26.11 How do Enzymes Work? Citrate Synthase
- Page ID
- 91066
Objectives
After completing this section, you should be able to
- describe and explain the general function of an enzyme like citrate synthase in a reaction.
- identify the structures of ten common coenzymes.

Oxaloacetate to Citrate Catalyzed by Citrate Synthase
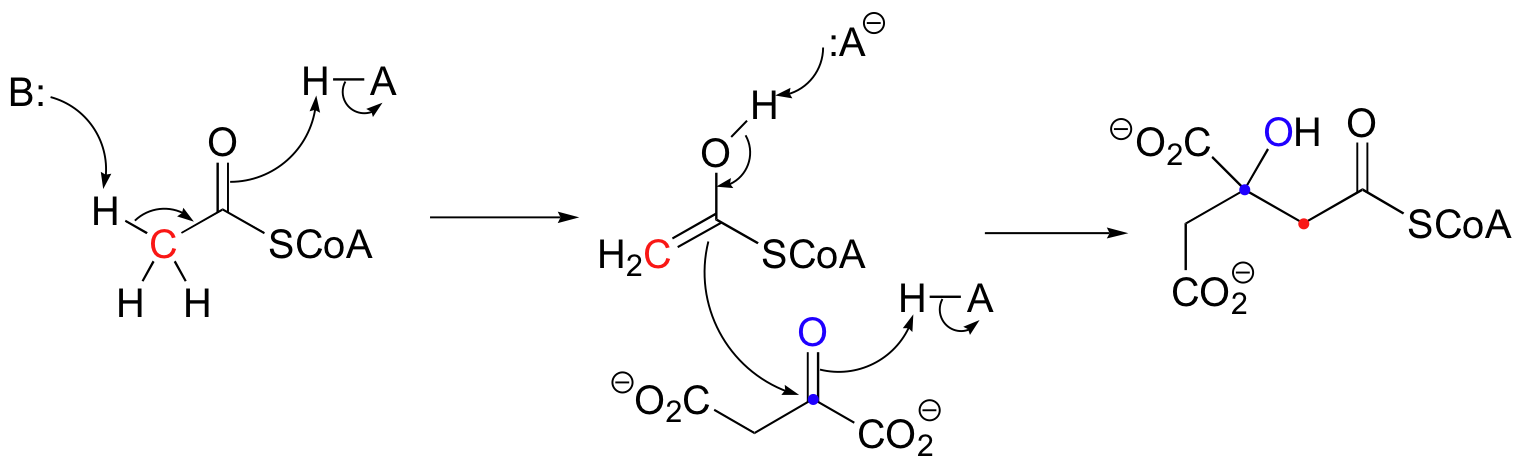
Citrate synthase is a protein with 433 amino acids with various functional groups that can react with substrates. This enzyme catalyzes oxaloacetate to eventually produce citrate as part of the citric acid (Krebs) cycle. In the first step of the citric acid (Krebs) cycle, acetyl CoA condenses with oxaloacetate to form (S)-citryl CoA. The carboxylate group of an aspartic acid (B:) on citrate synthase removes the acidic alpha proton on acetyl CoA, while a histidine site (H-A) donates a proton to form the enol. Then a second histidine site (H-A) protonates the carbonyl oxygen of oxaloacetate, while the carbon of the carbonyl is attacked by the enol. Simultaeously, that first histidine (:A-) deprotonates the acetyl CoA enol. (S)-citryl CoA is generated.
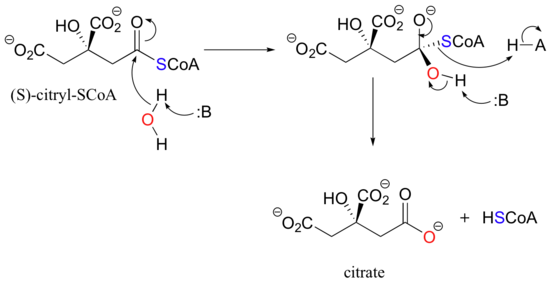
The acyl group of a thioester of (S)-citryl CoA can be transferred to a water molecule in a hydrolysis reaction to converting (S)-citryl CoA to citrate. Again histidine sites on citrate synthase are an integral part of the mechanism and assist with removal and addition of protons.
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
Organic Chemistry With a Biological Emphasis by Tim Soderberg (University of Minnesota, Morris)

