8.7: Enthalpy- A Measure of the Heat Evolved or Absorbed in a Reaction
- Page ID
- 98045
When physical or chemical changes occur, they are generally accompanied by a transfer of energy. The law of conservation of energy states that in any physical or chemical process, energy is neither created nor destroyed. In other words, the entire energy in the universe is conserved. In order to better understand the energy changes taking place during a reaction, we need to define two parts of the universe, called the system and the surroundings. The system is the specific portion of matter in a given space that is being studied during an experiment or an observation. The surroundings are everything in the universe that is not part of the system. In practical terms for a laboratory chemist, the system is the particular chemicals being reacted, while the surroundings is the immediate vicinity within the room. During most processes, energy is exchanged between the system and the surroundings. If the system loses a certain amount of energy, that same amount of energy is gained by the surroundings. If the system gains a certain amount of energy, that energy is supplied by the surroundings.
A chemical reaction or physical change is endothermic if heat is absorbed by the system from the surroundings. In the course of an endothermic process, the system gains heat from the surroundings and so the temperature of the surroundings decreases. The quantity of heat for a process is represented by the letter \(q\). The sign of \(q\) for an endothermic process is positive because the system is gaining heat. A chemical reaction or physical change is exothermic if heat is released by the system into the surroundings. Because the surroundings are gaining heat from the system, the temperature of the surroundings increases. The sign of \(q\) for an exothermic process is negative because the system is losing heat.
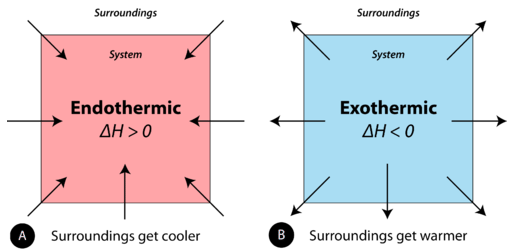
Enthalpy
Heat changes in chemical reactions are often measured in the laboratory under conditions in which the reacting system is open to the atmosphere. In that case, the system is at a constant pressure. Enthalpy \(\left( H \right)\) is the heat content of a system at constant pressure. Chemists routinely measure changes in enthalpy of chemical systems as reactants are converted into products. The heat that is absorbed or released by a reaction at constant pressure is the same as the enthalpy change, and is given the symbol \(\Delta H\). Unless otherwise specified, all reactions in this material are assumed to take place at constant pressure.
The change in enthalpy of a reaction is a measure of the differences in enthalpy of the reactants and products. The enthalpy of a system is determined by the energies needed to break chemical bonds and the energies needed to form chemical bonds. Energy needs to be put into the system in order to break chemical bonds, as they do not come apart spontaneously in most cases. Bond formation to produce products will involve release of energy. The change in enthalpy shows the trade-offs made in these two processes. Does it take more energy to break bonds than that needed to form bonds? If so, the reaction is endothermic and the enthalpy change is positive. If more energy is produced in bond formation than that needed for bond breaking, the reaction is exothermic and the enthalpy is negative.
Several factors influence the enthalpy of a system. Enthalpy is an extensive property, determined in part by the amount of material we work with. The state of reactants and products (solid, liquid, or gas) influences the enthalpy value for a system. The direction of the reaction affects the enthalpy value. A reaction that takes place in the opposite direction has the same numerical enthalpy value, but the opposite sign.
Thermochemical Equation
When methane gas is combusted, heat is released, making the reaction exothermic. Specifically, the combustion of \(1 \: \text{mol}\) of methane releases 890.4 kilojoules of heat energy. This information can be shown as part of the balanced equation:
\[\ce{CH_4} \left( g \right) + 2 \ce{O_2} \left( g \right) \rightarrow \ce{CO_2} \left( g \right) + 2 \ce{H_2O} \left( l \right) + 890.4 \: \text{kJ}\nonumber \]
The equation tells us that \(1 \: \text{mol}\) of methane combines with \(2 \: \text{mol}\) of oxygen to produce \(1 \: \text{mol}\) of carbon dioxide and \(2 \: \text{mol}\) of water. In the process, \(890.4 \: \text{kJ}\) is released and so it is written as a product of the reaction. A thermochemical equation is a chemical equation that includes the enthalpy change of the reaction. The process in the above thermochemical equation can be shown visually in Figure \(\PageIndex{2}\).
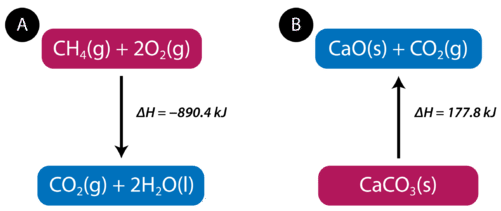
In the combustion of methane example, the enthalpy change is negative because heat is being released by the system. Therefore, the overall enthalpy of the system decreases. The heat of reaction is the enthalpy change for a chemical reaction. In the case above, the heat of reaction is \(-890.4 \: \text{kJ}\). The thermochemical reaction can also be written in this way:
\[\ce{CH_4} \left( g \right) + 2 \ce{O_2} \left( g \right) \rightarrow \ce{CO_2} \left( g \right) + 2 \ce{H_2O} \left( l \right) \: \: \: \: \: \Delta H = -890.4 \: \text{kJ}\nonumber \]
Heats of reaction are typically measured in kilojoules. It is important to include the physical states of the reactants and products in a thermochemical equation as the value of the \(\Delta H\) depends on those states.
Endothermic reactions absorb energy from the surroundings as the reaction occurs. When \(1 \: \text{mol}\) of calcium carbonate decomposes into \(1 \: \text{mol}\) of calcium oxide and \(1 \: \text{mol}\) of carbon dioxide, \(177.8 \: \text{kJ}\) of heat is absorbed. The process is shown visually in Figure \(\PageIndex{2B}\). The thermochemical reaction is shown below.
\[\ce{CaCO_3} \left( s \right) + 177.8 \: \text{kJ} \rightarrow \ce{CaO} \left( s \right) + \ce{CO_2} \left( g \right)\nonumber \]
Because the heat is absorbed by the system, the \(177.8 \: \text{kJ}\) is written as a reactant. The heat of reaction is positive for an endothermic reaction.
\[\ce{CaCO_3} \left( s \right) \rightarrow \ce{CaO} \left( s \right) + \ce{CO_2} \left( g \right) \: \: \: \: \: \Delta H = 177.8 \: \text{kJ}\nonumber \]
The way in which a reaction is written influences the value of the enthalpy change for the reaction. Many reactions are reversible, meaning that the product(s) of the reaction are capable of combining and reforming the reactant(s). If a reaction is written in the reverse direction, the sign of the \(\Delta H\) changes. For example, we can write an equation for the reaction of calcium oxide with carbon dioxide to form calcium carbonate.
\[\ce{CaO} \left( s \right) + \ce{CO_2} \left( g \right) \rightarrow \ce{CaCO_3} \left( s \right) + 177.8 \: \text{kJ}\nonumber \]
The reaction is exothermic and thus the sign of the enthalpy change is negative.
\[\ce{CaO} \left( s \right) + \ce{CO_2} \left( g \right) \rightarrow \ce{CaCO_3} \left( s \right) \: \: \: \: \: \Delta H = -177.8 \: \text{kJ}\nonumber \]
Stoichiometric Calculations and Enthalpy Changes
Chemistry problems that involve enthalpy changes can be solved by techniques similar to stoichiometry problems. Refer again to the combustion reaction of methane. Since the reaction of \(1 \: \text{mol}\) of methane released \(890.4 \: \text{kJ}\), the reaction of \(2 \: \text{mol}\) of methane would release \(2 \times 890.4 \: \text{kJ} = 1781 \: \text{kJ}\). The reaction of \(0.5 \: \text{mol}\) of methane would release \(\dfrac{890,4 \: \text{kJ}}{2} = 445.2 \: \text{kJ}\). As with other stoichiometry problems, the moles of a reactant or product can be linked to mass or volume.
Sulfur dioxide gas reacts with oxygen to form sulfur trioxide in an exothermic reaction, according to the following thermochemical equation.
\[2 \ce{SO_2} \left( g \right) + \ce{O_2} \left( g \right) \rightarrow 2 \ce{SO_3} \left( g \right) + 198 \: \text{kJ} \nonumber \nonumber \]
Calculate the enthalpy change that occurs when \(58.0 \: \text{g}\) of sulfur dioxide is reacted with excess oxygen.
Solution
Step 1: List the known quantities and plan the problem.
- Mass \(\ce{SO_2} = 58.0 \: \text{g}\)
- Molar mass \(\ce{SO_2} = 64.07 \: \text{g/mol}\)
- \(\Delta H = -198 \: \text{kJ}\) for the reaction of \(2 \: \text{mol} \: \ce{SO_2}\)
Unknown
- \(\Delta H = ? \: \text{kJ}\)
The calculation requires two steps. The mass of \(\ce{SO_2}\) is converted to moles. Then the moles of \(\ce{SO_2}\) is multiplied by the conversion factor of \(\left( \dfrac{-198 \: \text{kJ}}{2 \: \text{mol} \: \ce{SO_2}} \right)\).
Step 2: Solve.
\[\Delta H = 58.0 \: \text{g} \: \ce{SO_2} \times \dfrac{1 \: \text{mol} \: \ce{SO_2}}{64.07 \: \text{g} \: \ce{SO_2}} \times \dfrac{-198 \: \text{kJ}}{2 \: \text{mol} \: \ce{SO_2}} = 89.6 \: \text{kJ} \nonumber \nonumber \]
Step 3: Think about your result.
The mass of sulfur dioxide is slightly less than \(1 \: \text{mol}\). Since \(198 \: \text{kJ}\) is released for every \(2 \: \text{mol}\) of \(\ce{SO_2}\) that reacts, the heat released when about \(1 \: \text{mol}\) reacts is one half of 198. The \(89.6 \: \text{kJ}\) is slightly less than half of 198. The sign of \(\Delta H\) is negative because the reaction is exothermic.

