14.5: Characteristics of the Diels-Alder Reaction
- Page ID
- 67307
Objectives
After completing this section, you should be able to
- determine whether or not a given compound would behave as a reactive dienophile in a Diels-Alder reaction.
- predict the stereochemistry of the product obtained from the reaction of a given diene with a given dienophile.
- recognize that in order to undergo a Diels-Alder reaction, a diene must be able to assume ans-cis geometry, and determine whether or not a given diene can assume this geometry.
Make certain that you can define, and use in context, the key terms below.
- dienophile
- dimerization
Make sure that you understand that the s-cis and s-trans forms of a diene such as 1,3-butadiene are conformers, not isomers. Note that some textbooks can confuse the issue further by referring to a compound such as (2Z, 4Z)-hexadiene as cis, cis-2,4-hexadiene, and saying that the most stable form of this compound is its s-trans conformer!
In fulfilling Objective 2, above, you must recognize that the Diels-Alder reaction is stereospecific.
Finally, note reaction B in the reading shows 1,3-cyclopentadiene reacting with another molecule of 1,3-cyclopentadiene. When the same compound acts as both diene and dienophile in a Diels-Alder reaction to couple it is a dimerization.
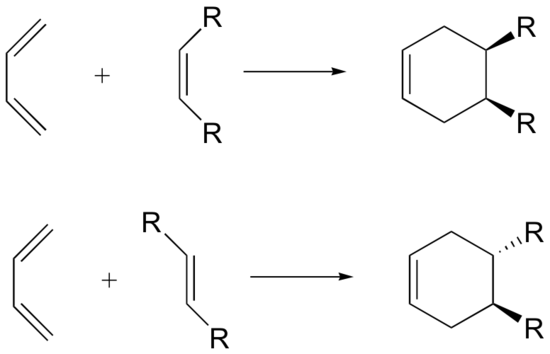
The Diels-Alder reaction is enormously useful for synthetic organic chemists, not only because ring-forming reactions are useful in general but also because in many cases two new stereocenters are formed, and the reaction is inherently stereospecific. A cis dienophile will generate a ring with cis substitution, while a trans dienophile will generate a ring with trans substitution:
In order for a Diels-Alder reaction to occur, the diene molecule must adopt what is called the s-cis conformation:
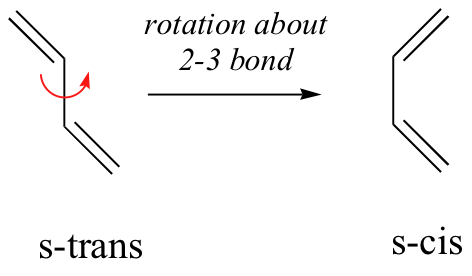
The s-cis conformation is higher in energy than the s-trans conformation, due to steric hindrance. For some dienes, extreme steric hindrance causes the s-cis conformation to be highly strained, and for this reason such dienes do not readily undergo Diels-Alder reactions.
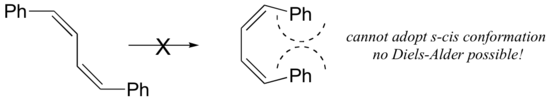
Cyclic dienes, on the other hand, are ‘locked’ in the s-cis conformation, and are especially reactive. The result of a Diels-Alder reaction involving a cyclic diene is a bicyclic structure:

Here, we see another element of stereopecificity: Diels-Alder reactions with cyclic dienes favor the formation of bicyclic structures in which substituents are in the endo position.

The endo position on a bicyclic structure refers to the position that is inside the concave shape of the larger (six-membered) ring. As you might predict, the exo position refers to the outside position.
The rate at which a Diels-Alder reaction takes place depends on electronic as well as steric factors. A particularly rapid Diels-Alder reaction takes place between cyclopentadiene and maleic anhydride.
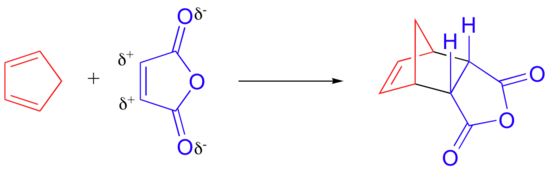
We already know that cyclopentadiene is a good diene because of its inherent s-cis conformation. Maleic anhydride is also a very good dienophile, because the electron-withdrawing effect of the carbonyl groups causes the two alkene carbons to be electron-poor, and thus a good target for attack by the pi electrons in the diene.
In general, Diels-Alder reactions proceed fastest with electron-donating groups on the diene (eg. alkyl groups) and electron-withdrawing groups on the dienophile.
Alkynes can also serve as dienophiles in Diels-Alder reactions:

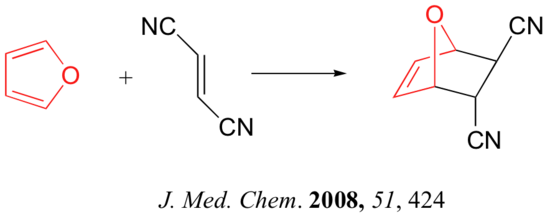
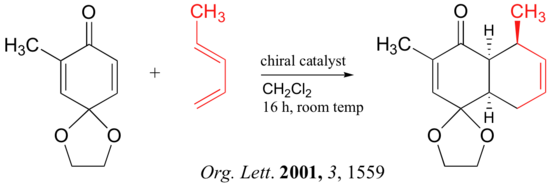
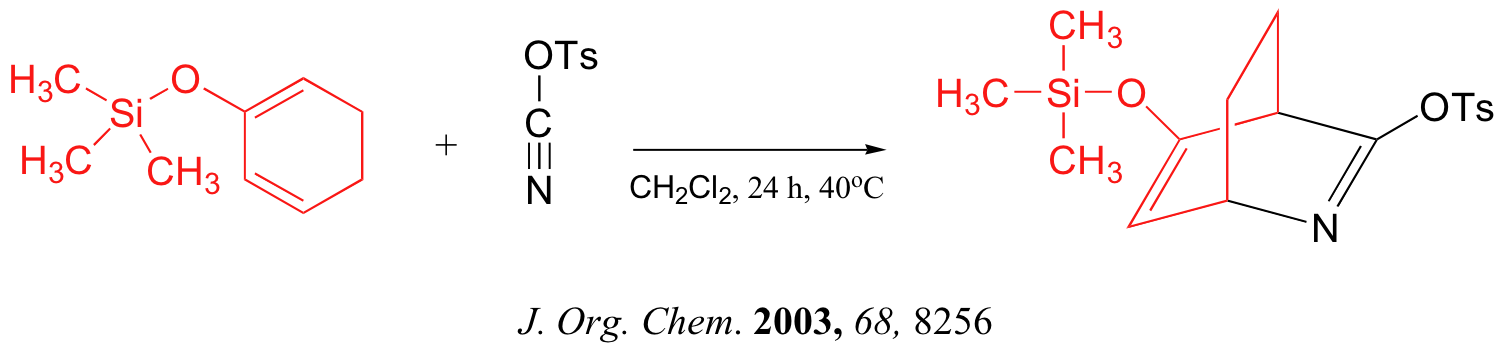
Below are just three examples of Diels-Alder reactions that have been reported in recent years:
The Diels-Alder reaction is just one example of a pericyclic reaction: this is a general term that refers to concerted rearrangements that proceed though cyclic transition states. Two well-studied intramolecular pericyclic reactions are known as the Cope rearrangement . . .
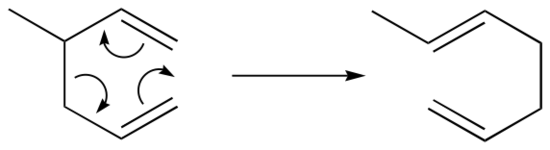
. . .and the Claisen rearrangement (when an oxygen is involved):
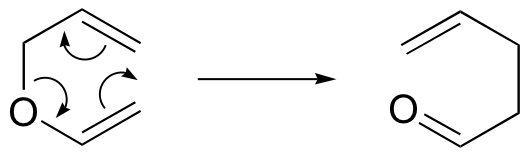
Notice that the both of these reactions require compounds in which two double bonds are separated by three single bonds.
Pericyclic reactions are rare in biological chemistry, but here is one example: the Claisen rearrangement catalyzed by chorismate mutase in the aromatic amino acid biosynthetic pathway.
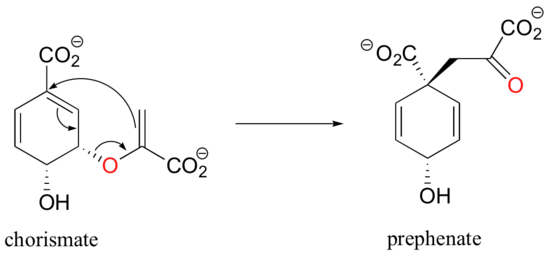
The study of pericyclic reactions is an area of physical organic chemistry that blossomed in the mid-1960s, due mainly to the work of R.B. Woodward, Roald Hoffman, and Kenichi Fukui. The Woodward-Hoffman rules for pericyclic reactions (and a simplified version introduced by Fukui) use molecular orbital theory to explain why some pericyclic processes take place and others do not. A full discussion is beyond the scope of this text, but if you go on to study organic chemistry at the advanced undergraduate or graduate level you are sure to be introduced to this fascinating area of inquiry.
Stereochemistry of the Diels-Alder reaction
We noted earlier that addition reactions of alkenes often exhibited stereoselectivity, in that the reagent elements in some cases added syn and in other cases anti to the the plane of the double bond. Both reactants in the Diels-Alder reaction may demonstrate stereoisomerism, and when they do it is found that the relative configurations of the reactants are preserved in the product (the adduct). The following drawing illustrates this fact for the reaction of 1,3-butadiene with (E)-dicyanoethene. The trans relationship of the cyano groups in the dienophile is preserved in the six-membered ring of the adduct. Likewise, if the terminal carbons of the diene bear substituents, their relative configuration will be retained in the adduct. Using the earlier terminology, we could say that bonding to both the diene and the dienophile is syn. An alternative description, however, refers to the planar nature of both reactants and terms the bonding in each case to be suprafacial (i.e. to or from the same face of each plane). This stereospecificity also confirms the synchronous nature of the 1,4-bonding that takes place.
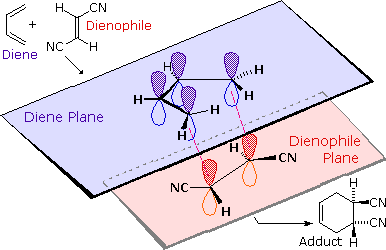
The essential characteristics of the Diels-Alder cycloaddition reaction may be summarized as follows:
- The reaction always creates a new six-membered ring. When intramolecular, another ring may also be formed.
- The diene component must be able to assume a s-cis conformation.
- Electron withdrawing groups on the dienophile facilitate reaction.
- Electron donating groups on the diene facilitate reaction.
- Steric hindrance at the bonding sites may inhibit or prevent reaction.
- The reaction is stereospecific with respect to substituent configuration in both the dienophile and the diene.
These features are illustrated by the following eight examples, one of which does not give a Diels-Alder cycloaddition.
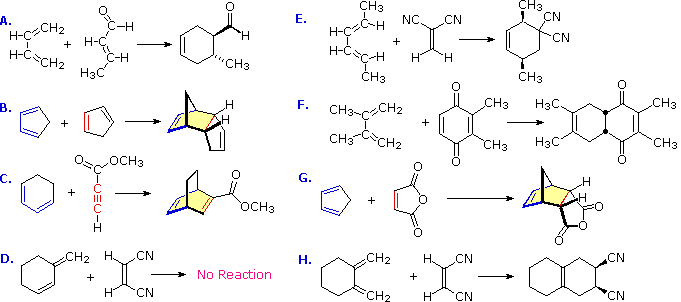
There is no reaction in example D because this diene cannot adopt an s-cis orientation. In examples B, C, F, G & H at least one of the reactants is cyclic so that the product has more than one ring, but the newly formed ring is always six-membered. In example B the the same cyclic compound acts as both the diene colored blue) and the dienophile (colored red). The adduct has three rings, two of which are the five-membered rings present in the reactant, and the third is the new six-membered ring (shaded light yellow). Example C has an alkyne as a dienophile (colored red), so the adduct retains a double bond at that location. This double bond could still serve as a dienophile, but in the present case the diene is sufficiently hindered to retard a second cycloaddition. The quinone dienophile in reaction F has two dienophilic double bonds. However, the double bond with two methyl substituents is less reactive than the unsubstituted dienophile due in part to the electron donating properties of the methyl groups and in part to steric hindrance. The stereospecificity of the Diels-Alder reaction is demonstrated by examples A, E & H. In A & H the stereogenic centers lie on the dienophile, whereas in E these centers are on the diene. In all cases the configuration of the reactant is preserved in the adduct.
Exercises
- Answer
-
Of the following dienes, which are s-trans and which are s-cis? Of those that are s-trans, are they able to rotate to become s-cis?
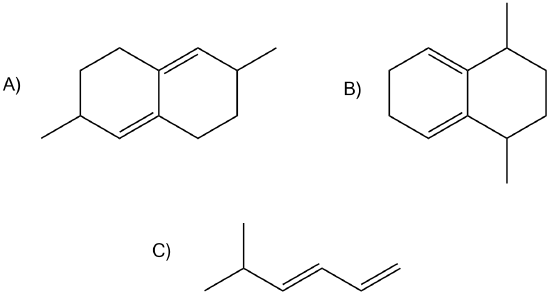
- Answer
-
A) s-trans, unable to rotate to become s-cis
B) s-cis
C) s-trans, can rotate to become s-cis.
Predict the product of the following reaction.
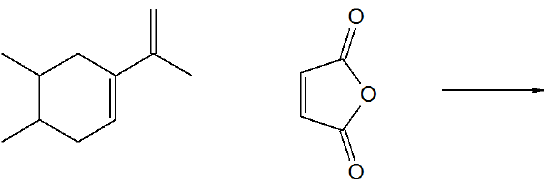
- Answer
-
.png?revision=1&size=bestfit&width=594&height=148)
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
Organic Chemistry With a Biological Emphasis by Tim Soderberg (University of Minnesota, Morris)