Answers to Chapter 08 & 09 Study Questions
- Page ID
- 11889
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\)
-
- 6
- 1
- 3 (two s electrons and one p electron)
- in order of increasing electronegativity: \(\mathrm{Ge < C < O}\)
-
- \(\ce{Li+}\)
- \(\ce{Na+}\)
- \(\ce{F}\)
-
- \(\ce{O^2-}\), \(\ce{F-}\), \(\ce{Na+}\), \(\ce{Mg^2+}\), \(\ce{Al^3+}\)
- \(\ce{S^2-}\), \(\ce{Cl-}\), \(\ce{K+}\), \(\ce{Ca^2+}\), \(\ce{Sc^3+}\)
- \(\ce{I-}\), \(\ce{Cs+}\), \(\ce{Ba^2+}\)
- isoelectronic
-
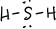
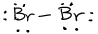
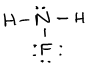
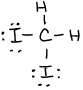
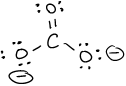
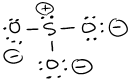
- \(\ce{P2Cl2}\)
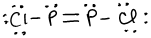
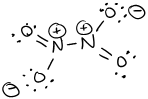
- \(\ce{N2O4}\)
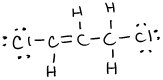
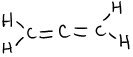 or
or 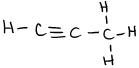
-
- bent, polar
- linear, nonpolar
- trigonal pyramid, polar
- tetrahedral, polar
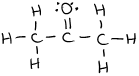 or
or 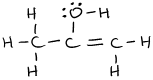
- Covalent bonds involve the sharing of electrons between two atoms (usually nonmetals). The orbitals of bonding electrons overlap so that the bonding electrons spend more time between the 2 atoms. A bond is polar when one of the atoms is more electronegative than the other atom. A molecule is polar when the overall molecule has a net dipole moment, that is, electrons spend more time on one side of the molecule than the other.
- Chemical properties are shared within a Group but not within a Period. Group number is a good predictor of chemical properties; Period number is not.
-
- \(\mathrm{C - F}\)
- \(\mathrm{Si - F}\)
-
- see-saw
- square planar
- trigonal bipyramid
- octahedral
-
- sp3
- sp
- sp3d2
- sp3d
- sp2
- sp2
-
- 4 sigma bonds
- 3 sigma bonds, 1 pi bond
- 2 sigma bonds, 2 pi bonds
- 9 sigma bonds, 1 pi bond

