7.1: The Discovery of Atomic Structure
- Page ID
- 98624
Skills to Develop
-
To become familiar with the experimental observations that led to the modern view of the structure of the atom.
Up to this point, we have been describing chemical reactions and solving chemical problems while making many assumptions about the underlying structure of matter. We have said that all matter is made of atoms; that atoms are composed of protons, neutrons, and electrons; that atoms can gain or lose electrons to form ions, or share electrons to form covalent bonds; that breaking chemical bonds requires energy while forming bonds releases energy. Hopefully you do believe these things. Certainly, the fact that we have been able to make sense of many of the features of chemical reactions by assuming these things lends some credence to them. However, you may still be left with questions. How do we know that these tiny atoms are composed of even tinier protons, neutrons, and electrons? Why do different atoms have different physical and chemical properties? Why are metals more likely to form cations and nonmetals to form anions? Why do different types of bonding exist, and why do certain compounds have one type or another? Why does breaking chemical bonds require energy, and why do some bonds require more energy to break than others? In order to answer these questions, we need a deeper understanding of the nature of atoms.
In the following chapter, we will begin to delve into the modern view of atomic structure by first examining the progression of our understanding of the atom from the late 19th century onward. Although you do not need to worry about memorizing historical details, we take this historical approach in order to highlight some of the key experimental evidence that support the modern theory of atoms that underpins all of chemistry. Many aspects of this theory are highly mathematical and difficult to grasp if one does not have an understanding of these key experiments and historical models. Finally, the story of the discovery of atomic structure serves as an excellent exemplar of the scientific method being put into practice, and a tribute to the many chemists and physicists who dedicated their work to this mystery.
The Discovery of the Electron
By the turn of the 20th century, most chemists and physicists were convinced of the real existence of atoms as the building blocks of matter (although truly definitive experimental evidence of this was not available until the early 1900s). Dalton's atomic theory had proved very capable at making sense of the quantitative laws of matter and the emerging science of organic chemistry that was developing. However, until that time, no one had suspected that atoms might themselves be composed of even smaller particles – in other words, that atoms are not the smallest indivisible components of matter.
Long before the end of the 19th century, it was well known that applying a high voltage to a gas contained at low pressure in a sealed tube (called a gas discharge tube) caused electricity to flow through the gas, which then emitted light (Figure \(\PageIndex{1}\)). Researchers trying to understand this phenomenon found that an unusual form of energy was also emitted from the cathode, or negatively charged electrode; this form of energy was called a cathode ray.
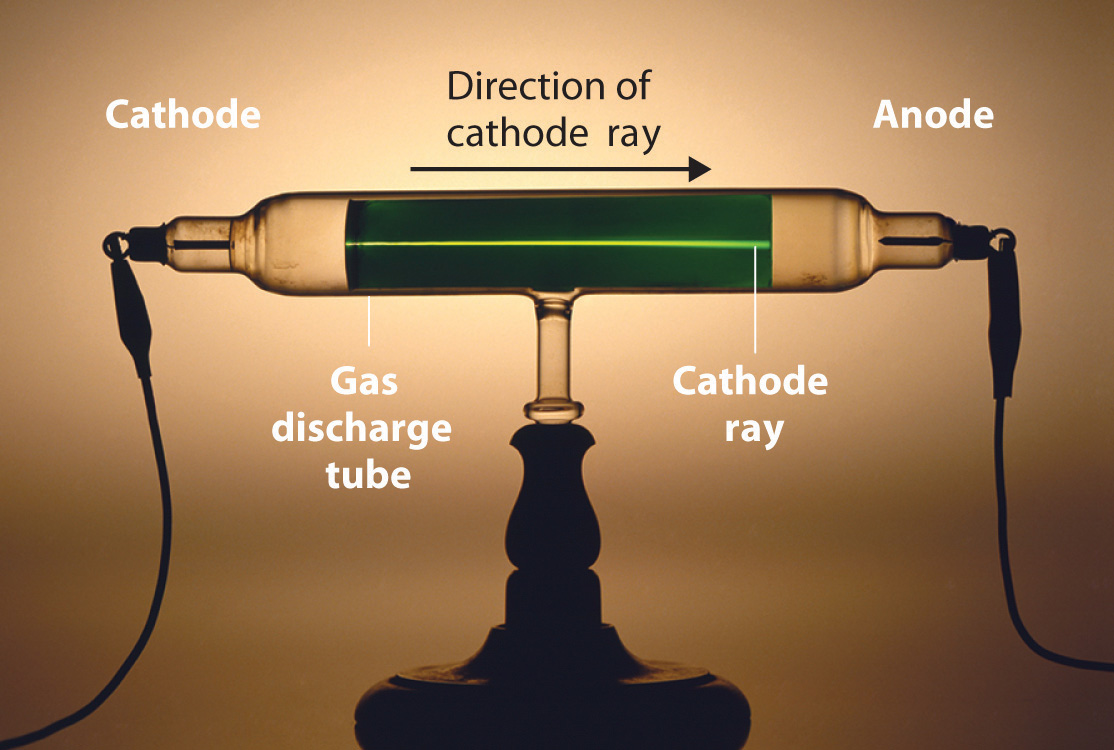
Figure \(\PageIndex{1}\): A Gas Discharge Tube Producing Cathode Rays. When a high voltage is applied to a gas contained at low pressure in a gas discharge tube, electricity flows through the gas, and energy is emitted in the form of light.
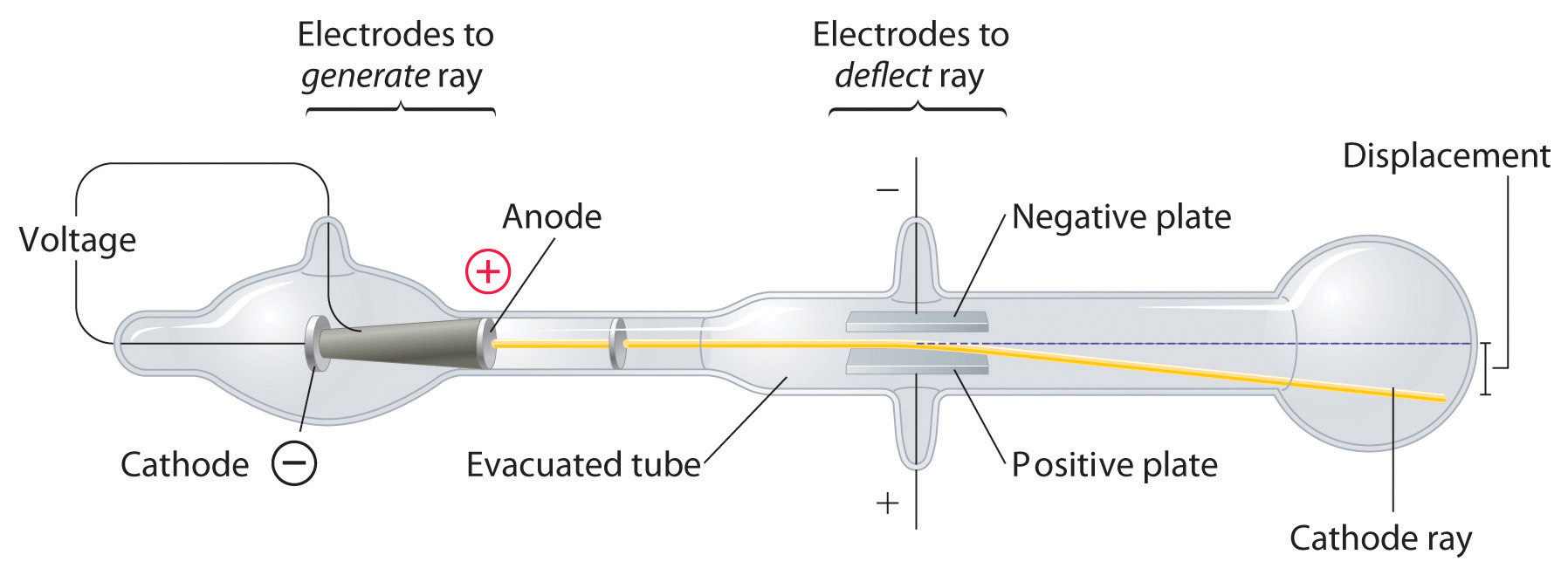
In 1897, the British physicist J. J. Thomson (1856–1940) demonstrated that cathode rays could be deflected, or bent, by magnetic or electric fields, which indicated that cathode rays consist of charged particles (Figure \(\PageIndex{2}\)). More importantly, by measuring the extent of the deflection of the cathode rays in magnetic or electric fields of various strengths, Thomson was able to calculate the mass-to-charge ratio of the particles. These particles were emitted by the negatively charged cathode and repelled by the negative terminal of an electric field. Because like charges repel each other and opposite charges attract, Thomson concluded that the particles had a net negative charge; these particles are now called electrons. Most relevant to the field of chemistry, Thomson found that the mass-to-charge ratio of cathode rays is independent of the nature of the metal electrodes or the gas, which suggested that electrons were fundamental components of all matter.
Figure \(\PageIndex{2}\): Deflection of Cathode Rays by an Electric Field. As the cathode rays travel toward the right, they are deflected toward the positive electrode (+), demonstrating that they are negatively charged.
The video below shows how J. J. Thompson used a cathode ray tube to measure the ratio of charge over mass of an electron.
Video \(\PageIndex{1}\): Measuring e/m For an Electron. Video from Davidson College demonstrating Thompson's e/m experiment.
Subsequently, the American scientist Robert Millikan (1868–1953) carried out a series of experiments using electrically charged oil droplets, which allowed him to calculate the charge on a single electron. With this information and Thomson’s mass-to-charge ratio, Millikan determined the mass of an electron:
\[\dfrac {mass}{charge} \times {charge} ={mass}\]
The mass of the electron calculated by this method was incredibly small – more than 1000 times smaller than the smallest known atom, hydrogen! Together, these experiments proved that not only are atoms not the smallest units of matter, but that atoms themselves are composed of even smaller subatomic particles including electrons.
It was at this point that two separate lines of investigation began to converge, both aimed at determining how and why matter emits energy.
Radioactivity
The second line of investigation began in 1896, when the French physicist Henri Becquerel (1852–1908) discovered that certain minerals, such as uranium salts, emitted a new form of energy. Becquerel’s work was greatly extended by Marie Curie (1867–1934) and her husband, Pierre (1854–1906); all three shared the Nobel Prize in Physics in 1903. Marie Curie coined the term radioactivity (from the Latin radius, meaning “ray”) to describe the emission of energy rays by matter. She found that one particular uranium ore, pitchblende, was substantially more radioactive than most, which suggested that it contained one or more highly radioactive impurities. Starting with several tons of pitchblende, the Curies isolated two new radioactive elements after months of work: polonium, which was named for Marie’s native Poland, and radium, which was named for its intense radioactivity. Pierre Curie carried a vial of radium in his coat pocket to demonstrate its greenish glow, a habit that caused him to become ill from radiation poisoning well before he was run over by a horse-drawn wagon and killed instantly in 1906. Marie Curie, in turn, died of what was almost certainly radiation poisoning.
Figure \(\PageIndex{3}\): Radium bromide illuminated by its own radioactive glow. This 1922 photo was taken in the dark in the Curie laboratory.
Building on the Curies’ work, the British physicist Ernest Rutherford (1871–1937) performed decisive experiments that led to the modern view of the structure of the atom. While working in Thomson’s laboratory shortly after Thomson discovered the electron, Rutherford showed that compounds of uranium and other elements emitted at least two distinct types of radiation. One was readily absorbed by matter and seemed to consist of particles that had a positive charge and were massive compared to electrons. Because it was the first kind of radiation to be discovered, Rutherford called these substances α particles. Rutherford also showed that the particles in the second type of radiation, β particles, had the same charge and mass-to-charge ratio as Thomson’s electrons; they are now known to be high-speed electrons. A third type of radiation, γ rays, was discovered somewhat later and found to be similar to the lower-energy form of radiation called x-rays, now used to produce images of bones and teeth.
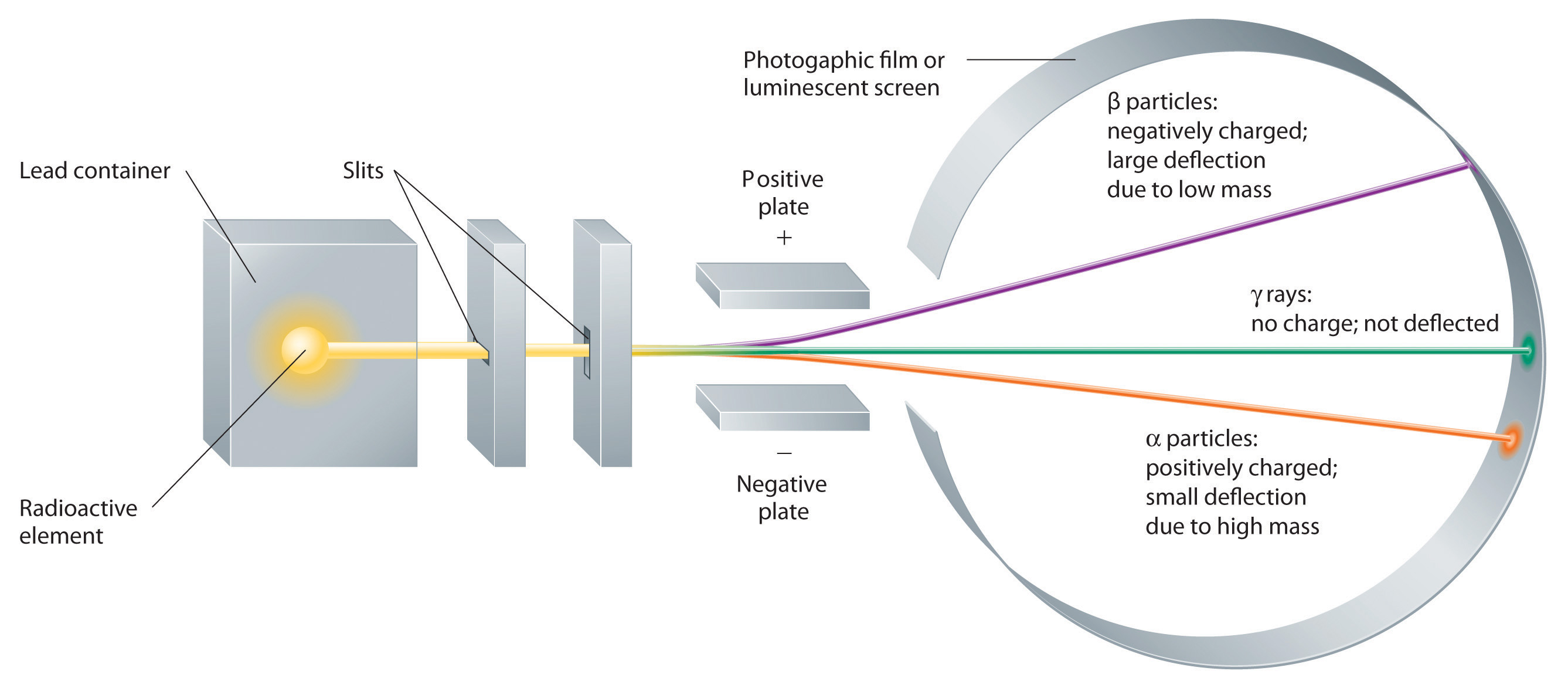
Figure \(\PageIndex{4}\): Effect of an Electric Field on α Particles, β Particles, and γ Rays. A negative electrode deflects negatively charged β particles, whereas a positive electrode deflects positively charged α particles. Uncharged γ rays are unaffected by an electric field. (Relative deflections are not shown to scale.)
These three kinds of radiation—α particles, β particles, and γ rays—are readily distinguished by the way they are deflected by an electric field and by the degree to which they penetrate matter. As Figure \(\PageIndex{3}\) illustrates, α particles and β particles are deflected in opposite directions; α particles are deflected to a much lesser extent because of their higher mass-to-charge ratio. In contrast, γ rays have no charge, so they are not deflected by electric or magnetic fields. Figure \(\PageIndex{5}\) shows that α particles have the least penetrating power and are stopped by a sheet of paper, whereas β particles can pass through thin sheets of metal but are absorbed by lead foil or even thick glass. In contrast, γ-rays can readily penetrate matter; thick blocks of lead or concrete are needed to stop them.
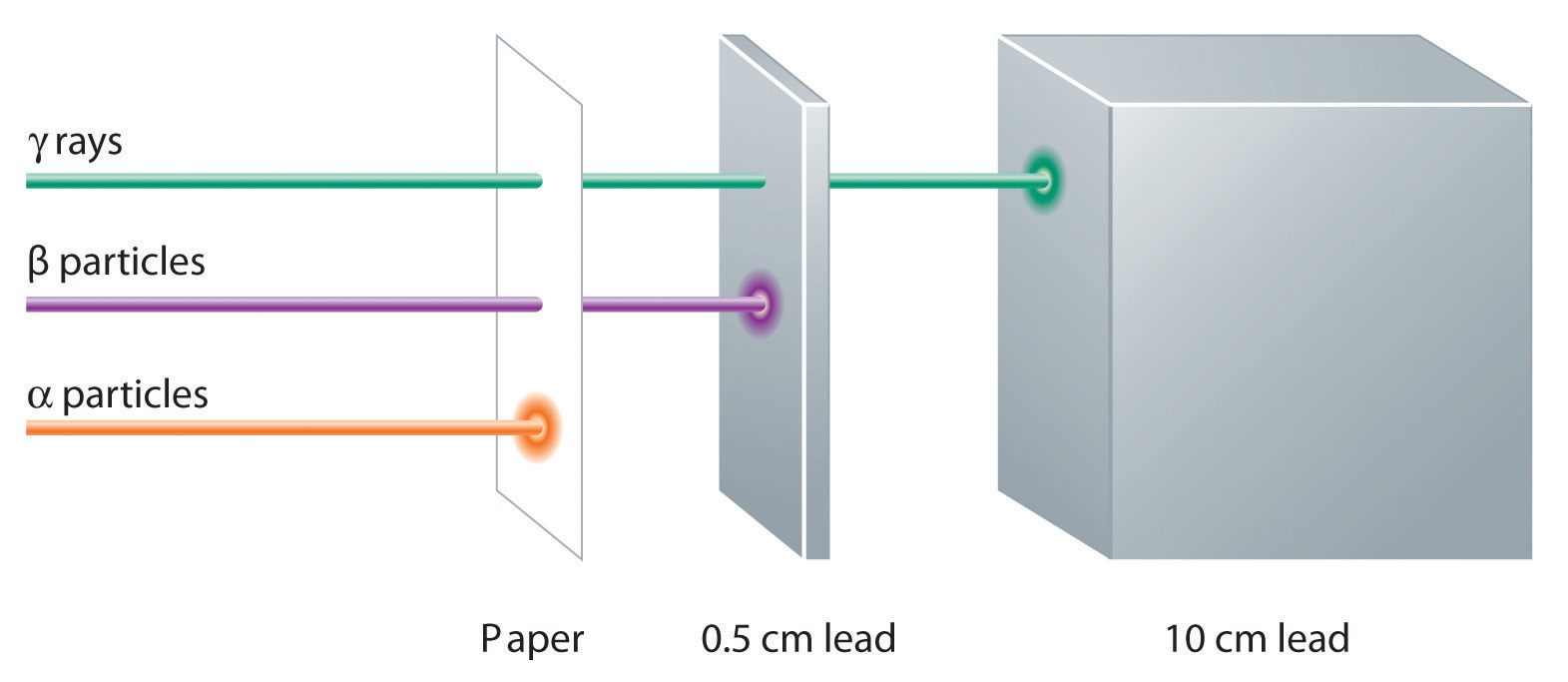
Figure \(\PageIndex{5}\): Relative Penetrating Power of the Three Types of Radiation. A sheet of paper stops comparatively massive α particles, whereas β particles easily penetrate paper but are stopped by a thin piece of lead foil. Uncharged γ rays penetrate the paper and lead foil; a much thicker piece of lead or concrete is needed to absorb them.
The Atomic Model
Once scientists concluded that all matter contains negatively charged electrons, it became clear that atoms, which are electrically neutral, must also contain positive charges to balance the negative ones. Thomson proposed that the electrons were embedded in a uniform sphere that contained both the positive charge and most of the mass of the atom, much like raisins in plum pudding or chocolate chips in a cookie (Figure \(\PageIndex{6}\)).
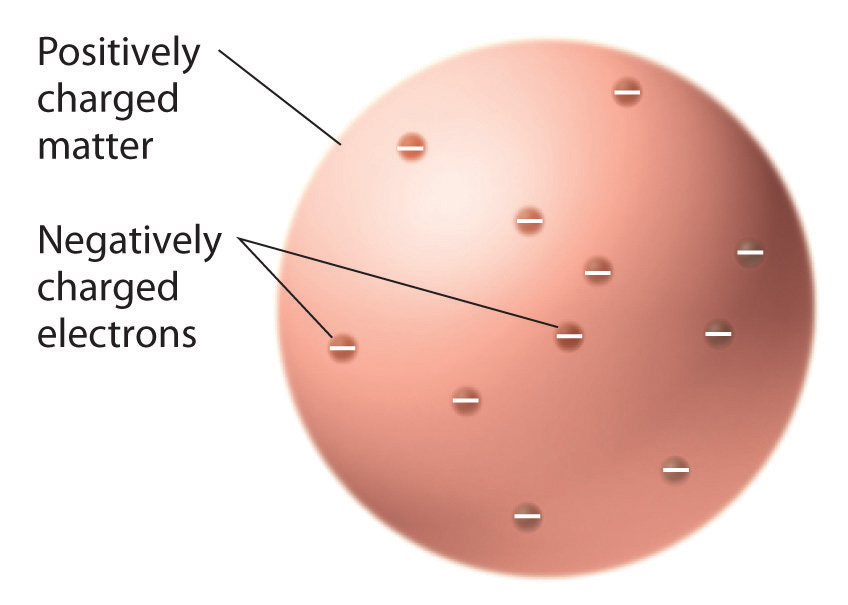
Figure \(\PageIndex{6}\): Thomson’s Plum Pudding Model of the Atom. In this model, the electrons are embedded in a uniform sphere of positive charge.
In a single famous experiment, however, Rutherford showed unambiguously that Thomson’s model of the atom was incorrect. Rutherford aimed a stream of α particles at a very thin gold foil target (Figure \(\PageIndex{7a}\)) and examined how the α particles were scattered by the foil. Gold was chosen because it could be easily hammered into extremely thin sheets, minimizing the number of atoms in the target. If Thomson’s model of the atom were correct, the positively-charged α particles should crash through the uniformly distributed mass of the gold target like cannonballs through the side of a wooden house. They might be moving a little slower when they emerged, but they should pass essentially straight through the target (Figure \(\PageIndex{7b}\)). To Rutherford’s amazement, a small fraction of the α particles were deflected at large angles, and some were reflected directly back at the source (Figure \(\PageIndex{7c}\)). According to Rutherford, “It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you.”
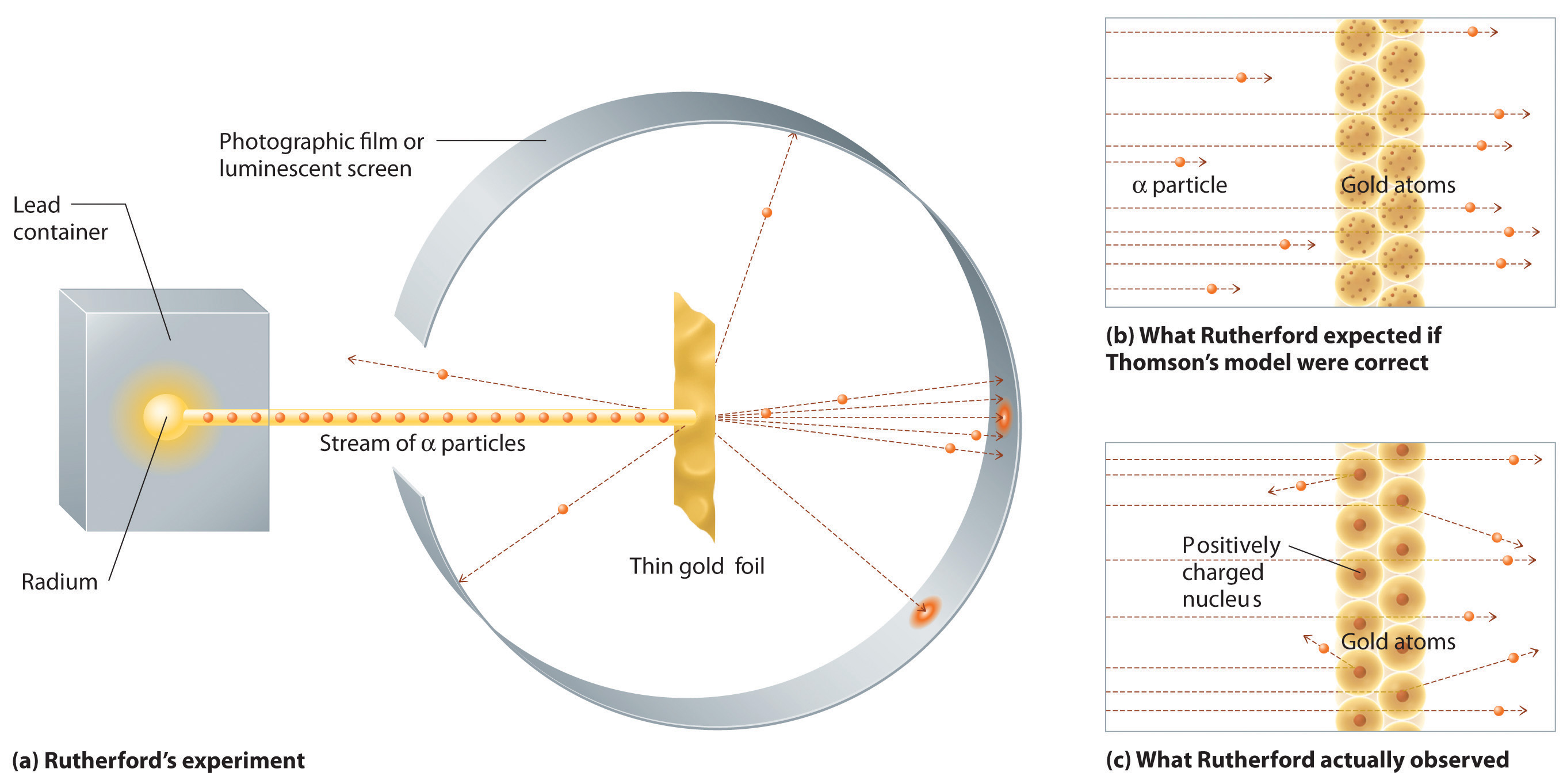
Figure \(\PageIndex{7}\): A Summary of Rutherford’s Experiments. (a) A representation of the apparatus Rutherford used to detect deflections in a stream of α particles aimed at a thin gold foil target. The particles were produced by a sample of radium. (b) If Thomson’s model of the atom were correct, the α particles should have passed straight through the gold foil. (c) However, a small number of α particles were deflected in various directions, including right back at the source. This could be true only if the positive charge were much more massive than the α particle. It suggested that the mass of the gold atom is concentrated in a very small region of space, which he called the nucleus.
Rutherford’s results were not consistent with a model in which the mass and positive charge are distributed uniformly throughout the volume of an atom. Instead, they strongly suggested that both the mass and positive charge are concentrated in a tiny fraction of the volume of an atom, which Rutherford called the nucleus. It made sense that a small fraction of the α particles collided with the dense, positively charged nuclei in either a glancing fashion, resulting in large deflections, or almost head-on, causing them to be reflected straight back at the source. For this and other insights, Rutherford was awarded the Nobel Prize in Chemistry in 1908.
Although Rutherford could not explain why repulsions between the positive charges in nuclei that contained more than one positive charge did not cause the nucleus to disintegrate, he reasoned that repulsions between negatively charged electrons would cause the electrons to be uniformly distributed throughout the atom’s volume. Today it is known that strong nuclear forces, which are much stronger than electrostatic interactions, hold the protons and the neutrons together in the nucleus.
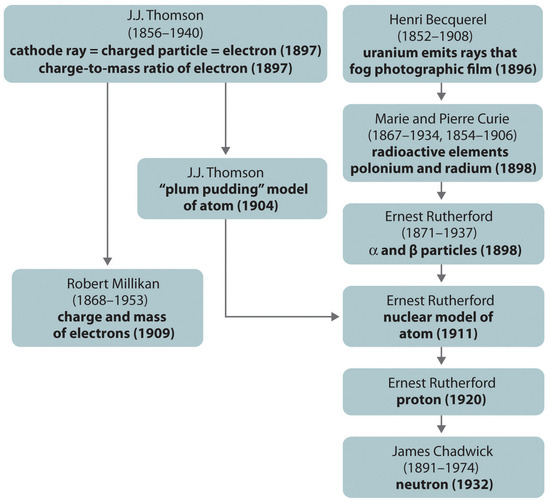
Figure \(\PageIndex{8}\): A Summary of the Historical Development of Models of the Components and Structure of the Atom. The dates in parentheses are the years in which the key experiments were performed.
The historical development of the different models of the atom’s structure is summarized in Figure \(\PageIndex{8}\). Rutherford established that the nucleus of the hydrogen atom was a positively charged particle, for which he coined the name proton in 1920. He also suggested that the nuclei of elements other than hydrogen must contain electrically neutral particles with approximately the same mass as the proton. The neutron, however, was not discovered until 1932, when James Chadwick (1891–1974, a student of Rutherford; Nobel Prize in Physics, 1935) discovered it. As a result of Rutherford’s work, it became clear that an α particle contains two protons and neutrons, and is therefore the nucleus of a helium atom.
Rutherford’s model of the atom is essentially the same as the modern model, except that it is now known that electrons are not uniformly distributed throughout an atom’s volume. Instead, they are distributed according to a set of principles described by quantum mechanics. Figure \(\PageIndex{9}\) shows how the model of the atom has evolved over time from the indivisible unit of Dalton to the modern view taught today. In the following sections, we will examine how the quantum mechanical model of the atom came to be developed.
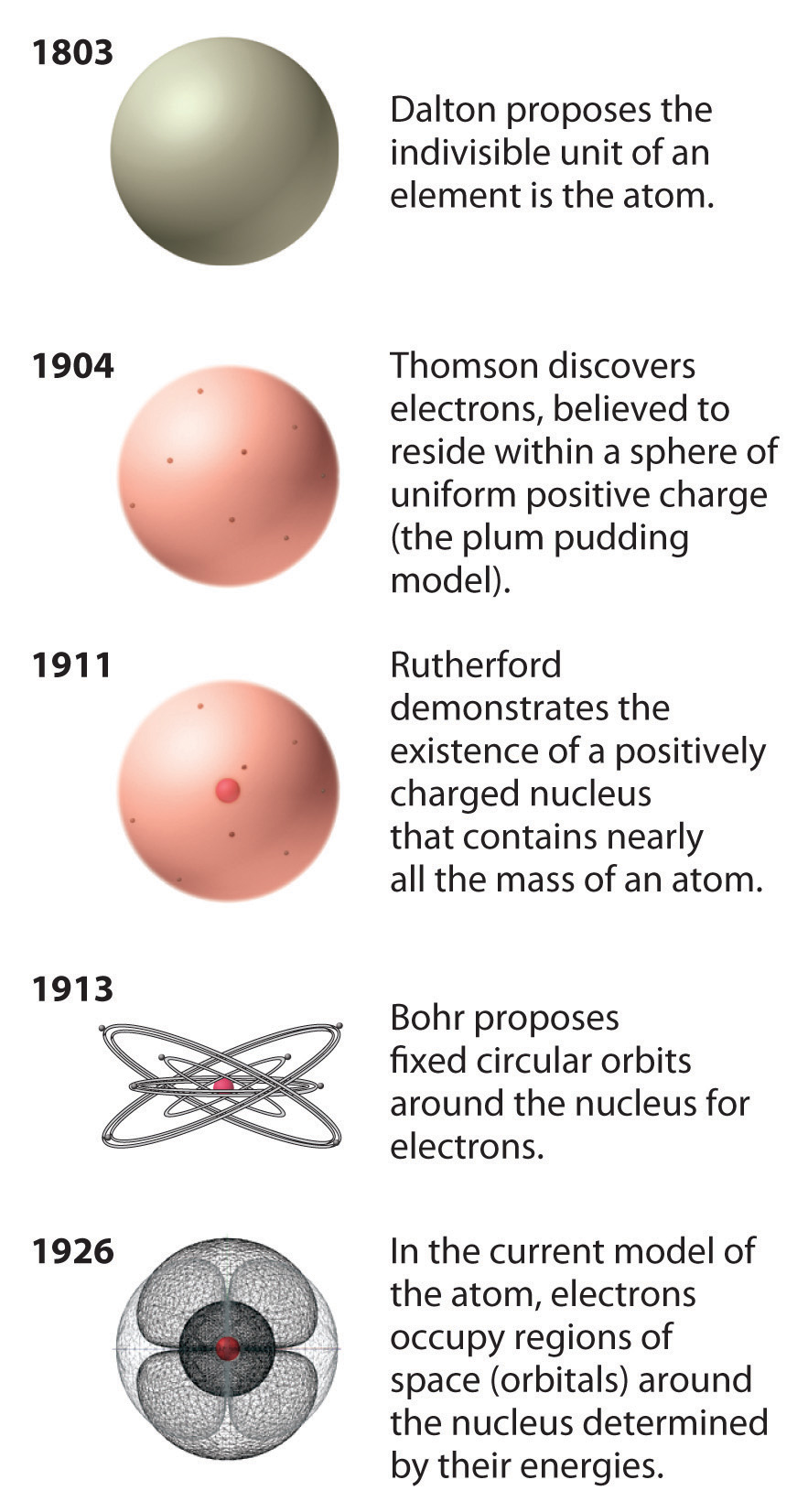
Figure \(\PageIndex{9}\): The Evolution of Atomic Theory, as Illustrated by Models of the Oxygen Atom. Bohr’s model and the current model are described in Chapter 6, "The Structure of Atoms."
Summary
Atoms are the ultimate building blocks of all matter. The modern atomic theory establishes the concepts of atoms and how they compose matter. Atoms, the smallest particles of an element that exhibit the properties of that element, consist of negatively charged electrons around a central nucleus composed of more massive positively charged protons and electrically neutral neutrons. Radioactivity is the emission of energetic particles and rays (radiation) by some substances. Three important kinds of radiation are α particles (helium nuclei), β particles (electrons traveling at high speed), and γ rays (similar to x-rays but higher in energy).