11.3: Characteristics of the SN2 Reaction
- Page ID
- 67264
Objectives
After completing this section, you should be able to
- discuss the role of steric effects in SN2 reactions.
- arrange a given series of alkyl halides in order of increasing or decreasing reactivity towards nucleophilic substitution through the SN2 mechanism.
- suggest a reason why vinyl halides and aryl halides do not undergo SN2 reactions.
- discuss how the nature of the nucleophile affects the rate of an SN2 reaction.
- arrange a given series of common nucleophiles (e.g., CN−, I−, Br− Cl−, H2O) in order of increasing or decreasing nucleophilicity.
- discuss how the nature of the leaving group affects the rate of an SN2 reaction.
- arrange a given series of leaving groups in order of increasing or decreasing ability to leave during an SN2 reaction.
- discuss the role played by the solvent in an SN2 reaction.
- give examples of the solvents which are commonly used for SN2 reactions, and identify those that promote a high reaction rate.
- predict which of two given SN2 reactions will proceed faster, by taking into account the structure of the substrates, the nucleophiles involved, leaving-group ability, solvent effects, or any combination of these factors.
Make certain that you can define, and use in context, the key terms below.
- leaving group
- polar aprotic solvent
- solvation
You may wish to review the discussion of acid-base theory given in Sections 2.7-2.11.
Both aryl and vinylic halides are relatively unreactive in SN2 displacement mechanisms, mostly because during the backside attack of the molecule the incoming nucleophile is sterically hindered by both substituents and electron density from any double bonds present. Also, leaving groups on sp2-hybridized carbons tend to be held tighter than sp3-hybridized carbons.
Solvation may be defined as the interaction between molecules of solvent and particles of solute. The result of solvation is to stabilize (i.e., lower the energy of) the solute particles. Solvents with lone pairs of electrons are good at solvating cations. Protic (i.e., hydroxylic) solvents are able to solvate anions through hydrogen bonding. As water has two lone pairs of electrons and is also protic, it is good at solvating both anions and cations.
Bimolecular Nucleophilic Substitution Reactions Are Concerted
Bimolecular nucleophilic substitution (SN2) reactions are concerted, meaning they are a one step process. This means that the process whereby the nucleophile attacks and the leaving group leaves is simultaneous. Hence, the bond-making between the nucleophile and the electrophilic carbon occurs at the same time as the bond-breaking between the electophilic carbon and the halogen.
The potential energy diagram for an SN2 reaction is shown below. Upon nucleophilic attack, a single transition state is formed. A transition state, unlike a reaction intermediate, is a very short-lived species that cannot be isolated or directly observed. Again, this is a single-step, concerted process with the occurrence of a single transition state.

Sterically Hindered Substrates Will Reduce the SN2 Reaction Rate
Now that we have discussed the effects that the leaving group, nucleophile, and solvent have on biomolecular nucleophilic substitution (SN2) reactions, it's time to turn our attention to how the substrate affects the reaction. Although the substrate, in the case of nucleophilic substitution of haloalkanes, is considered to be the entire molecule circled below, we will be paying particular attention to the alkyl portion of the substrate. In other words, we are most interested in the electrophilic center that bears the leaving group.
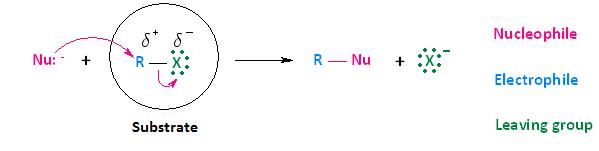
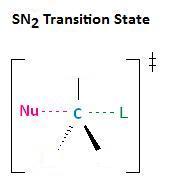
In the section Kinetics of Nucleophilic Substitution Reactions, we learned that the SN2 transition state is very crowded. Recall that there are a total of five groups around the electrophilic center, the nucleophile, the leaving group, and three substituents.
If each of the three substituents in this transition state were small hydrogen atoms, as illustrated in the first example below, there would be little steric repulsion between the incoming nucleophile and the electrophilic center, thereby increasing the ease at which the nucleophilic substitution reaction can occur. Remember, for the SN2 reaction to occur, the nucleophile must be able to attack the electrophilic center, resulting in the expulsion of the leaving group. If one of the hydrogens, however, were replaced with an R group, such as a methyl or ethyl group, there would be an increase in steric repulsion with the incoming nucleophile. If two of the hydrogens were replaced by R groups, there would be an even greater increase in steric repulsion with the incoming nucleophile.
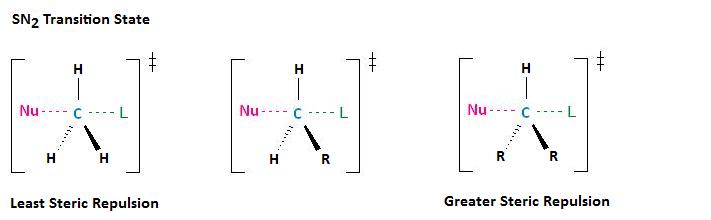
How does steric hindrance affect the rate at which an SN2 reaction will occur? As each hydrogen is replaced by an R group, the rate of reaction is significantly diminished. This is because the addition of one or two R groups shields the backside of the electrophilic carbon, impeding nucleophilic attack.
The diagram below illustrates this concept, showing that electrophilic carbons attached to three hydrogen atoms results in faster nucleophilic substitution reactions, in comparison to primary and secondary haloalkanes, which result in nucleophilic substitution reactions that occur at slower or much slower rates, respectively. Notice that a tertiary haloalkane, that which has three R groups attached, does not undergo nucleophilic substitution reactions at all. The addition of a third R group to this molecule creates a carbon that is entirely blocked.

Substitutes on Neighboring Carbons Slow Nucleophilic Substitution Reactions
Previously we learned that adding R groups to the electrophilic carbon results in nucleophilic substitution reactions that occur at a slower rate. What if R groups are added to neighboring carbons? It turns out that the addition of substitutes on neighboring carbons will slow nucleophilic substitution reactions as well.
In the example below, 2-methyl-1-bromopropane differs from 1-bromopropane in that it has a methyl group attached to the carbon that neighbors the electrophilic carbon. The addition of this methyl group results in a significant decrease in the rate of a nucleophilic substitution reaction.

If R groups were added to carbons farther away from the electrophilic carbon, we would still see a decrease in the reaction rate. However, branching at carbons farther away from the electrophilic carbon would have a much smaller effect.
What is a nucleophile?
Nucleophilic functional groups are those which have electron-rich atoms able to donate a pair of electrons to form a new covalent bond. In both laboratory and biological organic chemistry, the most relevant nucleophilic atoms are oxygen, nitrogen, and sulfur, and the most common nucleophilic functional groups are water, alcohols, phenols, amines, thiols, and occasionally carboxylates.
More specifically in laboratory reactions, halide and azide (N3-) anions are commonly seen acting as nucleophiles.
When thinking about nucleophiles, the first thing to recognize is that, for the most part, the same quality of 'electron-richness' that makes a something nucleophilic also makes it basic: nucleophiles can be bases, and bases can be nucleophiles. It should not be surprising, then, that most of the trends in basicity that we have already discussed also apply to nucleophilicity.
Some confusion in distinguishing basicity (base strength) and nucleophilicity (nucleophile strength) is inevitable. Since basicity is a less troublesome concept; it is convenient to start with it. Basicity refers to the ability of a base to accept a proton. Basicity may be related to the pKa of the corresponding conjugate acid, as shown below. The strongest bases have the weakest conjugate acids and vice versa. The range of basicities included in the following table is remarkable, covering over fifty powers of ten!
In an acid-base equilibrium the weakest acid and the weakest base will predominate (they will necessarily be on the same side of the equilibrium). Learning the pKa values for common compounds provides a useful foundation on which to build an understanding of acid-base factors in reaction mechanisms.
| Base | I (–) | Cl (–) | H2O | CH3CO2(–) | RS(–) | CN(–) | RO(–) | NH2(–) | CH3(–) |
|---|---|---|---|---|---|---|---|---|---|
| Conj. Acid | HI | HCl | H3O(+) | CH3CO2H | RSH | HCN | ROH | NH3 | CH4 |
| pKa | -9 | -7 | -1.7 | 4.8 | 8 | 9.1 | 16 | 33 | 48 |

Nucleophilicity is a more complex property. It commonly refers to the rate of substitution reactions at the halogen-bearing carbon atom of a reference alkyl halide, such as CH3-Br. Thus the nucleophilicity of the Nu:(–) reactant in the following substitution reaction varies as shown in the chart below:
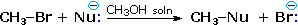 |
|---|
| Nucleophilicity: CH3CO2 (–) < Cl(–) < Br(–) < N3(–) < CH3O(–) < CN(–) < I(–) < CH3S(–) |
 |
Periodic trends and solvent effects in nucleophilicity
There are predictable periodic trends in nucleophilicity. Moving horizontally across the second row of the table, the trend in nucleophilicity parallels the trend in basicity:
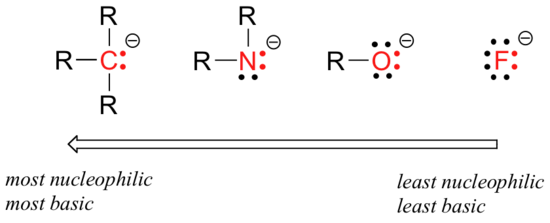
The reasoning behind the horizontal nucleophilicity trend is the same as the reasoning behind the basicity trend: more electronegative elements hold their electrons more tightly, and are less able to donate them to form a new bond. This horizontal trend also tells us that amines are more nucleophilic than alcohols, although both groups commonly act as nucleophiles in both laboratory and biochemical reactions.
Recall that the basicity of atoms decreases as we move vertically down a column on the periodic table: thiolate ions are less basic than alkoxide ions, for example, and bromide ion is less basic than chloride ion, which in turn is less basic than fluoride ion. Recall also that this trend can be explained by considering the increasing size of the 'electron cloud' around the larger ions: the electron density inherent in the negative charge is spread around a larger area, which tends to increase stability (and thus reduce basicity).
The vertical periodic trend for nucleophilicity is somewhat more complicated that that for basicity: depending on the solvent that the reaction is taking place in, the nucleophilicity trend can go in either direction. Let's take the simple example of the SN2 reaction below:
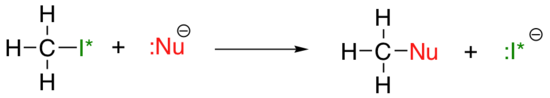
. . .where Nu- is one of the halide ions: fluoride, chloride, bromide, or iodide, and the leaving group I* is a radioactive isotope of iodine (which allows us to distinguish the leaving group from the nucleophile in that case where both are iodide). If this reaction is occurring in a protic solvent (that is, a solvent that has a hydrogen bonded to an oxygen or nitrogen - water, methanol and ethanol are the most important examples), then the reaction will go fastest when iodide is the nucleophile, and slowest when fluoride is the nucleophile, reflecting the relative strength of the nucleophile.

Relative nucleophilicity in a protic solvent
This of course, is opposite that of the vertical periodic trend for basicity, where iodide is the least basic. What is going on here? Shouldn't the stronger base, with its more reactive unbonded valence electrons, also be the stronger nucleophile?
As mentioned above, it all has to do with the solvent. Remember, we are talking now about the reaction running in a protic solvent like ethanol. Protic solvent molecules form very strong ion-dipole interactions with the negatively-charged nucleophile, essentially creating a 'solvent cage' around the nucleophile:

In order for the nucleophile to attack the electrophile, it must break free, at least in part, from its solvent cage. The lone pair electrons on the larger, less basic iodide ion interact less tightly with the protons on the protic solvent molecules - thus the iodide nucleophile is better able to break free from its solvent cage compared the smaller, more basic fluoride ion, whose lone pair electrons are bound more tightly to the protons of the cage.
The picture changes if we switch to a polar aprotic solvent, such as acetone, in which there is a molecular dipole but no hydrogens bound to oxygen or nitrogen. Now, fluoride is the best nucleophile, and iodide the weakest.
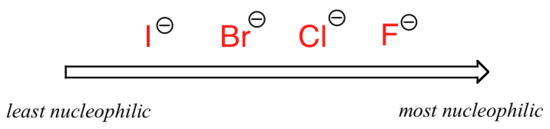
Relative nucleophilicity in a polar aprotic solvent
The reason for the reversal is that, with an aprotic solvent, the ion-dipole interactions between solvent and nucleophile are much weaker: the positive end of the solvent's dipole is hidden in the interior of the molecule, and thus it is shielded from the negative charge of the nucleophile.
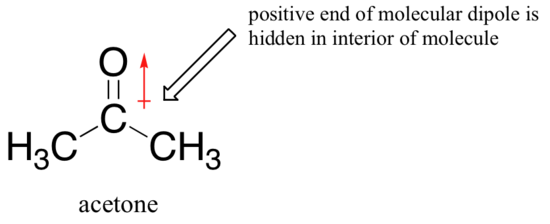
A weaker solvent-nucleophile interaction means a weaker solvent cage for the nucleophile to break through, so the solvent effect is much less important, and the more basic fluoride ion is also the better nucleophile.
Why not use a completely nonpolar solvent, such as hexane, for this reaction, so that the solvent cage is eliminated completely? The answer to this is simple - the nucleophile needs to be in solution in order to react at an appreciable rate with the electrophile, and a solvent such as hexane will not solvate an a charged (or highly polar) nucleophile at all. That is why chemists use polar aprotic solvents for nucleophilic substitution reactions in the laboratory: they are polar enough to solvate the nucleophile, but not so polar as to lock it away in an impenetrable solvent cage. In addition to acetone, three other commonly used polar aprotic solvents are acetonitrile, dimethylformamide (DMF), and dimethyl sulfoxide (DMSO).
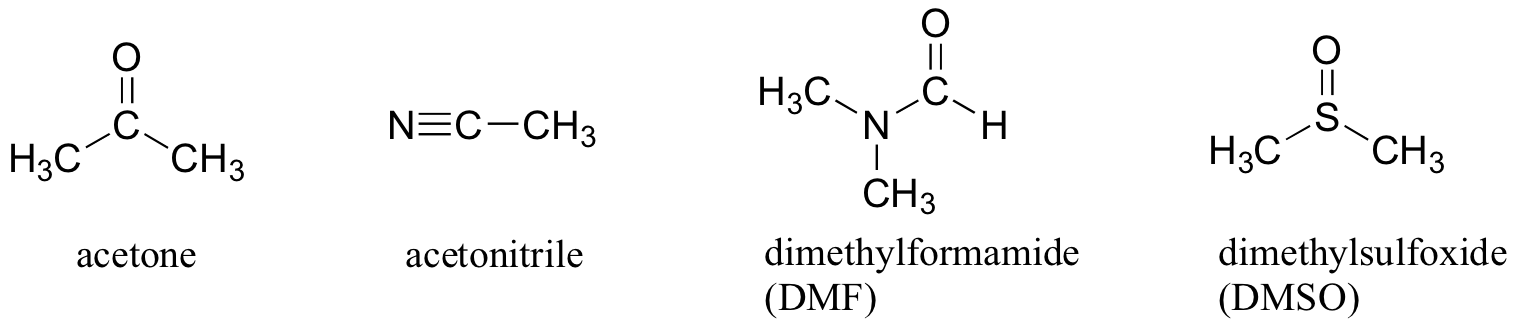
In biological chemistry, where the solvent is protic (water), the most important implication of the periodic trends in nucleophilicity is that thiols are more powerful nucleophiles than alcohols. The thiol group in a cysteine amino acid, for example, is a powerful nucleophile and often acts as a nucleophile in enzymatic reactions, and of course negatively-charged thiolates (RS-) are even more nucleophilic. This is not to say that the hydroxyl groups on serine, threonine, and tyrosine do not also act as nucleophiles - they do.
Resonance effects on nucleophilicity
Resonance effects also come into play when comparing the inherent nucleophilicity of different molecules. The reasoning involved is the same as that which we used to understand resonance effects on basicity. If the electron lone pair on a heteroatom is delocalized by resonance, it is inherently less reactive - meaning less nucleophilic, and also less basic. An alkoxide ion, for example, is more nucleophilic and more basic than a carboxylate group, even though in both cases the nucleophilic atom is a negatively charged oxygen. In the alkoxide, the negative charge is localized on a single oxygen, while in the carboxylate the charge is delocalized over two oxygen atoms by resonance.

The nitrogen atom on an amide is less nucleophilic than the nitrogen of an amine, due to the resonance stabilization of the nitrogen lone pair provided by the amide carbonyl group.

Influence of the solvent in an SN2 reaction
The rate of an SN2 reaction is significantly influenced by the solvent in which the reaction takes place. The use of protic solvents (those, such as water or alcohols, with hydrogen-bond donating capability) decreases the power of the nucleophile, because of strong hydrogen-bond interactions between solvent protons and the reactive lone pairs on the nucleophile. A less powerful nucleophile in turn means a slower SN2 reaction. SN2 reactions are faster in polar, aprotic solvents: those that lack hydrogen-bond donating capability. Below are several polar aprotic solvents that are commonly used in the laboratory:
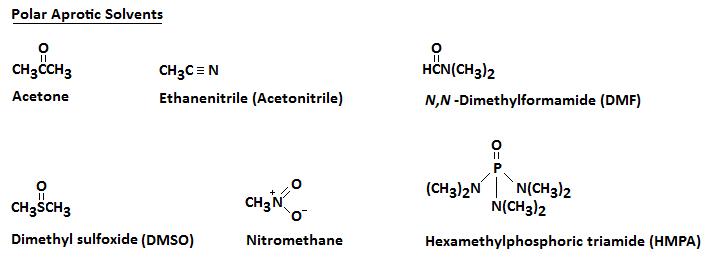
These aprotic solvents are polar but, because they do not form hydrogen bonds with the anionic nucleophile, there is a relatively weak interaction between the aprotic solvent and the nucleophile. By using an aprotic solvent we can raise the reactivity of the nucleophile. This can sometimes have dramatic effects on the rate at which a nucleophilic substitution reaction can occur. For example, if we consider the reaction between bromoethane and potassium iodide, the reaction occurs 500 times faster in acetone than in methanol.
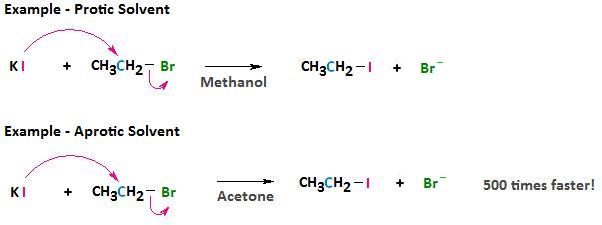
The leaving group
As Electronegativity Increases, Basicity Decreases: In general, if we move from the left of the periodic table to the right of the periodic table as shown in the diagram below, electronegativity increases. As electronegativity increases, basicity will decrease, meaning a species will be less likely to act as base; that is, the species will be less likely to share its electrons.

As Size Increases, Basicity Decreases:In general, if we move from the top of the periodic table to the bottom of the periodic table as shown in the diagram below, the size of an atom will increase. As size increases, basicity will decrease, meaning a species will be less likely to act as a base; that is, the species will be less likely to share its electrons.
Resonance Decreases Basicity:The third factor to consider in determining whether or not a species will be a strong or weak base is resonance. As you may remember from general chemistry, the formation of a resonance stabilized structure results in a species that is less willing to share its electrons. Since strong bases, by definition, want to share their electrons, resonance stabilized structures are weak bases.
Now that we understand how electronegativity, size, and resonance affect basicity, we can combine these concepts with the fact that weak bases make the best leaving groups. Think about why this might be true. In order for a leaving group to leave, it must be able to accept electrons. A strong bases wants to donate electrons; therefore, the leaving group must be a weak base. We will now revisit electronegativity, size, and resonance, moving our focus to the leaving group, as well providing actual examples.
As Electronegativity Increases, The Ability of the Leaving Group to Leave Increases
As mentioned previously, if we move from left to right on the periodic table, electronegativity increases. With an increase in electronegativity, basisity decreases, and the ability of the leaving group to leave increases. This is because an increase in electronegativity results in a species that wants to hold onto its electrons rather than donate them. The following diagram illustrates this concept, showing -CH3 to be the worst leaving group and F- to be the best leaving group. This particular example should only be used to facilitate your understanding of this concept. In real reaction mechanisms, these groups are not good leaving groups at all. For example, fluoride is such a poor leaving group that SN2 reactions of fluoroalkanes are rarely observed.
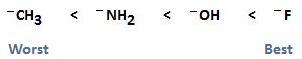
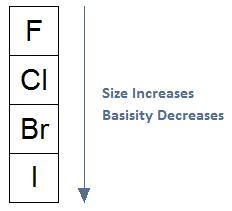
As Size Increases, The Ability of the Leaving Group to Leave Increases:Here we revisit the effect size has on basicity. If we move down the periodic table, size increases. With an increase in size, basicity decreases, and the ability of the leaving group to leave increases. The relationship among the following halogens, unlike the previous example, is true to what we will see in upcoming reaction mechanisms.

In each pair (A and B) below, which electrophile would be expected to react more rapidly in an SN2 reaction with the thiol group of cysteine as the common nucleophile?
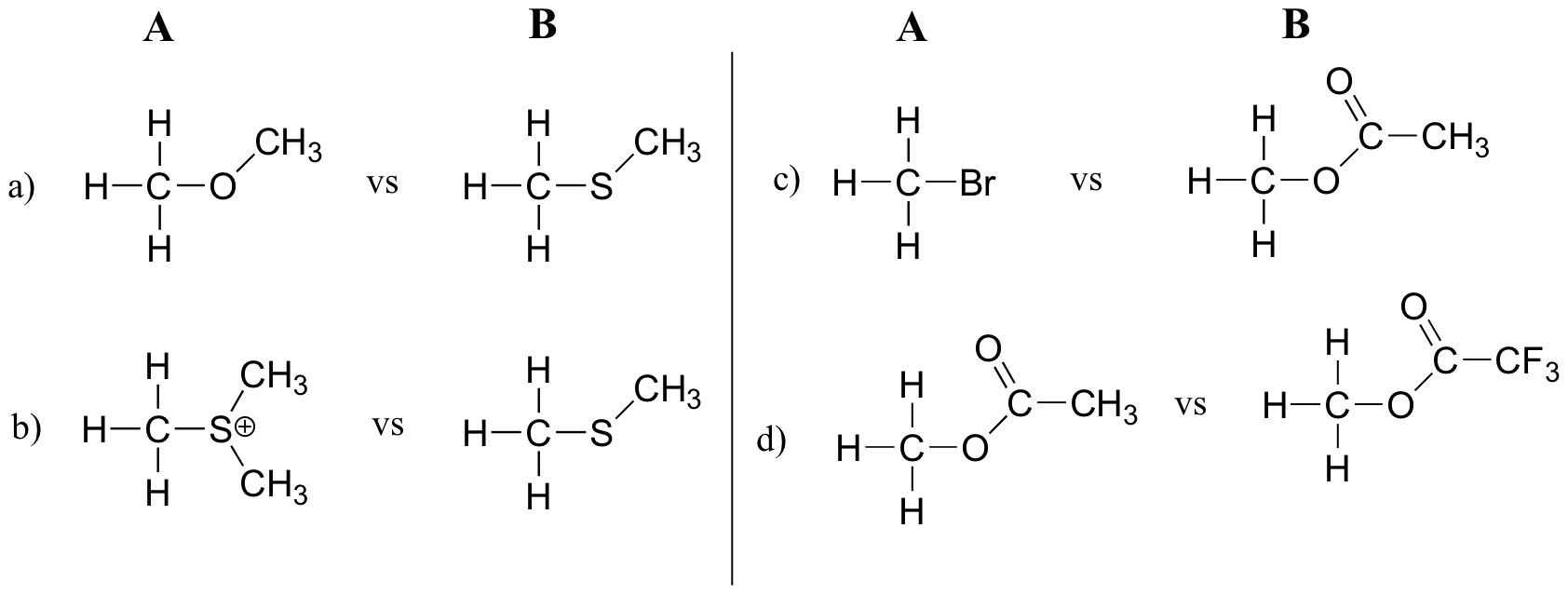
Solution
a) B (thiolate is a weaker base/better leaving group than alcohol)
b) A (sulfide is better leaving group than thiolate (sulfide is neutral and not at all basic)
c) A (bromide ion is a weaker base/better leaving group than acetate – compare pKa values of HBr and acetic acid).
d) B (inductive electron-withdrawing effect of fluorines make trifluoroacetate a weaker base/better leaving group)
Exercises
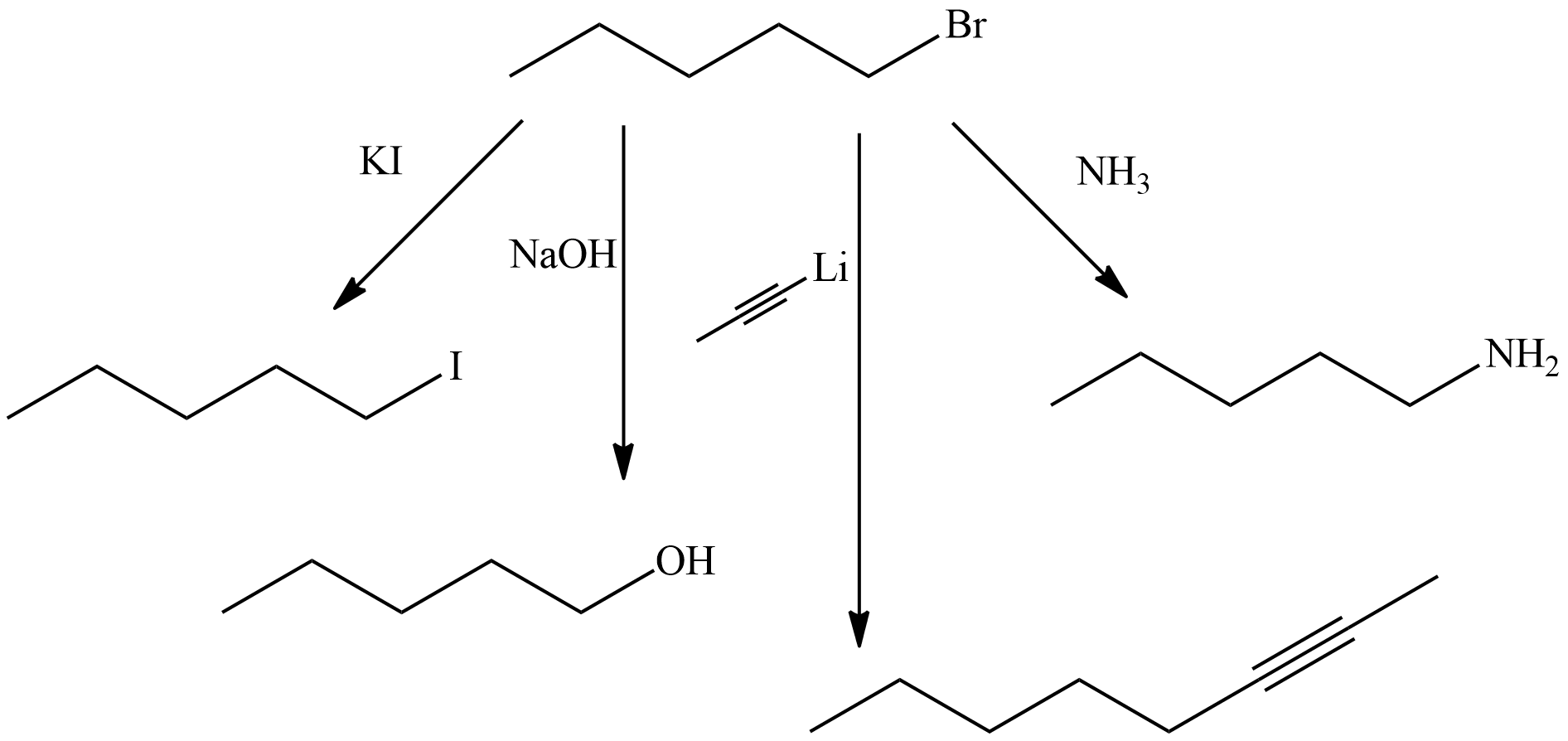
What product(s) do you expect from the reaction of 1-bromopentane with each of the following reagents in an SN2 reaction?
a) KI
b) NaOH
c) CH3C≡C-Li
d) NH3
- Answer
-
Which in the following pairs is a better nuceophile?
a) (CH3CH2)2N- or (CH3CH2)2NH
b) (CH3CH2)3N or (CH3CH2)3B
c) H2O or H2S
- Answer
-
a) (CH3CH2)2N- as there is a charge present on the nitrogen.
b) (CH3CH2)3N because a lone pair of electrons is present.
c) H2S as nucleophilicity generally increases going down the periodic table. [Assumes the absence of a polar aprotic solvent.]
Order the following in increasing reactivity for an SN2 reaction.
CH3CH2Br CH3CH2OTos (CH3CH2)3CCl (CH3CH2)2CHCl
- Answer
-
.png?revision=1&size=bestfit&width=464&height=95)
Solvents benzene, ether, chloroform are non-polar and not strongly polar solvents. What effects do these solvents have on an SN2 reaction?
- Answer
-
They will decrease the reactivity of the reaction.
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
Organic Chemistry With a Biological Emphasis by Tim Soderberg (University of Minnesota, Morris)
Jim Clark (Chemguide.co.uk)

