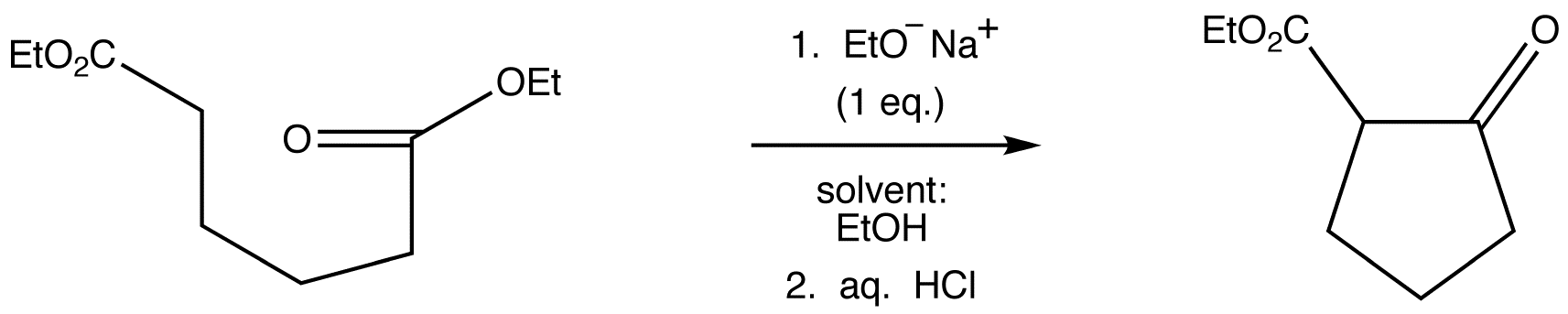23.9 Intramolecular Claisen Condensations - The Dieckmann Cyclization
- Page ID
- 91022
Objectives
After completing this section, you should be able to
- write an equation to illustrate an internal Claisen condensation, that is, a Dieckmann cyclization.
- identify the product formed when a 1,6‑ or 1,7‑diester undergoes an internal Claisen condensation.
- identify the diester needed to prepare a given cyclic β‑keto ester by an internal Claisen condensation.
- identify the structural features present in a diester that lead to the formation of more than one product in an internal Claisen condensation.
- Dieckmann cyclization
Essentially no new material is introduced in this section; Dieckmann cyclizations are intramolecular Claisen condensations. These reactions occur for 1,6‑ and 1,7‑diesters, as these substances result in the formation of compounds containing five‑ and six‑membered rings, respectively. You may recall that the formation of such ring systems is favoured because they are relatively free of strain.
Dieckmann Condensation
A diester can undergo an intramolecular reaction called a Dieckmann condensation.
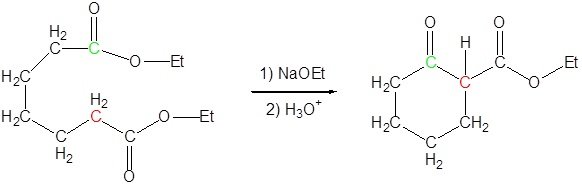
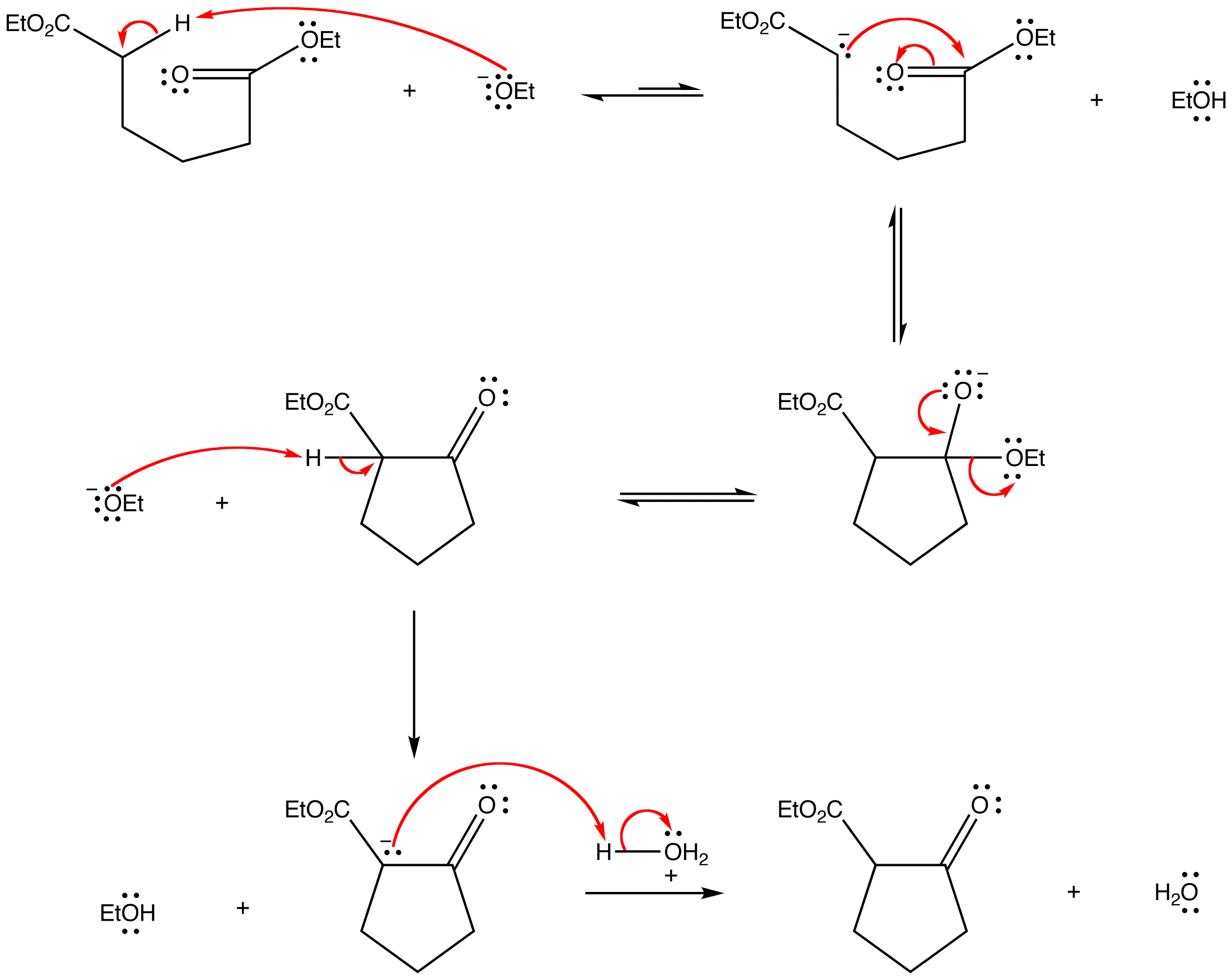
Mechanism
Exercises
Draw the expected product for the following reaction:
- Answer
-
The Dieckmann cyclization of the following molecule is expected to give a mixture of two cyclized products. Draw the structure of the two products and briefly explain why a mixture is formed.
- Answer
-
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
- Gamini Gunawardena from the OChemPal site (Utah Valley University)