7.1: Industrial Preparation and Use of Alkenes
- Page ID
- 67122
After completing this section, you should be able to
- discuss the industrial importance of ethylene (ethene) and propylene (propene).
- describe, briefly, the industrial process known as thermal cracking.
Among the most important and most abundant organic chemicals produced worldwide are the two simple alkenes, ethylene and propylene. They are used as the starting materials to synthesize numerous valuable compounds.
Produced from ethylene (ethene)
| Chemical | Uses |
|---|---|
| ethanol | solvent; constituent of cleaning preparations; in synthesis of esters |
| acetaldehyde | slug killer, in the form of methaldehyde (CH3CHO)4 |
| acetic acid | manufacture of vinyl acetate polymers, ethyl acetate solvent and cellulose acetate polymers |
| ethylene oxide | “cellosolves” (industrial solvents) |
| ethylene glycol | anti-freeze; production of DacronOR |
| ethylene dichloride | solvent; production of vinyl chloride |
| vinyl chloride | manufacture of poly (vinyl chloride)—PVC |
| vinyl acetate | manufacture of poly (vinyl acetate) used in paint emulsions, plywood adhesives and textiles |
| polyethylene | “plastic” bags; toys; packaging |
Produced from propylene (propene)
| Chemical | Uses |
|---|---|
| isopropyl alcohol | rubbing alcohol; cosmetics; synthesis of acetone |
| propylene oxide | manufacture of polyurethanes; polyesters |
| cumene | industrial preparation of phenol and acetone |
| polypropylene | molded articles (e.g., kitchenware); fibres for indoor-outdoor carpeting |
Catalytic cracking to produce propylene
Cracking is the name given to breaking up large hydrocarbon molecules into smaller and more useful bits. This is achieved by using high pressures and temperatures without a catalyst (thermal cracking), or at lower temperatures and pressures in the presence of a catalyst (catalytic cracking).
In the case of catalytic cracking, the source of the large hydrocarbon molecules is usually the gas oil fraction of crude oil (petroleum). Useful "straight run" products, such as gasoline, kerosene, diesel, butane and propane are separated from the crude oil mixture in the Atmospheric Distillation Unit (at atmospheric pressure). These "straight run" products only account for 30-50% of the crude oil, depending on its origin, the balance is in the form of atmospheric residue, typically boiling above about ~350°C/650°F. Atmospheric residue is typically routed to the Vacuum Distillation unit for separation into gas oil (usually termed vacuum gas oil, or VGO for short) and vacuum residue; via distillation at reduced pressure. VGO is the principle feed used in the Fluid Catalytic Cracking (FCC) process, though many FCC units also co-feed some portion of lower cost atmospheric residue too, typically 10-20% depending operational constraints.
The FCC unit employs a sophisticated powdered catalyst to lower the activation energy of cracking and thereby drive the cracking process. The particle size and density of the powder are set such that the catalyst powder is fluidizable - the powder behaves like a fluid when aerated. This allows the catalyst to form fluidized beds and be transported between the FCC reactor and regenerator with ease. FCC catalyst are considered to have two catalytically active parts: (a) the "zeolite", and (b) what is termed the "matrix".
The zeolite (referred to as Zeolite-Y) is a rare-earth stabilized synthetic form of a rare naturally occurring aluminosilicate mineral called faujasite. Ultrastabilization of Zeolite-Y (USY) further improves its hydrothermal stability; the final rare earth / ultrastable form of the zeolite is denoted RE-USY for short. RE-USY is a solid acid, with acidity equivalent to ~90% sulphuric acid. It is also highly crystalline and has a very regular micropore structure with mean pore mouth diameters in the range of diesel molecules - hence it behaves as a molecular sieve. Acid sites within the crystalline micropores very selectively crack diesel range molecules to gasoline and some light olefins (propene and butenes).
However, VGO consists of molecules that are bigger than diesel, too large to access the zeolite micropores. The catalyst "matrix" is designed to include a high surface area mesoporous alumina which has acid sites situated in larger pores that are able to pre-crack VGO molecules into the diesel range - in order to feed the zeolite so to speak. Optimization of FCC yields between gasoline and diesel can be achieved by adjusting the catalyst zeolite-to-matrix ratio. The main role of the FCC unit has until recently been the production of gasoline.
Propene is a very important product for polypropylene production and today 30-35% of the worlds propene monomer is made in the FCC process. Use of a co-catalysts called an "FCC Additive" is required to shift product selectivity towards propene in the FCC. FCC Additives are specially designed catalysts in separate particles that look much like the main FCC catalyst particles and can be added in any chosen proportion to achieve the desired selectivity shifts. Typically such Additives are so effective that they are only required as a minor part of the overall catalyst powder mixture in conventional FCC units (e.g. up to ~25%). An FCC Additive designed to maximize propene is very similar in concept to the main FCC catalyst above, its main active component is also a zeolite. However, in this case the zeolite has smaller crystalline micropores that will selectively admit gasoline range molecules for selectve cracking to propene and mixed butenes. This zeolite is usually called ZSM-5 for historical reasons (though more strictly it's scientific name is MFI - "Mobil Five"). Use of ZSM-5 Additives allows the FCC to double it's propene yield.
Ethene is not a catalytic product in the FCC, a small amount is formed via thermal cracking reactions which are considered undesirable in the FCC because they are non-selective and limit operations by increasing coke and light gas make. Every effort is made in the design and operation of an FCC unit to minimize thermal cracking because coke drives up the regenerator temperature and increases its air demand, while light gases are more difficult to compress in the product recovery section and limit product recovery. Ethene is a very minor product in the FCC that is not generally economically worth recovering.
The FCC process itself consists three main sections, the: (a) riser (reactor), (b) disengager, and (c) regenerator. Pre-heated liquid feed (typically at ~200°C/400°F) is injected into the lower section of the riser through feed nozzles that atomize the feed into tiny droplets (~50 micron droplets). The droplets contact hot freshly regenerated catalyst (typically at ~735°C/1350°F) where they vaporize and quench the mix zone temperature (typically to about ~570°C/1060°F). Volumetric expansion carries catalyst particles and feed vapours up the riser together, feed vapours diffuse into the catalyst (and FCC Additive) pores and crack. Cracking is endothermic, so the temperature continues to decrease until it reaches the top of the riser (typically at ~535°C/1000°F). Catalyst and product vapours are then disengaged using cyclones, the vapours leave the disengager to go to product separation and recovery section. A small amount of coke (~1 wt%) builds up on the catalyst during the reaction cycle, the disengaged catalyst is passed to the regenerator to burn off the coke and prepare the catalyst for the next cycle. The heat produced via combustion of coke on catalyst heats up the catalyst. The FCC is operated in heat-balance, where the heat of coke combustion provides the energy required to vaporize and crack the feed.
This is an example of heterogeneous catalysis - the catalyst is in a different phase (solid) from the reactants (vapours).
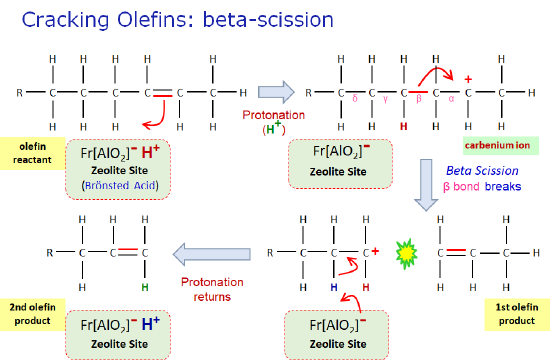
There are a complex combination of a number of catalyzed reactions taking place in the FCC unit. The initial step is formation of carbenium ions, either via protonation at Bronsted acid sites, or hydride abstraction at Lewis acid sites on the solid catalyst . Once formed carbenium ions undergo a number of reactions in parallel: beta-scission (cracking), isomerization (skeletal rearrangements), hydrogen transfer, and others. The relative rates of these reactions are optimized by adjusting operating conditions and catalyst design.
Bellow is a schematic of the beta-scission mechanism for an olefin (alkene): note the key steps: (1) initiation: carbenium ion formation via protonation from a Bronsted acid site within a zeolite, (2) propagation: beta-bond cleavage - the negatively charged electrons in the beta bond are attracted to the positive charge on the cabenium ion, (3) termination: the carbenium ion intermediate returns a proton to the zeolite site to complete the catalytic cycle.
Propene is an important material for making plastics and producing other organic chemicals, polypropylene is made by polymerization of propene monomers into long polymeric chains.
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
Jim Clark (Chemguide.co.uk)

