23.4 Using Aldol Reactions in Synthesis
- Page ID
- 91017
Objectives
After completing this section, you should be able to identify the aldehyde or ketone and other necessary reagents that should be used to prepare a given enone by an aldol condensation.
This section stresses the importance of being able to think logically.
The experience that you have already gained through designing multi‑step syntheses and solving road‑map problems should help you to recognize when an aldol reaction may have been one of the steps in the synthesis of a given compound.
It is important that you recognize that the aldol condensation is an important part of a synthetic chemist’s repertoire, both because it involves the formation of a new carbon‑carbon bond, and also because it yields a product containing two functional groups.
Aldol reactions are excellent methods for the synthesis of many enones or beta hydroxy carbonyls. Because of this, being able to predict when an aldol reaction might be used in a synthesis in an important skill. This accomplished by mentally breaking apart the target molecule and then considering what the starting materials might be.
Fragments which are easily made by an aldol reaction
Steps to 'reverse' the aldol reaction (from the final aldol product towards identifying the starting compounds).
1) From an enone break the double bond and form two single bonds. Place and OH on the bond furthest from the carbonyl and an H on the bond closest to the carbonyl.
2) From the aldol product break the C-C bond between the alpha carbon and the carbon attached to the OH. Then turn the OH into a carbonyl and add an hydrogen to the other carbon.
Worked Example 23.4.1
Show how the following molecule could be made using an aldol condensation?
- Answer
-
Analysis: The C=C bond in the target molecule is cleaved to form two fragments. The fragment with the carbonyl gains two alpha hydrogens. The other fragment gains a =O to form a carbonyl. Both fragments end up producing the same reactant which is typical for an aldol condensation.
Solution
Exercises
Which of the following molecules could be made using an aldol reaction or condensation. Please show the starting material for the reactions that are possible.
- Answer
-
Molecules A and C are possible.
Solution for molecule A
Solution for molecule C
Please show the reaction steps required to make 4-methyl-2-pentanol from an aldol condensation.
- Answer
-
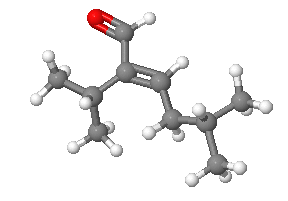
Please design a synthesis for the following molecule: Red = Oxygen, Grey = Carbon, White = Hydrogen
- Answer
-
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)

