11.10: The E1 and E1cB Reactions
- Page ID
- 67271
Objectives
After completing this section, you should be able to
- write the mechanism for a typical E1 reaction.
- explain why E1 elimination often accompanies SN1 substitution.
- write an equation to describe the kinetics of an E1 reaction.
- discuss the stereochemistry of E1 reactions.
- account for the lack of a deuterium isotope effect in E1 reactions.
Make certain that you can define, and use in context, the key terms below.
- E1 reaction
- E1cB reaction
The abbreviation E1 stands for “unimolecular elimination”; that is, an E1 reaction is an elimination reaction in which only one species is involved in the rate-limiting step.
Unimolecular Elimination (E1) is a reaction in which the removal of an HX substituent results in the formation of a double bond. It is similar to a unimolecular nucleophilic substitution reaction (SN1) in various ways. One being the formation of a carbocation intermediate. Also, the only rate determining (slow) step is the dissociation of the leaving group to form a carbocation, hence the name unimolecular. Thus, since these two reactions behave similarly, they compete against each other. Many times, both these reactions will occur simultaneously to form different products from a single reaction. However, one can be favored over another through thermodynamic control. Although Elimination entails two types of reactions, E1 and E2, we will focus mainly on E1 reactions with some reference to E2.
General E1 Reaction
An E1 reaction involves the deprotonation of a hydrogen nearby (usually one carbon away, or the beta position) the carbocation resulting in the formation of an alkene product. In order to accomplish this, a Lewis base is required. For a simplified model, we’ll take B to be a Lewis base, and LG to be a halogen leaving group.
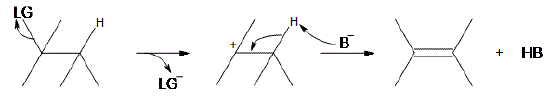
As can be seen above, the preliminary step is the leaving group (LG) leaving on its own. Because it takes the electrons in the bond along with it, the carbon that was attached to it loses its electron, making it a carbocation. Once it becomes a carbocation, a Lewis Base (\(B^-\)) deprotonates the intermediate carbocation at the beta position, which then donates its electrons to the neighboring C-C bond, forming a double bond. Unlike E2 reactions, which require the proton to be anti to the leaving group, E1 reactions only require a neighboring hydrogen. This is due to the fact that the leaving group has already left the molecule. The final product is an alkene along with the HB byproduct.
Reactivity
Due to the fact that E1 reactions create a carbocation intermediate, rules present in \(S_N1\) reactions still apply.
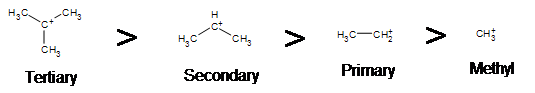
As expected, tertiary carbocations are favored over secondary, primary and methyl’s. This is due to the phenomena of hyperconjugation, which essentially allows a nearby C-C or C-H bond to interact with the p orbital of the carbon to bring the electrons down to a lower energy state. Thus, this has a stabilizing effect on the molecule as a whole. In general, primary and methyl carbocations do not proceed through the E1 pathway for this reason, unless there is a means of carbocation rearrangement to move the positive charge to a nearby carbon. Secondary and Tertiary carbons form more stable carbocations, thus this formation occurs quite rapidly.
Secondary carbocations can be subject to the E2 reaction pathway, but this generally occurs in the presence of a good / strong base. Adding a weak base to the reaction disfavors E2, essentially pushing towards the E1 pathway. In many instances, solvolysis occurs rather than using a base to deprotonate. This means heat is added to the solution, and the solvent itself deprotonates a hydrogen. The medium can effect the pathway of the reaction as well. Polar protic solvents may be used to hinder nucleophiles, thus disfavoring E2 / Sn2 from occurring.
How are Regiochemistry & Stereochemistry involved?
In terms of regiochemistry, Zaitsev's rule states that although more than one product can be formed during alkene synthesis, the more substituted alkene is the major product. This infers that the hydrogen on the most substituted carbon is the most probable to be deprotonated, thus allowing for the most substituted alkene to be formed.
Unlike E2 reactions, E1 is not stereospecific. Thus, a hydrogen is not required to be anti-periplanar to the leaving group.

In this mechanism, we can see two possible pathways for the reaction. One in which the methyl on the right is deprotonated, and another in which the CH2 on the left is deprotonated. Either one leads to a plausible resultant product, however, only one forms a major product. As stated by Zaitsev's rule, deprotonation of the most substituted carbon results in the most substituted alkene. This then becomes the most stable product due to hyperconjugation, and is also more common than the minor product.
The Connection Between SN1 and E1
The E1 mechanism is nearly identical to the SN1 mechanism, differing only in the course of reaction taken by the carbocation intermediate. As shown by the following equations, a carbocation bearing beta-hydrogens may function either as a Lewis acid (electrophile), as it does in the SN1 reaction, or a Brønsted acid, as in the E1 reaction.
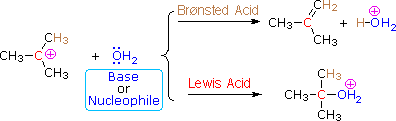
Thus, hydrolysis of tert-butyl chloride in a mixed solvent of water and acetonitrile gives a mixture of 2-methyl-2-propanol (60%) and 2-methylpropene (40%) at a rate independent of the water concentration. The alcohol is the product of an SN1 reaction and the alkene is the product of the E1 reaction. The characteristics of these two reaction mechanisms are similar, as expected. They both show first order kinetics; neither is much influenced by a change in the nucleophile/base; and both are relatively non-stereospecific.
(CH3)3C–Cl + H2O ——> [ (CH3)3C(+) ] + Cl(–) + H2O ——> (CH3)3C–OH + (CH3)2C=CH2 + HCl + H2O
To summarize, when carbocation intermediates are formed one can expect them to react further by one or more of the following modes:
- The cation may bond to a nucleophile to give a substitution product.
- The cation may transfer a beta-proton to a base, giving an alkene product.
- The cation may rearrange to a more stable carbocation, and then react by mode #1 or #2.
Since the SN1 and E1 reactions proceed via the same carbocation intermediate, the product ratios are difficult to control and both substitution and elimination usually take place.
Having discussed the many factors that influence nucleophilic substitution and elimination reactions of alkyl halides, we must now consider the practical problem of predicting the most likely outcome when a given alkyl halide is reacted with a given nucleophile. As we noted earlier, several variables must be considered, the most important being the structure of the alkyl group and the nature of the nucleophilic reactant. The nature of the halogen substituent on the alkyl halide is usually not very significant if it is Cl, Br or I. In cases where both SN2 and E2 reactions compete, chlorides generally give more elimination than do iodides, since the greater electronegativity of chlorine increases the acidity of beta-hydrogens. Indeed, although alkyl fluorides are relatively unreactive, when reactions with basic nucleophiles are forced, elimination occurs (note the high electronegativity of fluorine).
The E1cB Reaction
Although E1 reactions typically involves a carbocation intermediate, the E1cB reactoin utilizes a carbanion intermediate. This reaction is generally utilized when a poor leaving group, such an and alcohol, is involved. This poor leaving group makes the direct E1 or E2 reactions difficult. This reaction is used later in a reaction called an aldol condensation.
The product of this \(\beta\)-elimination reaction is an α,β-unsaturated aldehyde or ketone. Base-catalyzed elimination occurs with heating. The additional stability provided by the conjugated carbonyl system of the product makes some aldol reactions thermodynamically and mixtures of stereoisomers (E & Z) are obtained from some reactions.
Figure: General reaction for an E1cB condensation
Going from reactants to products simply

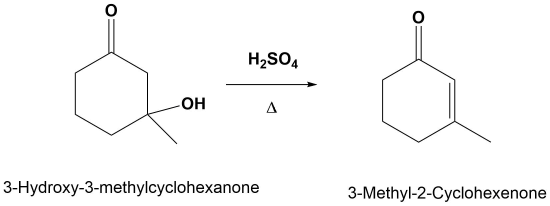
E1cB Mechanism
1) Form resonance stabilized anion
2) Form for conjugated alkene
Note! The double bond always forms in conjugation with the carbonyl.
Exercises
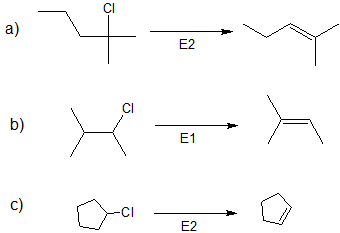
1. Predict the dominant elimination mechanism (E1 or E2) for each reaction below. Explain your reasoning.
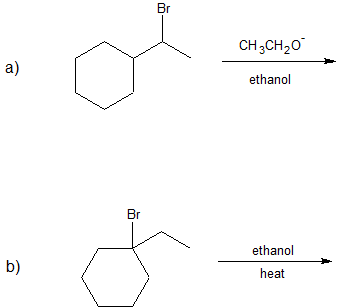
2) Specify the reaction conditions to favor the indicated elimination mechanism.
- Answer
-
1)

2)
a) strong base, such as hydroxide, an alkoxide, or equivalent
b) water or alcohol or equivalent weak base with heat
c) strong base, such as hydroxide, an alkoxide, or equivalent
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
Organic Chemistry With a Biological Emphasis by Tim Soderberg (University of Minnesota, Morris)

