape is less pronounced with a tetramethylammonium counterion.[3] One tool for the detection of carbanions in solution is proton NMR.[4] A spectrum of cyclopentadiene in DMSO shows four vinylic protons at 6.5 ppm and two methylene bridge protons at 3 ppm whereas the cyclopentadienyl anion has a single resonance at 5.50 ppm.
Carbon acids
Any molecule containing a C-H can lose a proton forming the carbanion. Hence any hydrocarbon containing C-H bonds can be considered an acid with a corresponding pKa value. Methane is certainly not an acid in its classical meaning yet its estimated pKa is 56. Compare this to acetic acid with pKa 4.76. The same factors that determine the stability of the carbanion also determine the order in pKa in carbon acids. These values are determined for the compounds either in water in order to compare them to ordinary acids, indimethyl sulfoxide in which the majority of carbon acids and their anions are soluble or in the gas phase. With DMSO the acidity window for solutes is limited to its own pKa of 35.5.
Note that the anions formed by ionization of acetic acid, ethanol or aniline are not carbanions.
Starting from methane in Table 1, the acidity increases:
- when the anion is aromatic, either because the added electron causes the anion to become aromatic (as in indene and cyclopentadiene), or because the negative charge on carbon can be delocalized over several already-aromatic rings (as in triphenylmethane or the carborane superacid).
- when the carbanion is surrounded by strongly electronegative groups, through the partial neutralisation of the negative charge (as in malononitrile).
- when the carbanion is immediately next to a carbonyl group. The α-protons of carbonyl groups are acidic because the negative charge in the enolate can be partially distributed in the oxygen atom. Meldrum's acid and barbituric acid, historically named acids, are in fact a lactone and a lactam respectively, but their acidic carbon protons make them acidic. The acidity of carbonyl compounds is an important driving force in many organic reactions such as the aldol reaction.
Chiral carbanions
With the molecular geometry for a carbanion described as a trigonal pyramid the question is whether or not carbanions can display chirality, because if the activation barrier for inversion of this geometry is too low any attempt at introducing chirality will end inracemization, similar to the nitrogen inversion. However, solid evidence exists that carbanions can indeed be chiral for example in research carried out with certain organolithium compounds.
The first ever evidence for the existence of chiral organolithium compounds was obtained in 1950. Reaction of chiral 2-iodooctane with sec-butyllithium in petroleum ether at −70 °C followed by reaction with dry ice yielded mostly racemic 2-methylbutyric acid but also an amount of optically active 2-methyloctanoic acid which could only have formed from likewise optical active 2-methylheptyllithium with the carbon atom linked to lithium the carbanion:[6]
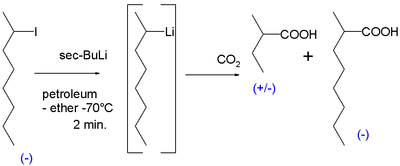
On heating the reaction to 0 °C the optical activity is lost. More evidence followed in the 1960s. A reaction of the cis isomer of 2-methylcyclopropyl bromide with sec-butyllithium again followed by carboxylation with dry ice yielded cis-2-methylcyclopropylcarboxylic acid. The formation of the trans isomer would have indicated that the intermediate carbanion was unstable.[7]
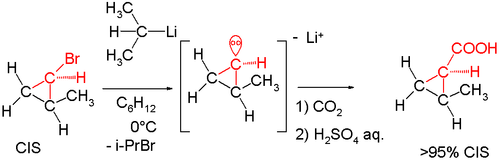
In the same manner the reaction of (+)-(S)-l-bromo-l-methyl-2,2-diphenylcyclopropane with n-butyllithium followed by quench with methanol resulted in product with retention of configuration:[8]

Of recent date are chiral methyllithium compounds:[9]
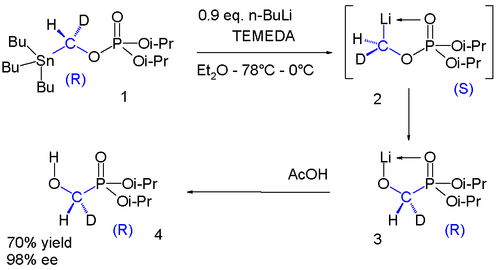
The phosphate 1 contains a chiral group with a hydrogen and a deuterium substituent. The stannyl group is replaced by lithium to intermediate 2 which undergoes a phosphate-phosphorane rearrangement to phosphorane 3 which on reaction with acetic acid givesalcohol 4. Once again in the range of −78 °C to 0 °C the chirality is preserved in this reaction sequence.[10]
A carbanionic structure first made an appearance in the reaction mechanism for the benzoin condensation as correctly proposed by Clarke and Lapworth in 1907.[11] In 1904 Schlenk prepared Ph3C-NMe4+ in a quest for pentavalent nitrogen (fromTetramethylammonium chloride and Ph3CNa) [12] and in 1914 he demonstrated how triarylmethyl radicals could be reduced to carbonions by alkali metals.[13] The phrase carbanion was introduced by Wallis and Adams in 1933 as the negatively charged counterpart of the carbonium ion. [14][15]
External links
- Large database of Bordwell pKa values at www.chem.wisc.edu Link
- Large database of Bordwell pKa values at daecr1.harvard.edu Link
References
- Organic Chemistry - Robert Thornton Morrison, Robert Neilson Boyd
- The isolation and x-ray structures of lithium crown ether salts of the free phenyl carbanions [CHPh2]- and [CPh3]- Marilyn M. Olmstead, Philip P. Power; J. Am. Chem. Soc.; 1985; 107(7); 2174-2175. doi:10.1021/ja00293a059
- Harder, Sjoerd (2002). "Schlenk's Early “Free” Carbanions". Chemistry - A European Journal 8 (14): 3229–3232. doi:10.1002/1521-3765(20020715)8:14<3229::AID-CHEM3229>3.0.CO;2-3.
- A Simple and Convenient Method for Generation and NMR Observation of Stable Carbanions. Hamid S. Kasmai Journal of Chemical Education • Vol. 76 No. 6 June 1999
- Equilibrium acidities in dimethyl sulfoxide solution Frederick G. Bordwell Acc. Chem. Res.; 1988; 21(12) pp 456 - 463; doi:10.1021/ar00156a004
- FORMATION OF OPTICALLY ACTIVE 1-METHYLHEPTYLLITHIUM Robert L. Letsinger J. Am. Chem. Soc.; 1950; 72(10) pp 4842 - 4842; doi:10.1021/ja01166a538
- The Configurational Stability of cis- and trans-2-Methylcyclopropyllithium and Some Observations on the Stereochemistry of their Reactions with Bromine and Carbon Dioxide Douglas E. Applequist and Alan H. Peterson J. Am. Chem. Soc.; 1961; 83(4) pp 862 - 865;doi:10.1021/ja01465a030
- Cyclopropanes. XV. The Optical Stability of 1-Methyl-2,2-diphenylcyclopropyllithium H. M. Walborsky, F. J. Impastato, and A. E. Young J. Am. Chem. Soc.; 1964; 86(16) pp 3283 - 3288; doi:10.1021/ja01070a017
- Preparation of Chiral -Oxy-[2H1]methyllithiums of 99% ee and Determination of Their Configurational Stability Dagmar Kapeller, Roland Barth, Kurt Mereiter, and Friedrich Hammerschmidt J. Am. Chem. Soc.; 2007; 129(4) pp 914 - 923; (Article) doi:10.1021/ja066183s
- Enantioselectivity determined by NMR spectroscopy after derivatization with Mosher's acid
- Clarke, R. W. L.; Lapworth, A. (1907). "LXV.?An extension of the benzoin synthesis". Journal of the Chemical Society, Transactions 91: 694. doi:10.1039/CT9079100694.
- Schlenk, W.; Weickel, T.; Herzenstein, A. (1910). "Ueber Triphenylmethyl und Analoga des Triphenylmethyls in der Biphenylreihe. [Zweite Mittheilung über „Triarylmethyle”.]". Justus Liebig's Annalen der Chemie 372: 1. doi:10.1002/jlac.19103720102.
- Schlenk, W.; Marcus, E. (1914). "Über Metalladditinen an freie organische Radikale. (Über Triarylmethyle. XII.)". Berichte der deutschen chemischen Gesellschaft 47 (2): 1664. doi:10.1002/cber.19140470256.
- Wallis, E. S.; Adams, F. H. (1933). "The Spatial Configuration of the Valences in Tricovalent Carbon Compounds1". Journal of the American Chemical Society 55 (9): 3838. doi:10.1021/ja01336a068.
- Tidwell, T. T. (1997). "The first century of physical organic chemistry: A prologue". Pure and Applied Chemistry 69 (2): 211–214. doi:10.1351/pac199769020211.





















