16.6: Trisubstituted Benzenes- Additivity of Effects
- Page ID
- 67330
Objectives
After completing this section, you should be able to
- predict the position or positions at which electrophilic substitution will occur when a third substituent is introduced into a disubstituted benzene ring.
- explain the observed substitution pattern when a third substituent is introduced into a disubstituted benzene ring.
Orientational Interaction of Substituents
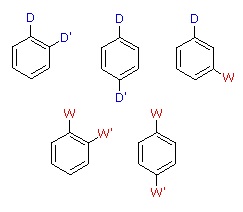
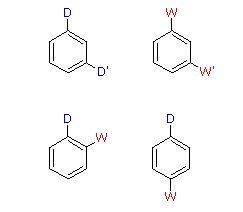
When a benzene ring has two substituent groups, each exerts an influence on subsequent substitution reactions. The activation or deactivation of the ring can be predicted more or less by the sum of the individual effects of these substituents. The site at which a new substituent is introduced depends on the orientation of the existing groups and their individual directing effects. We can identify two general behavior categories, as shown in the following table. Thus, the groups may be oriented in such a manner that their directing influences act in concert, reinforcing the outcome; or are opposed (antagonistic) to each other. Note that the orientations in each category change depending on whether the groups have similar or opposite individual directing effects.
|
Antagonistic or Non-Cooperative |
Reinforcing or Cooperative |
|
|
|
| D = Electron Donating Group (ortho/para-directing) W = Electron Withdrawing Group (meta-directing) |
|
Reinforcing or Cooperative Substitutions
The products from substitution reactions of compounds having a reinforcing orientation of substituents are easier to predict than those having antagonistic substituents. For example, the six equations shown below are all examples of reinforcing or cooperative directing effects operating in the expected manner. Symmetry, as in the first two cases, makes it easy to predict the site at which substitution is likely to occur. Note that if two different sites are favored, substitution will usually occur at the one that is least hindered by ortho groups.
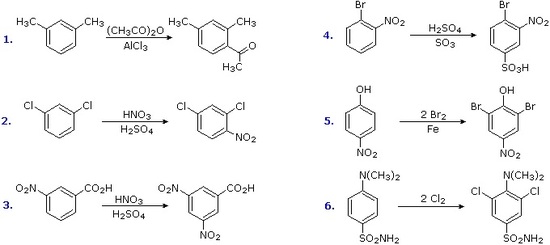
The first three examples have two similar directing groups in a meta-relationship to each other. In examples 4 through 6, oppositely directing groups have an ortho or para-relationship. The major products of electrophilic substitution, as shown, are the sum of the individual group effects. The strongly activating hydroxyl (–OH) and amino (–NH2) substituents favor dihalogenation in examples 5 and six.
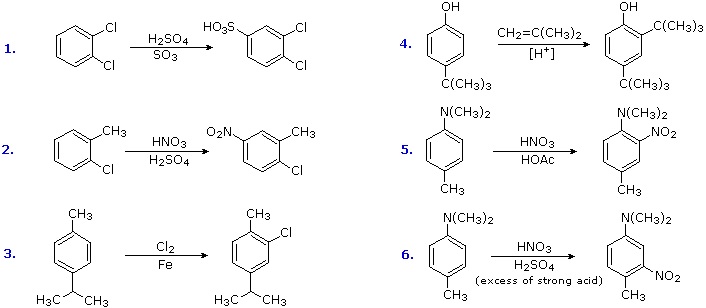
Antagonistic or Non-Cooperative Substitutions
Substitution reactions of compounds having an antagonistic orientation of substituents require a more careful analysis. If the substituents are identical, as in example 1 below, the symmetry of the molecule will again simplify the decision. When one substituent has a pair of non-bonding electrons available for adjacent charge stabilization, it will normally exert the product determining influence, examples 2, 4 & 5, even though it may be overall deactivating (case 2). Case 3 reflects a combination of steric hindrance and the superior innate stabilizing ability of methyl groups relative to other alkyl substituents. Example 6 is interesting in that it demonstrates the conversion of an activating ortho/para-directing group into a deactivating meta-directing "onium" cation [–NH(CH3)2(+) ] in a strong acid environment.
Exercise
Predict the products of the following reactions:
- Answer
-
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry