11.1: The Discovery of Nucleophilic Substitution Reactions
- Page ID
- 67262
Objectives
After completing this section, you should be able to
- write an equation to represent the Walden inversion.
- write a short paragraph describing the Walden inversion.
- describe, using equations, a series of reactions interconverting two enantiomers of 1-phenyl-2-propanol which led to the conclusion that nucleophilic substitution of primary and secondary alkyl halides proceeds with inversion of configuration.
The IUPAC name for malic acid is 2-hydroxybutanedioic acid. This acid is produced by apples, a fact which seems to have been appreciated by the British novelist Thomas Hardy in The Woodlanders:
Up, upward they crept, a stray beam of the sun alighting every now and then like a star on the blades of the pomace-shovels, which had been converted to steel mirrors by the action of the malic acid.
In 1896, the German chemist Paul Walden discovered that he could interconvert pure enantiomeric (+) and (-) malic acids through a series of reactions. This conversion meant that there was some kind of change in the stereo chemistry made during the reaction.
There reactions are currently referred to as nucleophilic substitution reaction because each step involves the substitution of one nucleophile by another. Therese reactions are one of the most common and versatile reaction types in organic chemistry.
Further investigations into these reaction were under taken during the 1920's and 1930's to clarify the mechanism and clarify how the inversion of configurations occur. These reactions involved the nucleophilic substitution of an alkyl p-toluenesulfonate (called a tosylate group). For this purpose the tosylate groups acts as a halogen substituent. In the series of reactions (+)-1-phenyl-2-propanol is interconverted with (-)-1-phenyl-2-propanol.
It was determined that the reaction with acetate was causing the stereochemical configuration to be inverted.
Exercise
1) Predict the product of a nucleophilic substitution of (S)-2-bromopentane reacting with CH3CO2-, Show stereochemistry.
- Answer
-
1)
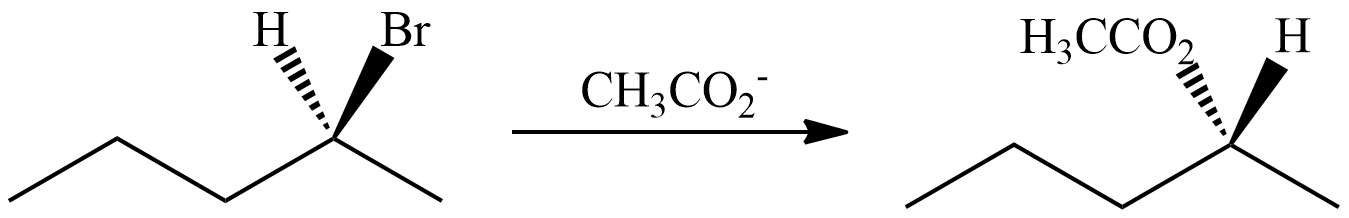
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)

