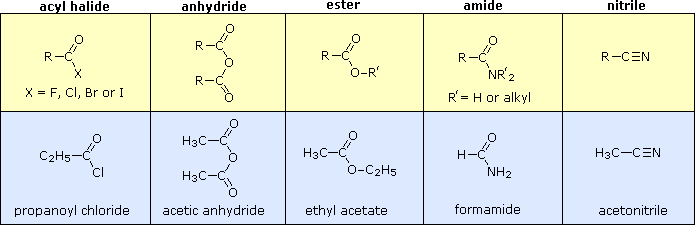
20.0 Introduction
- Page ID
- 90980
Objectives
After completing this section, you should be able to
- list a number of naturally occurring carboxylic acids.
- state one of the industrial uses of acetic acid.
The global demand for acetic acid is about 6.5 million tonnes per year. A common industrial preparation of acetic acid is the catalytic oxidation of methanol with carbon monoxide. Much of the acetic acid produced is converted to vinyl acetate polymer and used in adhesives and paints.
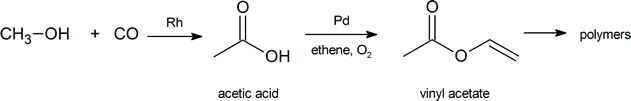
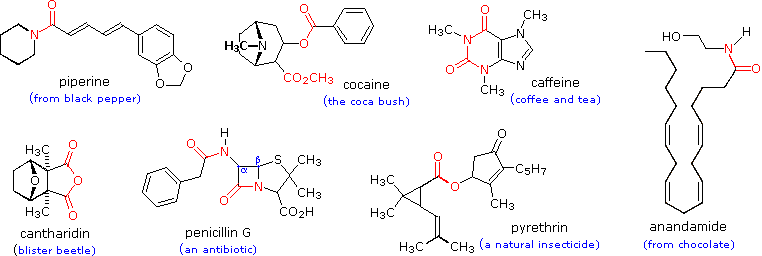
Not only are carboxylic acids valuable, but they and their derivatives are useful starting materials for many synthetic products. Finally, carboxylic acids and their derivatives are also found in a variety of natural systems and important biological pathways.
Carboxylic Acids
Serving wine usually involves a rather elaborate ceremony in which the host tastes the wine before pouring it for the guests. One reason for this is the possibility that the wine may have been spoiled by exposure to air.
Certain bacterial enzymes are capable of converting ethanol to ethanoic acid (acetic acid) when oxygen is present:
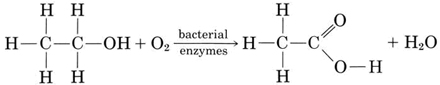
The same reaction occurs when cider changes into vinegar, which contains 4 to 5 percent acetic acid. Acetic acid gives vinegar its sour taste and pungent odor and can do the same thing to wine.
Acetic acid, CH3COOH, is an example of the class of compounds called carboxylic acids, each of which contains one or more carboxyl groups, COOH. The general formula of a carboxylic acid is RCOOH. Some other examples are
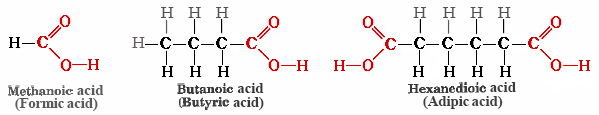
Formic acid (the name comes from Latin word formica meaning “ant“) is present in ants and bees and is responsible for the burning pain of their bites and stings. Butyric acid, a component of rancid butter and Limburger cheese, has a vile odor. Adipic acid is an example of a dicarboxylic acid—it has two functional groups—and is used to make nylon.
Carboxylic Acid Derivatives
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Ed Vitz (Kutztown University), John W. Moore (UW-Madison), Justin Shorb (Hope College), Xavier Prat-Resina (University of Minnesota Rochester), Tim Wendorff, and Adam Hahn.