6.1: Kinds of Organic Reactions
- Page ID
- 67108
Objective
After completing this section, you should be able to list and describe the four important “kinds” of reactions that occur in organic chemistry.
Make certain that you can define, and use in context, the key terms below.
- addition reaction
- elimination reaction
- rearrangement reaction
- substitution reaction
It is sufficient that you know the general form of each kind of reaction. However, given a chemical equation, you should be able to recognize which kind of reaction it involves.
If you scan any organic textbook you will encounter what appears to be a very large, often intimidating, number of reactions. These are the "tools" of a chemist, and to use these tools effectively, we must organize them in a sensible manner and look for patterns of reactivity that permit us make plausible predictions. Most of these reactions occur at special sites of reactivity known as functional groups, and these constitute one organizational scheme that helps us catalog and remember reactions.
Ultimately, the best way to achieve proficiency in organic chemistry is to understand how reactions take place, and to recognize the various factors that influence their course.
First, we identify four broad classes of reactions based solely on the structural change occurring in the reactant molecules. This classification does not require knowledge or speculation concerning reaction paths or mechanisms. The four main reaction classes are additions, eliminations, substitutions, and rearrangements.
Addition Reaction
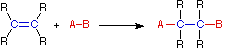
Elimination Reaction
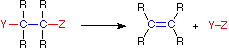
Substitution Reaction
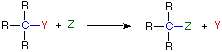
Rearrangement Reaction
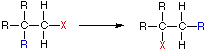
In an addition reaction the number of σ-bonds in the substrate molecule increases, usually at the expense of one or more π-bonds. The reverse is true of elimination reactions, i.e.the number of σ-bonds in the substrate decreases, and new π-bonds are often formed. Substitution reactions, as the name implies, are characterized by replacement of an atom or group (Y) by another atom or group (Z). Aside from these groups, the number of bonds does not change. A rearrangement reaction generates an isomer, and again the number of bonds normally does not change.
The examples illustrated above involve simple alkyl and alkene systems, but these reaction types are general for most functional groups, including those incorporating carbon-oxygen double bonds and carbon-nitrogen double and triple bonds. Some common reactions may actually be a combination of reaction types.
The reaction of an ester with ammonia to give an amide, as shown below, appears to be a substitution reaction ( Y = CH3O & Z = NH2 ); however, it is actually two reactions, an addition followed by an elimination.
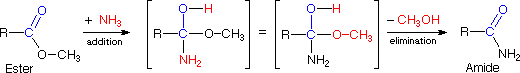
The addition of water to a nitrile does not seem to fit any of the above reaction types, but it is simply a slow addition reaction followed by a rapid rearrangement, as shown in the following equation. Rapid rearrangements of this kind are called tautomerizations.
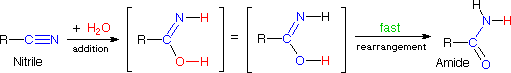
Exercise
Classify each reaction as addition, elimination, substitution, or rearrangement.
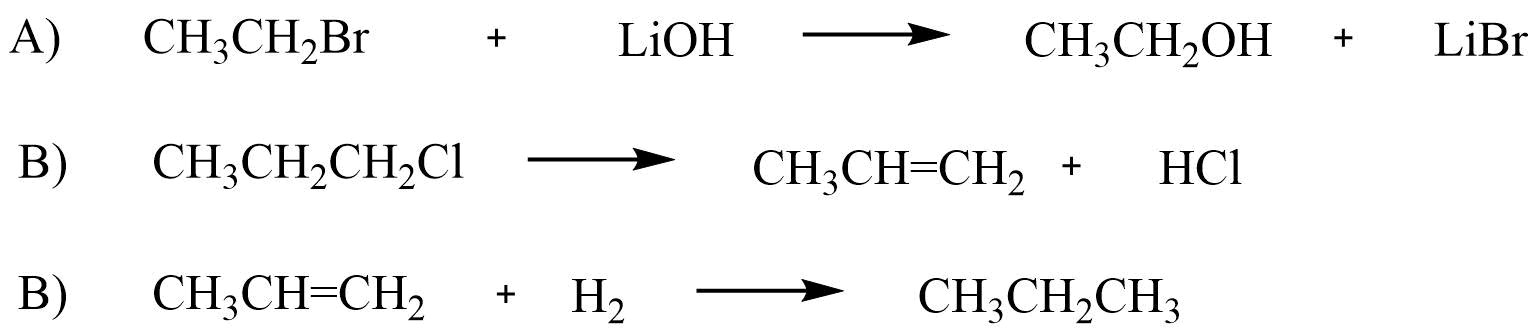
- Answer
-
A) Substitution
B) Elimination
C) Addition
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry

