22.1 Keto-Enol Tautomerism
- Page ID
- 91005
Objectives
After completing this section, you should be able to
- write an equation to illustrate keto‑enol tautomerism.
- write a detailed mechanism for acid‑catalyzed keto‑enol tautomerism.
- write a detailed mechanism for base‑catalyzed keto‑enol tautomerism.
- draw the structure of the enol form of a given carbonyl compound.
Make certain that you can define, and use in context, the key terms below.
- enol
- keto
- tautomerism
- tautomers
- enolate ion
Keto‑enol tautomerism was first introduced in Section 9.4, in the discussion of the hydration of alkynes. The subject was raised again in the chapter entitled A Preview of Carbonyl Compounds, during the brief overview of the alpha‑substitution reactions of carbonyl compounds. You may wish to review these sections before proceeding.
Often, the position of a carbon atom near a carbonyl group is designated using Greek letters. The atom adjacent to the carbonyl is alpha, the next removed is beta and so on. The carbon in the carbonyl group is used as reference point and is not assigned a Greek letter. Likewise, hydrogens bare the same Greek letter as the carbon atoms to which they are attached. α-Hydrogens are bonded to α-carbons and β-hydrogens are bonded to β-carbons etc.
The presence of α-hydrogens in a molecule provides the possibility of certain chemical reactions, which will be discussed in this chapter and in Chapter 23. Because of this, the ability to identify α-hydrogens is an important skill. As shown below, pentanal has two α-hydrogens. Note that aldehyde hydrogens are not given a Greek letter, they are simply referred to as an aldehyde hydrogen.
α-hydrogens, which are attached to a carbon directly adjacent to a carbonyl group, display unusual acidity. This is almost exclusively due to the resonance stabilization of the carbanion conjugate base, called an enolate, as illustrated in the diagram below. The effect of the the stabilizing C=O is seen when comparing the pKa for the α-hydrogens of aldehydes (~16-18), ketones (~19-21), and esters (~23-25) to that of a typical alkyl C-H bond (~40-50).
Indicate any α-hydrogens contained in the following molecules:
Solution
Because of the acidity of α hydrogens carbonyls undergo keto-enol tautomerism. Tautomers are rapidly interconverted constitutional isomers, usually distinguished by a different bonding location for a labile hydrogen atom and a differently located double bond. The equilibrium between tautomers is not only rapid under normal conditions, but it often strongly favors one of the isomers (acetone, for example, is 99.999% keto tautomer). Even in such one-sided equilibria, evidence for the presence of the minor tautomer comes from the chemical behavior of the compound. Tautomeric equilibria are catalyzed by traces of acids or bases that are generally present in most chemical samples.
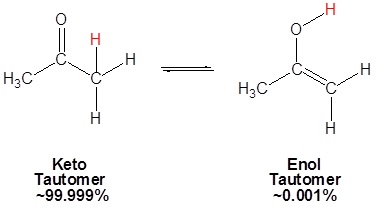
Mechanism for Enol Formation
Acid conditions
1) Protonation of the Carbonyl
2) Enol formation
Basic conditions
1) Enolate formation
2) Protonation
How Enols React
Exercises
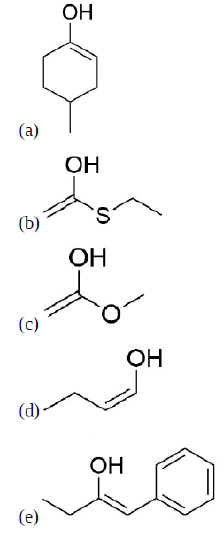
Draw the enol forms of the following molecules
- 4-methylcyclohexanone
- Ethyl thioactetate
- Methyl acetate
- Butanal
- 1-phenyl-2-butanone
- Answer
-
How many α-hydrogens do each of the molecules from the previous question have? Label them.
- Answer
-
(a)
(b)
(c)
(d)
(e)
Under normal conditions cyclohexanone exists in the enol tautomer in a much higher percentage than acetone. Explain.
- Answer
-
The enol tautomer of cyclohexanone has more alky substituents than the enol tautomer of acetone. This makes the double bond of the enol double bond of cyclohexanone more stable and easier to form.
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)

