13.3: The Vibration-Rotation Spectrum
- Page ID
- 63742
Vibration-rotation interaction describes the inversely proportional relationship of the rotational constant and the vibrational state. The rotational constants decrease as the vibrational states increase, and their interaction influences the frequencies at where the lines of R and P branches occurred. This effect can be observed well on a rotational-vibrational spectrum, and the vibration-rotation relationship also has its own mathematical form.
Introduction
When a rotational transition occurs with a vibrational transition, the vibrational-rotational spectrum of R branch and P branch is observed. Experimental data shows that the P branch has closer spaces between the lines as the frequencies decrease while R branch has further spaces between the lines as the frequencies increase. On the spectrum, R region is on the left and P region is on the right separated by a large gap in between. There are other graphs show differently, but the regions can be labeled using their spacing characteristic discussed below.
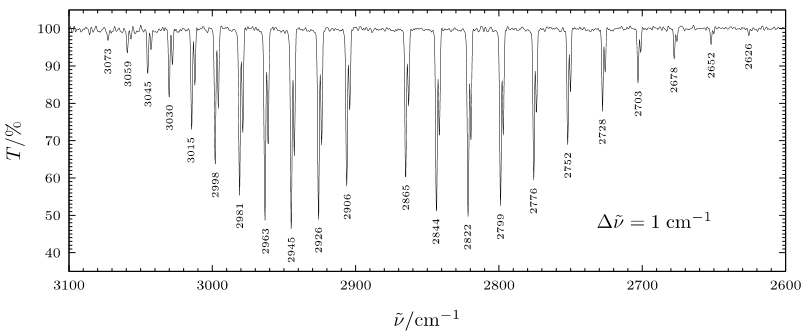
Figure 1: Rotational-Vibrational Spectrum of HCl. Figure used with permission from Wikipedia.
Vibration-Rotation Interaction
Consider the rotational constant for a rigid rotator \(\tilde{B}=h/8\pi^2c\mu R_e^2\) where \(\tilde{B}\) is the rotational constant, and \(R_e^2\) is the bond length. It is clear from the equation that \(\tilde{B}\) depends on \(R_e^2\) in a inverse manner, so \(\tilde{B}\) will decrease as \(R_e^2\) increase, and vice versa.
Vibrational state and the bond length also possess a relationship, and that is bond length, \(R_e^2\), will increase as the vibrational state, \(v\) increases. High vibrational states have large vibrational amplitudes (the amplitude can be visualized as the distance from one side to the other on the harmonic-oscillator parabola, and higher states has greater distance). On a potential graph, imagine a diatomic molecule with one of the atom fixed at the origin, the bond is along the x-axis, and the molecule vibrate toward and away from the origin along x-axis. The range at how much the bond can stretch or squeeze depend on the vibrational amplitude. So, at high vibrational state (thus high amplitude), the bond can squeeze in and stretch out more (just a little more) on the regular harmonic-oscillator potential, but it can stretch out greatly on an anharmonic oscillator. That's why bond length increase with vibrational state.
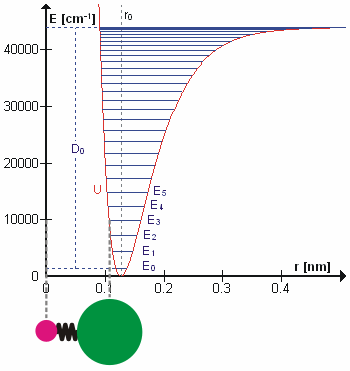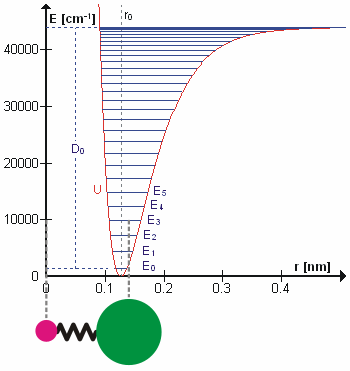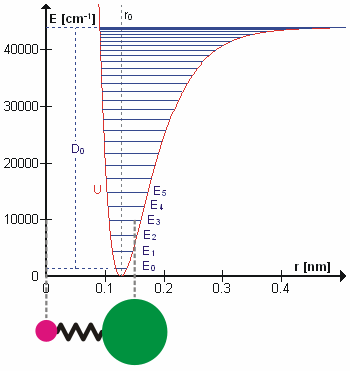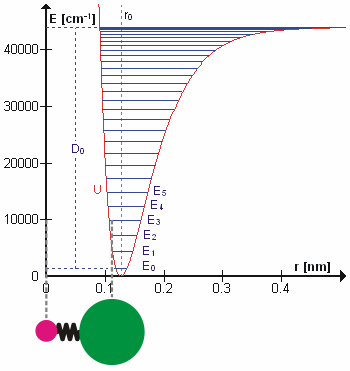
Figure 2: Anharmonic Oscillator and Bond Length. Figure used with permission from Wikipedia.
So far, there are two relationship established above
\(\tilde{B} \propto R_e^2\) (inversely proportional)
and
\(R_e^2 \propto v\) (directly proportional)
Therefore,
\(\tilde{B} \propto v\) (inversely proportional)
The rotational constant dependent on the vibrational state is denoted as \(\tilde{B}_v\), and the dependence of \(\tilde{B}\) on \(v\) is the vibrational-rotational interaction.
Frequencies of R and P Branches
The energies of the rigid rotator-harmonic oscillator is
\[\tilde{E}_{v, J} = G(v) + F(J) = \tilde{\nu} (v+\dfrac{1}{2}) + \tilde{B} J (J+1)\]
or in term of \(\tilde{B}_v\)
\[\tilde{E}_{v, J} = \tilde{\nu} (v+\dfrac{1}{2}) + \tilde{B}_v J (J+1)\]
Frequencies of vibrational transition from \(v = 0 \rightarrow 1\) can be calculated from the equation above as the following:
For R branch, allowed \(J = 0, 1, 2, ...\)
\[\tilde{\nu}_R(\Delta J = +1) = \tilde{E}_{1, J+1} - \tilde{E}_{0, J}\]
\[= \dfrac{3}{2} \tilde{\nu} + \tilde{B}_1 (J+1)(J+2) - \dfrac{1}{2} \tilde{\nu} - \tilde{B}_0 J(J+1)\]
\[= \tilde{\nu}+2\tilde{B}_1+(3\tilde{B}_1-\tilde{B}_0)J+(\tilde{B}_1-\tilde{B}_0)J^2\]
For P branch, allowed \(J = 1, 2, 3, ...\)
\[\tilde{\nu}_P(\Delta J = -1) = \tilde{E}_{1, J-1} - \tilde{E}_{0, J}\]
\[= \tilde{\nu}-(\tilde{B}_1+\tilde{B}_0)J+(\tilde{B}_1-\tilde{B}_0)J^2\]
According to the vibration-rotation interaction, \(\tilde{B}_1 < \tilde{B}_0\) because \(R_e^2 (v=1) > R_e^2 (v=0)\).
Decrease in Spacing of Lines in the R Branch with Increasing J
A closer look at
\[\tilde{\nu}_R = \tilde{\nu}+2\tilde{B}_1+(3\tilde{B}_1-\tilde{B}_0)J+(\tilde{B}_1-\tilde{B}_0)J^2\]
shows that the last term in the parentheses \((\tilde{B}_1-\tilde{B}_0)\) will be always negative because \(\tilde{B}_1 < \tilde{B}_0\), and it also multiplies with \(J^2\), so the square term \((\tilde{B}_1-\tilde{B}_0)J^2\) will give a larger negative value for increasing \(J\). As a result, \(\tilde{\nu}_R=\tilde{\nu} + \)(smaller and smaller value) as J increase. Because the rotational frequencies keep getting smaller, the spacing between lines in R branch decreases as J increases.
Increasing in Spacing of Lines in the P Branch with Decreasing J
Using the same analysis as above,
\[\tilde{\nu}_P = \tilde{\nu}-(\tilde{B}_1+\tilde{B}_0)J+(\tilde{B}_1-\tilde{B}_0)J^2\]
shows that the last term in the parentheses \((\tilde{B}_1-\tilde{B}_0)\) will be always negative because \(\tilde{B}_1 < \tilde{B}_0\), and it also multiplies with \(J^2\), so the square term \((\tilde{B}_1-\tilde{B}_0)J^2\) will give a larger negative value even for decreasing \(J\). As a result, \(\tilde{\nu}_R=\tilde{\nu} - \)(smaller and smaller value) as J decreases. Because the rotational frequencies keep getting larger, the spacing between lines in P branch increase as J decreases.
References
- McQuarrie, Donald A. Quantum Chemistry. New York: University Science Books, 2007.
Problems
- Why does the bond length increase as the vibrational state increases? Answer
- Explain the decrease in line spacing for the R region as J increase. Answer
- Explain the increase in line spacing for the P region as J decrease. Answer
- Despite being parts of the same spectrum, why do P region and R region have different line spacing characteristics as discussed above. (Hint: Are their spacing characteristics really different?)
- Given the relationship
\[\tilde{B}_v=\tilde{B}_e-\tilde{\alpha}(v=\dfrac{1}{2})\]
What type of graph is expected for the graph of \(\tilde{B}_v\) vs. \(v"\)?
Answers
- Above
- Above
- Above
- The spacing characteristics of R and P are exactly the same because the spacing always decreases as J increase regardless of the region. Spacing in P increases only because we look at it backwardly as J decreases.
- The equation should give a linear relationship, so the graph is a straight line with a negative slope.
Contributors
- Dayton Dang

