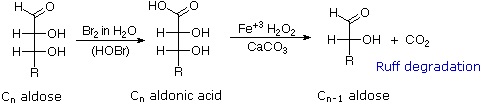25.6 Reactions of Monosaccharides
- Page ID
- 91047
Objectives
After completing this section, you should be able to
-
- write equations to illustrate that the hydroxyl groups of carbohydrates can react to form esters and ethers.
- identify the product formed when a given monosaccharide is reacted with acetic anhydride or with silver oxide and an alkyl halide.
- identify the reagents required to convert a given monosaccharide to its ester or ether.
-
- write an equation to show how a monosaccharide can be converted to a glycoside using an alcohol and an acid catalyst.
- identify the product formed when a given monosaccharide is treated with an alcohol and an acid catalyst.
- write a detailed mechanism for the formation of a glycoside by the reaction of the cyclic form of a monosaccharide with an alcohol and an acid catalyst.
- identify the ester formed by phosphorylation in biologically important compounds.
-
- identify the product formed when a given monosaccharide is reduced with sodium borohydride.
- identify the monosaccharide which should be reduced in order to form a given polyalcohol (alditol).
-
- explain that a sugar with an aldehyde or hemiacetal can be oxidized to the corresponding carboxylic acid (also known as aldonic acid). Note: The sugar is able to reduce an oxidizing agent, and is thus called a reducing sugar. Tests for reducing sugars include the use of Tollens’ reagent, Fehling’s reagent and Benedict’s reagent.
- explain why certain ketoses, such as fructose, behave as reducing sugars even though they do not contain an aldehyde group.
- identify warm HNO3 as the reagent needed to form dicarboxylic acid (an aldaric acid).
-
- describe the chain‑lengthening effect of the Kiliani‑Fischer synthesis.
- predict the product that would be produced by the Kiliani‑Fischer synthesis of a given aldose.
- identify the aldose that would yield a given product following Kiliani‑Fischer synthesis.
-
- describe the chain‑shortening effect of the Wohl degradation.
- predict the product that would be produced by the Wohl degradation of a given aldose.
- identify the aldose or aldoses that would yield a given product following Wohl degradation.
Make certain that you can define, and use in context, the key terms below.
- aldaric acid
- aldonic acid
- alditol
- aldonic acid
- glycoside
- Kiliani-Fischer synthesis
- neighbouring group effect
- reducing sugar
- Wohl degradation
While several reactions are covered in this section, keep in mind that you have encountered them in previous sections. The active functional groups on monosaccharides are essentially carbonyls and hydroxyls. Although they now are a part of much larger molecules, their chemistry should be familiar.
The formation of esters and ethers is quite straightforward and should not require further clarification.
Note that glycosides are in fact acetals, and that glycoside formation is therefore analogous to acetal formation. To refresh your memory about the chemistry of acetals, quickly review Section 19.10
Ester and Ether Formation
The -OH groups on a monosaccharide can be readily converted to esters and ethers. Esterfication can be done with an acid chloride (Section 21.4) or acid anhydride (Section 21.5), while treatment with an alkyl halide by a Williamson ether synthesis (Section 18.2) leads to the ether.
Glycoside Formation
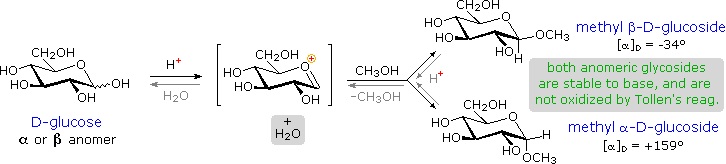
Acetal derivatives formed when a monosaccharide reacts with an alcohol in the presence of an acid catalyst are called glycosides. This reaction is illustrated for glucose and methanol in the diagram below. In naming of glycosides, the "ose" suffix of the sugar name is replaced by "oside", and the alcohol group name is placed first. As is generally true for most acetals, glycoside formation involves the loss of an equivalent of water. The diether product is stable to base and alkaline oxidants such as Tollen's reagent. Since acid-catalyzed aldolization is reversible, glycosides may be hydrolyzed back to their alcohol and sugar components by aqueous acid.
The anomeric methyl glucosides are formed in an equilibrium ratio of 66% alpha to 34% beta. From the structures in the previous diagram, we see that pyranose rings prefer chair conformations in which the largest number of substituents are equatorial. In the case of glucose, the substituents on the beta-anomer are all equatorial, whereas the C-1 substituent in the alpha-anomer changes to axial. Since substituents on cyclohexane rings prefer an equatorial location over axial (methoxycyclohexane is 75% equatorial), the preference for alpha-glycopyranoside formation is unexpected, and is referred to as the anomeric effect.
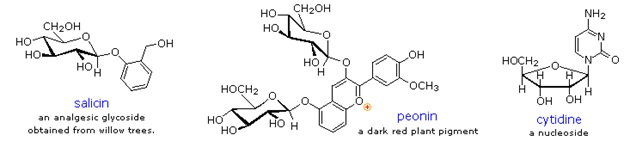
Glycosides abound in biological systems. By attaching a sugar moiety to a lipid or benzenoid structure, the solubility and other properties of the compound may be changed substantially. Because of the important modifying influence of such derivatization, numerous enzyme systems, known as glycosidases, have evolved for the attachment and removal of sugars from alcohols, phenols and amines. Chemists refer to the sugar component of natural glycosides as the glycon and the alcohol component as the aglycon.
Two examples of naturally occurring glycosides and one example of an amino derivative are displayed above. Salicin, one of the oldest herbal remedies known, was the model for the synthetic analgesic aspirin. A large class of hydroxylated, aromatic oxonium cations called anthocyanins provide the red, purple and blue colors of many flowers, fruits and some vegetables. Peonin is one example of this class of natural pigments, which exhibit a pronounced pH color dependence. The oxonium moiety is only stable in acidic environments, and the color changes or disappears when base is added. The complex changes that occur when wine is fermented and stored are in part associated with glycosides of anthocyanins. Finally, amino derivatives of ribose, such as cytidine play important roles in biological phosphorylating agents, coenzymes and information transport and storage materials.
Biological Ester Formation: Phosphorylation
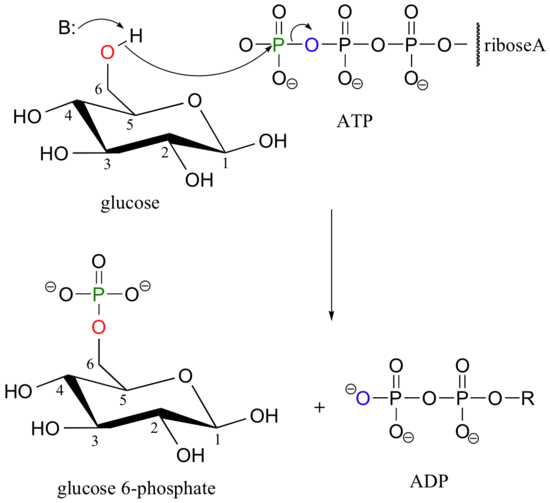
Recall that almost all biomolecules are charged species, which 1) keeps them water soluble, and 2) prevents them from diffusing across lipid bilayer membranes. Although many biomolecules are ionized by virtue of negatively charged carboxylate and positively charged amino groups, the most common ionic group in biologically important organic compounds is phosphate - thus the phosphorylation of alcohol groups is a critical metabolic step. In alcohol phosphorylations, ATP is almost always the phosphate donor, and the mechanism is very consistent: the alcohol oxygen acts as a nucleophile, attacking the gamma-phosphorus of ATP and expelling ADP (look again, for example, at the glucose kinase reaction that we first saw in section 10.1D).
Oxidation
As noted above, sugars may be classified as reducing or non-reducing based on their reactivity with Tollens', Benedict's or Fehling's reagents. If a sugar is oxidized by these reagents it is called reducing, since the oxidant (Ag(+) or Cu(+2)) is reduced in the reaction, as evidenced by formation of a silver mirror or precipitation of cuprous oxide. The Tollens' test is commonly used to detect aldehyde functions; and because of the facile interconversion of ketoses and aldoses under the basic conditions of this test, ketoses such as fructose also react and are classified as reducing sugars.
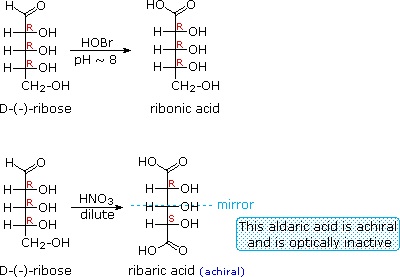
When the aldehyde function of an aldose is oxidized to a carboxylic acid the product is called an aldonic acid. Because of the 2º hydroxyl functions that are also present in these compounds, a mild oxidizing agent such as hypobromite must be used for this conversion (equation 1). If both ends of an aldose chain are oxidized to carboxylic acids the product is called an aldaric acid. By converting an aldose to its corresponding aldaric acid derivative, the ends of the chain become identical (this could also be accomplished by reducing the aldehyde to CH2OH, as noted below). Such an operation will disclose any latent symmetry in the remaining molecule. Thus, ribose, xylose, allose and galactose yield achiral aldaric acids which are, of course, not optically active. The ribose oxidation is shown in equation 2 below.
|
1. |
|
|
2. |
|
|
3. |
|
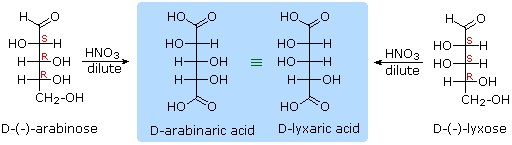
Other aldose sugars may give identical chiral aldaric acid products, implying a unique configurational relationship. The examples of arabinose and lyxose shown in equation 3 above illustrate this result. Remember, a Fischer projection formula may be rotated by 180º in the plane of projection without changing its configuration.
Reduction
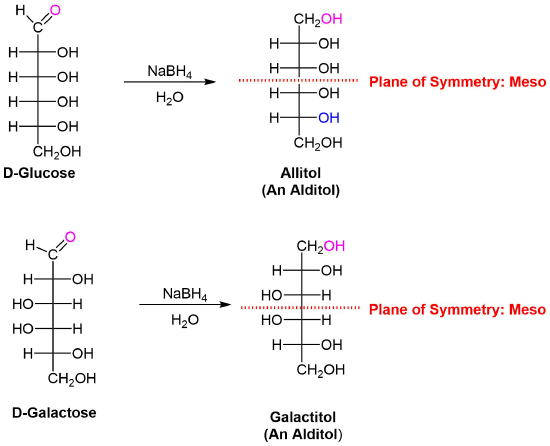
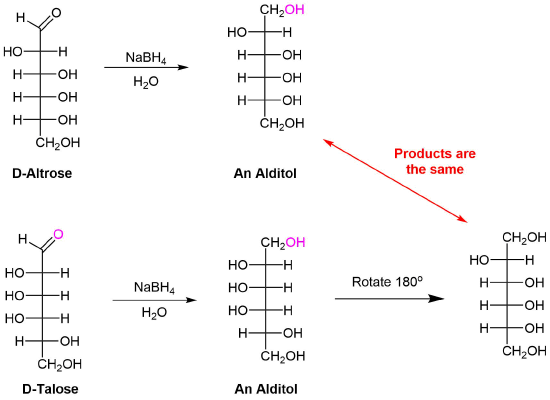
Sodium borohydride reduction of an aldose makes the ends of the resulting alditol chain identical, HOCH2(CHOH)nCH2OH, thereby accomplishing the same configurational change produced by oxidation to an aldaric acid. Thus, allitol and galactitol from reduction of allose and galactose are achiral, and altrose and talose are reduced to the same chiral alditol. A summary of these redox reactions, and derivative nomenclature is given in the following table.
|
\(HOBr\) Oxidation |
\(\longrightarrow\) |
\(HOCH_2(CHOH)_nCO_2H\) |
|
\(HNO_3\) Oxidation |
\(\longrightarrow\) |
\(H_2OC(CHOH)_nCO_2H\) |
|
\(NaBH_4\) Reduction |
\(\longrightarrow\) |
\(HOCH_2(CHOH)_nCH_2OH\) |
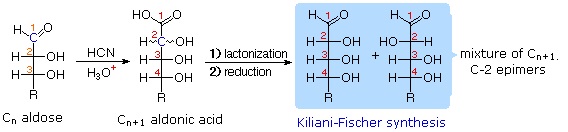
Chain Shortening and Lengthening
|
1. Ruff Degradation |
|
|
2. Kiliani-Fischer Synthesis |
|
These two procedures permit an aldose of a given size to be related to homologous smaller and larger aldoses. The importance of these relationships may be seen in the array of aldose structures presented earlier, where the structural connections are given by the dashed blue lines. Thus Ruff degradation of the pentose arabinose gives the tetrose erythrose. Working in the opposite direction, a Kiliani-Fischer synthesis applied to arabinose gives a mixture of glucose and mannose. An alternative chain shortening procedure known as the Wohl degradation is essentially the reverse of the Kiliani-Fischer synthesis.
Note that in the Kiliani-Fischer synthesis the first step is to generate a cyanohydrin intermediate, which is then has its nitrile group hydrolyzed to the carboxylic acid. From there the cyclic ester (lactone) is formed and reduced to the final products (epimers).
Wohl Degradation
The ability to shorten (degrade) an aldose chain by one carbon was an important tool in the structure elucidation of carbohydrates. This was commonly accomplished by the Ruff procedure. An interesting alternative technique, known as the Wohl degradation has also been used. The following equation illustrates the application of this procedure to the aldopentose, arabinose. Based on your knowledge of carbonyl chemistry, and considering that the Wohl degradation is in essence the reverse of the Kiliani-Fischer synthesis.
Exercises
1) D-arabinose and D-lyxose produce the same chiral aldaric acid product when oxidized with dilute HNO3. Please explain.
2) Which two D-aldohexoses are oxidized to produce an optically inactive (meso) aldaric acid?
- Answer
-
1) Remember, a Fischer projection formula may be rotated by 180º in the plane of projection without changing its configuration.
2) D-Allose and
D-galactose.
1) Allitol and galactitol from the reduction of allose and galactose are achiral. Explain why this occurs.
2) Altrose and talose are reduced to the same chiral alditol. Explain why this occurs.
- Answer
-
1)
2)
1) What products would you expect from Kiliani–Fischer reaction of
2) What aldopentose would be expected to produce a mixture of
- Answer
-
1) D-Gluose and D-idose
2) D-Threose
Two of the four D- aldopentoses yield
- Answer
-
D-Ribose and D-arabinose
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry