10.2: Preparing Alkyl Halides from Alkanes - Radical Halogenation
- Page ID
- 67252
After completing this section, you should be able to
- explain why the radical halogenation of alkanes is not usually a particularly good method of preparing pure samples of alkyl halides.
- use \(\ce{\sf{C–H}}\) bond energies to account for the fact that in radical chlorinations, the reactivity of hydrogen atoms decreases in the order
tertiary > secondary > primary.
- predict the approximate ratio of the expected products from the monochlorination of a given alkane.
The following terms are synonymous:
- methyl hydrogens, primary hydrogens, and 1° hydrogens.
- methylene hydrogens, secondary hydrogens, and 2° hydrogens.
- methine hydrogens, tertiary hydrogens, and 3° hydrogens.
Note that in radical chlorination reactions, the reactivity of methine, methylene and methyl hydrogens decreases in the ratio of approximately 5 : 3.5 : 1. This will aid in the prediction of expected products from the monochlorination of a given alkane.
Methane and chlorine
If a mixture of methane and chlorine is exposed to a flame, it explodes - producing carbon and hydrogen chloride. This is not a very useful reaction! The reaction we are going to explore is a more gentle one between methane and chlorine in the presence of ultraviolet light - typically sunlight. This is a good example of a photochemical reaction - a reaction brought about by light.
\[CH_4 + Cl_2 + energy → CH_3Cl + HCl\]
The organic product is chloromethane. One of the hydrogen atoms in the methane has been replaced by a chlorine atom, so this is a substitution reaction. However, the reaction doesn't stop there, and all the hydrogens in the methane can in turn be replaced by chlorine atoms. Multiple substitution is dealt with on a separate page, and you will find a link to that at the bottom of this page.
Substitution reactions happen in which hydrogen atoms in the methane are replaced one at a time by chlorine atoms. You end up with a mixture of chloromethane, dichloromethane, trichloromethane and tetrachloromethane.
![]()
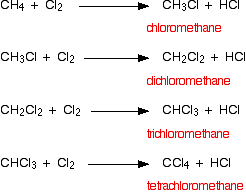
The original mixture of a colorless and a green gas would produce steamy fumes of hydrogen chloride and a mist of organic liquids. All of the organic products are liquid at room temperature with the exception of the chloromethane which is a gas.
If you were using bromine, you could either mix methane with bromine vapor, or bubble the methane through liquid bromine - in either case, exposed to UV light. The original mixture of gases would, of course, be red-brown rather than green.
You wouldn't choose to use these reactions as a means of preparing these organic compounds in the lab because the mixture of products would be too tedious to separate. The mechanisms for the reactions are explained on separate pages.
Alkanes (the most basic of all organic compounds) undergo very few reactions. One of these reactions is halogenation, or the substitution of a single hydrogen on the alkane for a single halogen to form a haloalkane. This reaction is very important in organic chemistry because it opens a gateway to further chemical reactions.
Halogenation Reaction
While the reactions possible with alkanes are few, there are many reactions that involve haloalkanes. In order to better understand the mechanism (a detailed look at the step by step process through which a reaction occurs), we will closely examine the chlorination of methane. When methane (CH4) and chlorine (Cl2) are mixed together in the absence of light at room temperature nothing happens. However, if the conditions are changed, so that either the reaction is taking place at high temperatures (denoted by Δ) or there is ultra violet irradiation, a product is formed, chloromethane (CH3Cl).
Energetics
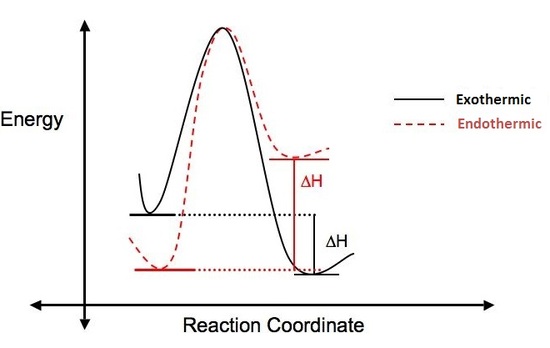
Why does this reaction occur? Is the reaction favorable? A way to answer these questions is to look at the change in enthalpy (\(\Delta{H}\)) that occurs when the reaction takes place.
ΔH = (Energy put into reaction) – (Energy given off from reaction)
If more energy is put into a reaction than is given off, the ΔH is positive, the reaction is endothermic and not energetically favorable. If more energy is given off in the reaction than was put in, the ΔH is negative, the reaction is said to be exothermic and is considered favorable. The figure below illustrates the difference between endothermic and exothermic reactions.
ΔH can also be calculated using bond dissociation energies (ΔH°):
\[\Delta{H} = \sum \Delta{H^°} \text{ of bonds broken} - \sum \Delta{H^°} \text{ of bonds formed}\]
Let’s look at our specific example of the chlorination of methane to determine if it is endothermic or exothermic:
.jpg?revision=2&size=bestfit&width=386&height=228)
Since, the ΔH for the chlorination of methane is negative, the reaction is exothermic. Energetically this reaction is favorable. In order to better understand this reaction we need to look at the mechanism ( a detailed step by step look at the reaction showing how it occurs) by which the reaction occurs.
Radical Chain Mechanism
The reaction proceeds through the radical chain mechanism. The radical chain mechanism is characterized by three steps: initiation, propagation and termination. Initiation requires an input of energy but after that the reaction is self-sustaining. The first propagation step uses up one of the products from initiation, and the second propagation step makes another one, thus the cycle can continue until indefinitely.
Step 1: Initiation
Initiation breaks the bond between the chlorine molecule (Cl2). For this step to occur energy must be put in, this step is not energetically favorable. After this step, the reaction can occur continuously (as long as reactants provide) without input of more energy. It is important to note that this part of the mechanism cannot occur without some external energy input, through light or heat.
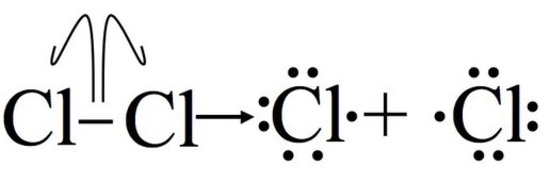
Step 2: Propagation
The next two steps in the mechanism are called propagation steps. In the first propagation step, a chlorine radical combines with a hydrogen on the methane. This gives hydrochloric acid (HCl, the inorganic product of this reaction) and the methyl radical. In the second propagation step more of the chlorine starting material (Cl2) is used, one of the chlorine atoms becomes a radical and the other combines with the methyl radical.
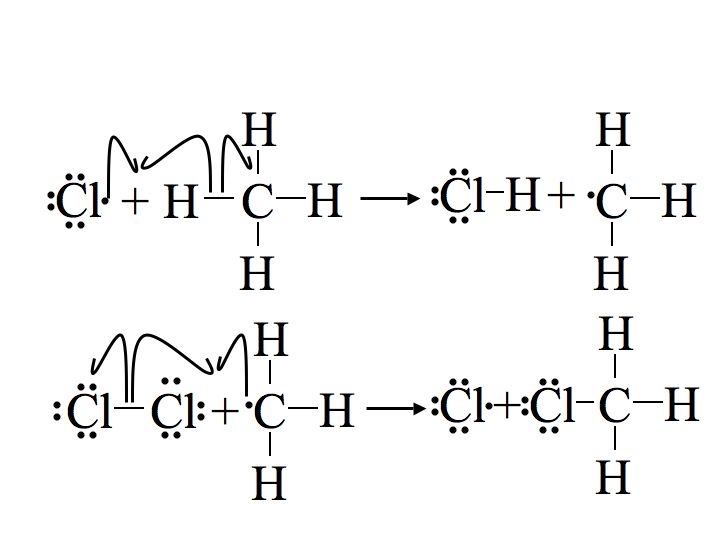
The first propagation step is endothermic, meaning it takes in heat (requires 2 kcal/mol) and is not energetically favorable. In contrast the second propagation step is exothermic, releasing 27 kcal/mol. Since the second propagation step is so exothermic, it occurs very quickly. The second propagation step uses up a product from the first propagation step (the methyl radical) and following Le Chatelier's principle, when the product of the first step is removed the equilibrium is shifted towards it's products. This principle is what governs the unfavorable first propagation step's occurance.
.gif?revision=2&size=bestfit&width=452&height=339)
Step 3: Termination
In the termination steps, all the remaining radicals combine (in all possible manners) to form more product (CH3Cl), more reactant (Cl2) and even combinations of the two methyl radicals to form a side product of ethane (CH3CH3).
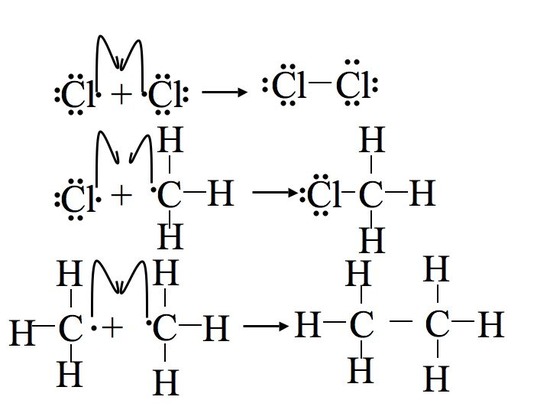
Problems with the Chlorination of Methane
The chlorination of methane does not necessarily stop after one chlorination. It may actually be very hard to get a monosubstituted chloromethane. Instead di-, tri- and even tetra-chloromethanes are formed. One way to avoid this problem is to use a much higher concentration of methane in comparison to chloride. This reduces the chance of a chlorine radical running into a chloromethane and starting the mechanism over again to form a dichloromethane. Through this method of controlling product ratios one is able to have a relative amount of control over the product.
Chlorination of other alkanes
When alkanes larger than ethane are halogenated, isomeric products are formed. Thus chlorination of propane gives both 1-chloropropane and 2-chloropropane as mono-chlorinated products. Four constitutionally isomeric dichlorinated products are possible, and five constitutional isomers exist for the trichlorinated propanes. Can you write structural formulas for the four dichlorinated isomers?
\[CH_3CH_2CH_3 + 2Cl_2 \rightarrow \text{Four} \; C_3H_6Cl_2 \; \text{isomers} + 2 HCl\]
The halogenation of propane discloses an interesting feature of these reactions. All the hydrogens in a complex alkane do not exhibit equal reactivity. For example, propane has eight hydrogens, six of them being structurally equivalent primary, and the other two being secondary. If all these hydrogen atoms were equally reactive, halogenation should give a 3:1 ratio of 1-halopropane to 2-halopropane mono-halogenated products, reflecting the primary/secondary numbers. This is not what we observe. Light-induced gas phase chlorination at 25 ºC gives 45% 1-chloropropane and 55% 2-chloropropane.
CH3-CH2-CH3 + Cl2 → 45% CH3-CH2-CH2Cl + 55% CH3-CHCl-CH3
These results suggest strongly that 2º-hydrogens are inherently more reactive than 1º-hydrogens, by a factor of about 3:1. Further experiments showed that 3º-hydrogens are even more reactive toward halogen atoms. Thus, light-induced chlorination of 2-methylpropane gave predominantly (65%) 2-chloro-2-methylpropane, the substitution product of the sole 3º-hydrogen, despite the presence of nine 1º-hydrogens in the molecule.
(CH3)3CH + Cl2 → 65% (CH3)3CCl + 35% (CH3)2CHCH2Cl
If you are uncertain about the terms primary (1º), secondary (2º) & tertiary (3º) Click Here.
It should be clear from a review of the two steps that make up the free radical chain reaction for halogenation that the first step (hydrogen abstraction) is the product determining step. Once a carbon radical is formed, subsequent bonding to a halogen atom (in the second step) can only occur at the radical site. Consequently, an understanding of the preference for substitution at 2º and 3º-carbon atoms must come from an analysis of this first step.
First Step: R3CH + X· → R3C· + H-X
Second Step: R3C· + X2 → R3CX + X·
Since the H-X product is common to all possible reactions, differences in reactivity can only be attributed to differences in C-H bond dissociation energies. In our previous discussion of bond energy we assumed average values for all bonds of a given kind, but now we see that this is not strictly true. In the case of carbon-hydrogen bonds, there are significant differences, and the specific dissociation energies (energy required to break a bond homolytically) for various kinds of C-H bonds have been measured. These values are given in the following table.
| R (in R–H) | methyl | ethyl | i-propyl | t-butyl | phenyl | benzyl | allyl | vinyl |
|---|---|---|---|---|---|---|---|---|
| Bond Dissociation Energy (kcal/mole) |
103 | 98 | 95 | 93 | 110 | 85 | 88 | 112 |
The difference in C-H bond dissociation energy reported for primary (1º), secondary (2º) and tertiary (3º) sites agrees with the halogenation observations reported above, in that we would expect weaker bonds to be broken more easily than are strong bonds. By this reasoning we would expect benzylic and allylic sites to be exceptionally reactive in free radical halogenation, as experiments have shown. The methyl group of toluene, C6H5CH3, is readily chlorinated or brominated in the presence of free radical initiators (usually peroxides), and ethylbenzene is similarly chlorinated at the benzylic location exclusively. The hydrogens bonded to the aromatic ring (referred to as phenyl hydrogens above) have relatively high bond dissociation energies and are not substituted.
C6H5CH2CH3 + Cl2 → C6H5CHClCH3 + HCl
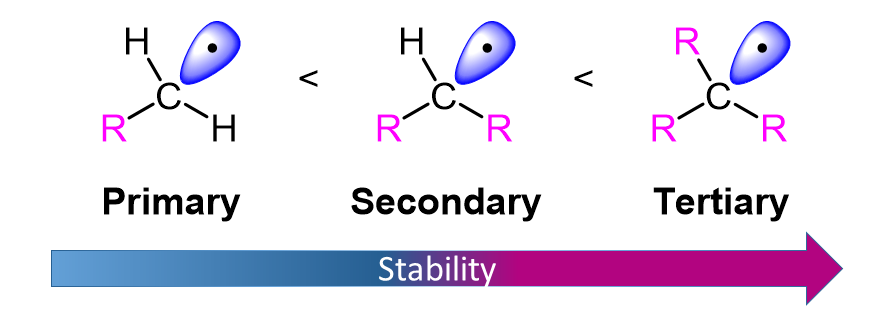
Relative Stability of Free Radicals
Exercises
Write out the complete mechanism for the chlorination of methane.
- Answer
-
The answer to this problem is actually above in the initiation, propagation and termination descriptions.
Explain, in your own words, how the first propagation step can occur without input of energy if it is energetically unfavorable.
- Answer
-
Since the second step in propagation is energetically favorable and fast, it drives the equilibrium toward products, even though the first step is not favorable.
Which step of the radical chain mechanism requires outside energy? What can be used as this energy?
- Answer
-
Initiation step requires energy which can be in the form of light or het.
Having learned how to calculate the change in enthalpy for the chlorination of methane apply your knowledge and using the table provided below calculate the change in enthalpy for the bromination of ethane.
| Compound | Bond Dissociation Energy (kcal/mol) |
| CH3CH2-H | 101 |
| CH3CH2-Br | 70 |
| H-Br | 87 |
| Br2 | 46 |
- Answer
-
To calculate the enthalpy of reaction, you subtract the BDE of the bonds formed from the BDE of the bonds broken.
Bonds broken are C-H and Br-Br.
Bonds formed are H-Br adn C-Br.
Bonds broken - bonds formed = change in enthalpy
(101 kcal/mol + 46 kcal/mol) - (87 kcal/mol + 70 kcal/mol) = change in enthalpy
-10 kcal/mol = change in enthalpy for bromination of ethane.
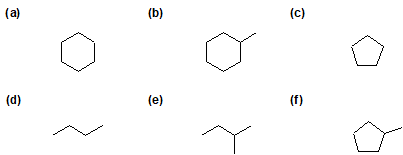
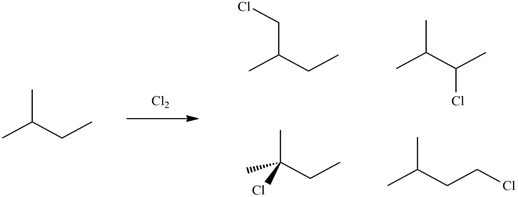
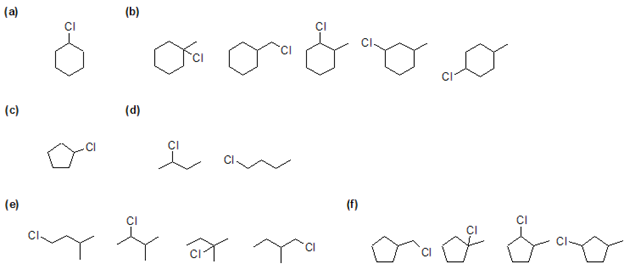
1) Predict the mono-substituted halogenated product(s) of chlorine gas reacting with 2-methylbutane.
2) For the following compounds, give all possible monochlorinated derivatives.
- Answer
-
1)
2)
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
Jim Clark (Chemguide.co.uk)
- Kristen Kelley and Britt Farquharson

