22.7 Alkylation of Enolate Ions
- Page ID
- 91011
Objectives
After completing this section, you should be able to
- write a general mechanism for the attack of an enolate anion on an alkyl halide.
- write a reaction sequence to illustrate the preparation of carboxylic acids via the malonic ester synthesis.
- identify the product formed, and all the intermediates, in a given malonic ester synthesis.
- identify all of the compounds needed to prepare a given carboxylic acid by a malonic ester synthesis.
- write a detailed mechanism for each of the steps involved in a malonic ester synthesis.
- write a reaction sequence to illustrate the preparation of ketones through the acetoacetic ester synthesis.
- identify the product formed, and all the intermediates, in a given acetoacetic ester synthesis.
- identify all of the compounds needed to prepare a given ketone by an acetoacetic ester synthesis.
- write a detailed mechanism for each of the steps involved in an acetoacetic ester synthesis.
- identify the product or products formed when a given lactone, ester, nitrile or ketone is treated with lithium diisopropylamide followed by an alkyl halide.
- identify the compounds needed to prepare a given α‑substituted ketone, ester, lactone or nitrile by a method involving the alkylation of an enolate anion.
Make certain that you can define, and use in context, the key terms below.
- alkylation
- malonic ester synthesis
The two syntheses discussed in this section provide routes to a wide variety of carboxylic acids and methyl ketones. You may wish to review the factors influencing SN2 reactions (Section 11.3) in conjunction with this section.
You should try to memorize the structures of malonic ester and ethyl acetoacetate. The IUPAC names of these compounds are shown in the table below.
| Structure | Common name | IUPAC name |
|---|---|---|
 |
malonic acid | propanedioic acid |
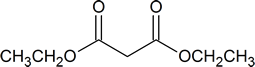 |
malonic ester or diethyl malonate |
diethyl propanedioate |
 |
acetoacetic acid | 3‑oxobutanoic acid |
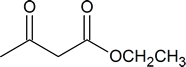 |
ethyl acetoacetate or acetoacetic ester |
ethyl 3‑oxobutanoate |
Enolates can act as a nucleophile in SN2 type reactions. Overall an α hydrogen is replaced with an alkyl group. This reaction is one of the more important for enolates because a carbon-carbon bond is formed. These alkylations are affected by the same limitations as SN2 reactions previously discussed. A good leaving group, X= chloride, bromide, iodide, tosylate, should be used. Also, secondary and tertiary leaving groups should not be used because of poor reactivity and possible competition with elimination reactions. Lastly, it is important to use a strong base, such as LDA or sodium amide, for this reaction. Using a weaker base such as hydroxide or an alkoxide leaves the possibility of multiple alkylation’s occurring.
Malonic Ester Synthesis
Malonic ester is a reagent specifically used in a reaction which converts alkyl halides to carboxylic acids called the Malonic Ester Synthesis. Malonic ester synthesis is a synthetic procedure used to convert a compound that has the general structural formula 1 into a carboxylic acid that has the general structural formula 2.
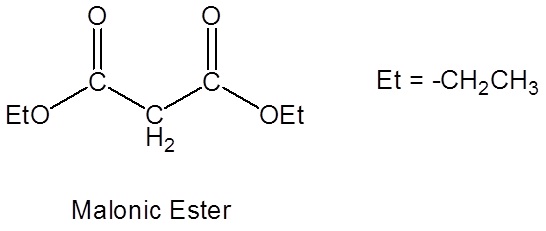
Due to the fact that Malonic ester’s α hydrogens are adjacent to two carbonyls, they can be deprotonated by sodium ethoxide (NaOEt) to form Sodio Malonic Ester.
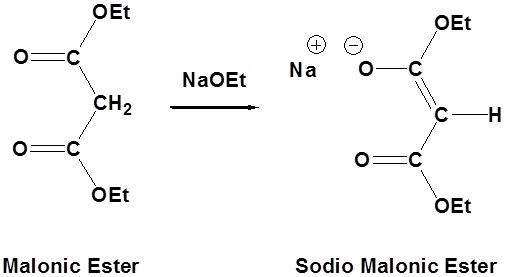
Because Sodio Malonic Ester is an enolate, it can then be alkylated with alkyl halides.
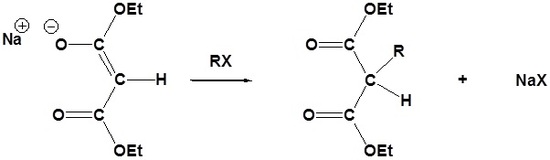
After alkylation the product can be converted to a dicarboxylic acid through saponification and subsequently one of the carboxylic acids can be removed through a decarboxylation step.
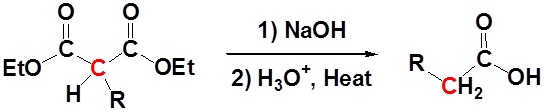
Mechanism
1) Saponification
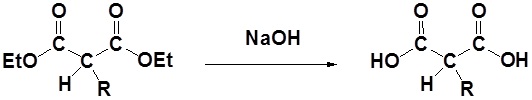
2) Decarboxylation
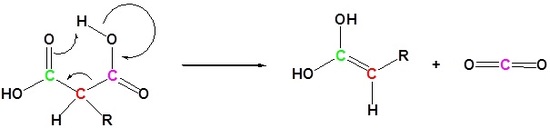
3) Tautomerization

All of the steps together form the Malonic ester synthesis.
\[RX \rightarrow RCH_2CO_2H\]
Example
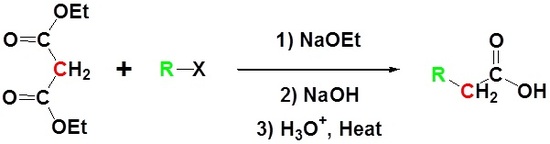
The Acetoacetic Ester Synthesis
The acetoacetic ester synthesis allows for the conversion of ethyl acetoacetate into a methyl ketone with one or two alkyl groups on the alpha carbon.
Steps
1) Deprotonation with ethoxide
2) Alkylation via and SN2 Reaction
3) Hydrolysis and decarboxylation
Addition of a second alky group
After the first step and additional alkyl group can be added prior to the decarboxylation step. Overall this allows for the addition of two different alkyl groups.
Direct Alkylation
Both the malonic ester synthesis and acetoacetic ester synthesis can be preformed with relatively mild bases, such as sodium ethoxide, because of the unusually acidic nature of the dicarbonyl compounds. However, if a stronger base is used, such as LDA, monocarbonyl compounds can also be alkylated in the alpha position.
Mechanism
1) Enolate formation
2) SN2 attack
Enolate of Unsymmetrical Carbonyl Compounds
Now let’s consider what happens when an unsymmetrical carbonyl is treated with a base. In the case displayed below there are two possible enolates which can form. The removal of the 2o hydrogen forms the kinetic enolate and is formed faster because it is less substituted and thereby less sterically hindered. The removal of the 3o hydrogen forms the thermodynamic enolate which is more stable because it is more substituted.
Kinetic Enolates
Kinetic enolates are formed when a strong bulky base like LDA is used. The bulky base finds the 2o hydrogen less sterically hindered and preferable removes it. Low temperature are typically used when forming the kinetic enolate to prevent equilibration to the more stable thermodynamic enolate. Typically a temperature of -78 oC is used.
Thermodynamic Enolates
The thermodynamic enolate is favored by conditions which allow for equilibration. The thermodynamic enolate is usually formed by using a strong base at room temperature. At equilibrium the lower energy of the thermodynamic enolate is preferred, so that the more stable, more stubstituted enolate is formed.
Exercises
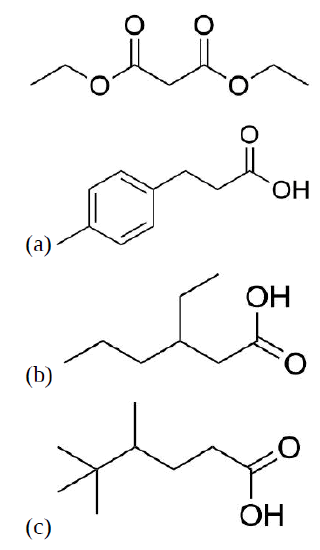
Propose a synthesis for each of the following molecules from this malonic ester.
- Answer
-
(a) 1) Malonic Ester, NaOEt, 2) 4-Methylbenzyl Bromide, 3) Base, 4) Acid, Heat
(b) 1) Malonic Ester, NaOEt, 2) 3-bromohexane, 3) Base, 4) Acid, Eat
(c) 1) Malonic Ester, NaOEt, 2) 1-Bromo-2,3,3-trimethylbutane, 3) Base, 4) Acid, Heat
Why can't you prepare tri substituted acetic acids from a malonic ester?
- Answer
-
Malonic esters only contain two acid protons.
Propose a synthesis for the following molecule via a malonic ester.
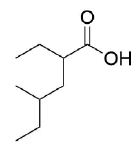
- Answer
-
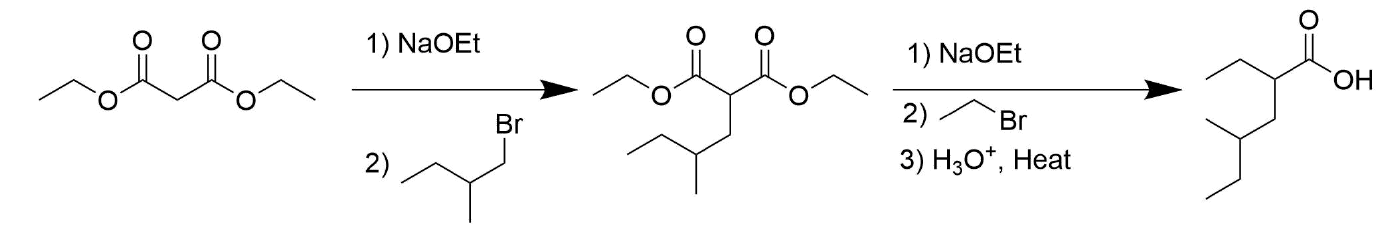
How might you prepare the following compounds from an alkylation reaction?
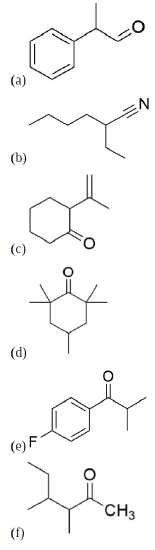
- Answer
-
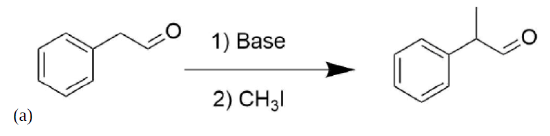
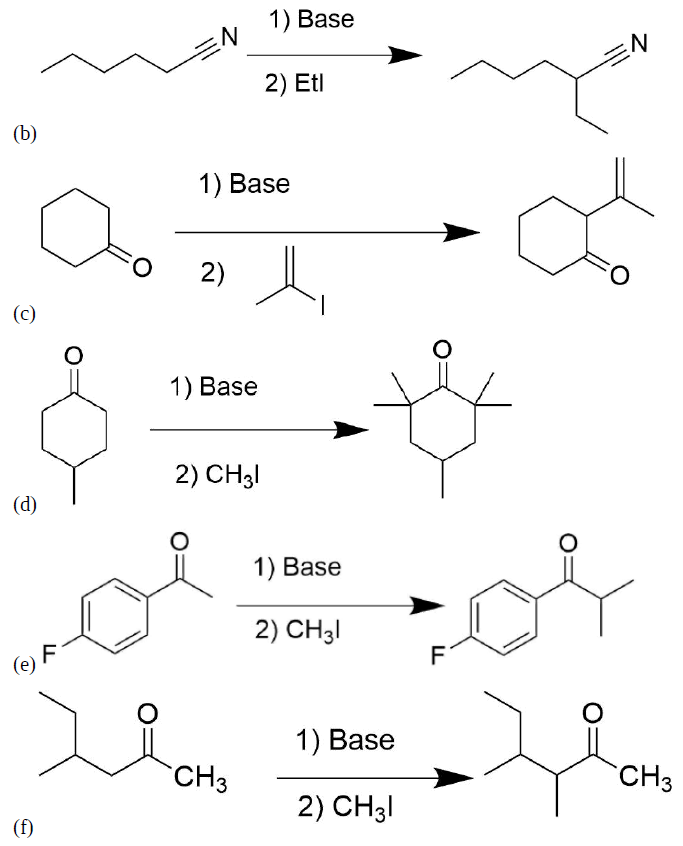
Please write the structure of the product for the following reactions.
- Answer
-
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
Prof. Steven Farmer (Sonoma State University)

