7.1: Structure and Composition of the Atmosphere
- Last updated
- Save as PDF
- Page ID
- 291393
Life as we know it on the Earth is entirely dependent on the tenuous layer of gas that clings to the surface of the globe, adding about 1% to its diameter and an insignificant amount to its total mass. And yet the atmosphere serves as the earth’s window and protective shield, as a medium for the transport of heat and water, and as source and sink for exchange of carbon, oxygen, and nitrogen with the biosphere. The atmosphere acts as a compressible fluid tied to the earth by gravitation; as a receptor of solar energy and a thermal reservoir, it constitutes the working fluid of a heat engine that transports and redistributes matter and energy over the entire globe. The atmosphere is also a major temporary repository of a number of chemical elements that move in a cyclic manner between the hydrosphere, atmosphere, and the upper lithosphere. Finally, the atmosphere is a site for a large variety of complex photochemically initiated reactions involving both natural and anthropogenic substances.
On the scale of cubic meters the air is a homogeneous mixture of its constituent gases, but on a larger scale the atmosphere is anything but uniform. Variations of temperature, pressure, and moisture content in the layers of air near the earth’s surface give rise to the dynamic effects we know as the weather.
Although the density of the atmosphere decreases without limit with increasing height, for most practical purposes one can roughly place its upper boundary at about 500 km. However, half the mass of the atmosphere lies within 5 km, and 99.99% within 80 km of the surface. The average atmospheric pressure at sea level is 1.01 x 105 pascals, or 1010 millibars. A 1-cm2 cross section of the earth’s surface supports a column weighing 1030 g; the total mass of the atmosphere is about 5.27 x 1021 g.
About 80% of the mass of the atmosphere resides in the first 10 km; this well-mixed region of fairly uniform composition is known as the troposphere.
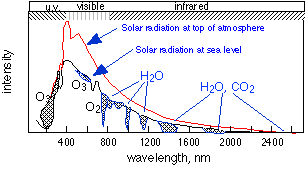
Solar irradiation of the Earth
The gases ozone, water vapor, and carbon dioxide are only minor components of the atmosphere, but they exert a huge effect on the Earth by absorbing radiation in the ranges indicated by the shading. Ozone in the upper atmosphere filters out the ultraviolet light below about 360 nm that is destructive of life. O2, H2O, CO2 and CH4 are "greenhouse" gases that trap some of the heat absorbed from the Sun and prevent it from re-radiating back into space.
Structure of the atmosphere
We commonly think of gas molecules as moving about in a completely random manner, but the Earth’s gravitational field causes downward motions to be very slightly favored so that the molecules in any thin layer of the air collide more frequently with those in the layer below. This gives rise to a pressure gradient that is the most predictable and well known structural characteristic of the atmosphere. This gradient is described by an exponential law which predicts that the atmospheric pressure should decrease by 50% for every 6 km increase in altitude. This law also predicts that the composition of a gas mixture will change with altitude, the lower-molecular weight components being increasingly favored at higher altitudes. However, this gravitational fractionation effect is completely obliterated below about 160 km owing to turbulence and convective flows (winds).
The atmosphere is divided vertically into several major regions which are distinguished by the sign of the temperature gradient. In the lowermost region, the troposphere, the temperature falls with increasing altitude. The major source of heat input into this part of the atmosphere is long-wave radiation from the earth’s surface, while the major loss is radiation into space.
At higher elevations the temperature begins to rise with altitude as we move into a region in which heat is produced by exothermic chemical reactions, mainly the decomposition of ozone that is formed photochemically from dioxygen in the stratosphere. At still higher elevations the ozone gives out and the temperature begins to drop; this is the mesosphere, which is finally replaced by the thermosphere which consists largely of a plasma (gaseous ions). This outer section of the atmosphere which extends indefinitely to perhaps 2000 km is heated by absorption of intense u.v. radiation from the Sun and also from the solar wind, a continual rain of electrons, protons, and other particles emitted from the Sun’s surface.

Structure of the atmosphere. The main divisions of the atmosphere are defined by the elevations at which the sign of the temperature gradient changes. The chemical formulas at the right show the major species of interest in the various regions. The shaded D- and E-layers are regions of high ion concentrations that reflect radio waves and are important in long-distance communication.
Composition of the atmosphere
Except for water vapor, whose atmospheric abundance varies from practically zero up to 4%, the fractions of the major atmospheric components N2, O2, and Ar are remarkably uniform below about 100 km. At greater heights, diffusion becomes the principal transport process, and the lighter gases become relatively more abundant. In addition, photochemical processes result in the formation of new species whose high reactivities would preclude their existence in significant concentrations at the higher pressures found at lower elevations.
The atmospheric gases fall into three abundance categories: major, minor, and trace. Nitrogen, the most abundant component, has accumulated over time as a result of its geochemical inertness; a very small fraction of it passes into the other phases as a result of biological activity and natural fixation by lightning. It is believed that denitrifying bacteria in marine sediments may provide the major route for the return of N2 to the atmosphere. Oxygen is almost entirely of biological origin, and cycles through the hydrosphere, the biosphere, and sedimentary rocks. Argon consists mainly of Ar40 which is a decay product of K40 in the mantle and crust.
| nitrogen | N2 | 78.08 % |
|---|---|---|
| oxygen | O2 | 20.95 % |
| argon | Ar | 0.93 % |
The most abundant of the minor gases aside from water vapor is carbon dioxide, about which more will be said below. Next in abundance are neon and helium. Helium is a decay product of radioactive elements in the earth, but neon and the other inert gases are primordial, and have probably been present in their present relative abundances since the earth’s formation. Two of the minor gases, ozone and carbon monoxide, have abundances that vary with time and location. A variable abundance implies an imbalance between the rates of formation and removal. In the case of carbon monoxide, whose major source is anthropogenic (a small amount is produced by biological action), the variance is probably due largely to localized differences in fuel consumption, particularly in internal combustion engines. The nature of the carbon monoxide sink (removal mechanism) is not entirely clear; it may be partly microbial.
| water | H2O | 0-4 % |
|---|---|---|
| carbon dioxide | CO2 | 325 ppm |
| neon | Ne | 18 ppm |
| helium | He | 5 ppm |
| methane | CH4 | 2 ppm |
| krypton | Kr | 1 ppm |
| hydrogen | H2 | 0.5 ppm |
| nitrous oxide | N2O | 0.3 ppm |
| carbon monoxide | CO | 0.05-0.2 ppm |
| ozone | O3 | 0.02 - 10 ppm |
| xenon | Xe | 0.08 ppm |
Table 3:Trace components
| ammonia | NH3 | 4 ppb |
| nitrogen oxide | NO | 1 ppb |
| sulfur dioxide | SO2 | 1 ppb |
| hydrogen sulfide | H2S | 0.05 ppb |
Ozone
Ozone is formed by the reaction of O2 with oxygen atoms produced photochemically. As a consequence the abundance of ozone varies with the time of day, the concentration of O atoms from other sources (photochemical smog, for example), and particularly with altitude; at 30 km, the ozone concentration reaches a maximum of 12 ppm.
Carbon dioxide
The concentration of atmospheric carbon dioxide, while fairly uniform globally, is increasing at a rate of 0.2-0.7% per year as a result of fossil fuel burning. The present CO2 content of the atmosphere is about 129  1018 g. Most of the CO2, however, is of natural origin, and represents the smallest part of the total carbonate reservoir that includes oceanic CO2, HCO3–, and carbonate sediments. The latter contain about 600 times as much CO2 as the atmosphere, and the oceans contain about 50 times as much. These relative amounts are controlled by the rates of the reactions that interconvert the various forms of carbonate.
1018 g. Most of the CO2, however, is of natural origin, and represents the smallest part of the total carbonate reservoir that includes oceanic CO2, HCO3–, and carbonate sediments. The latter contain about 600 times as much CO2 as the atmosphere, and the oceans contain about 50 times as much. These relative amounts are controlled by the rates of the reactions that interconvert the various forms of carbonate.
The surface conditions on the earth are sensitively dependent on the atmospheric CO2 concentration. This is due mainly to the strong infrared absorption of CO2, which promotes the absorption and trapping of solar heat (see below). Since CO2 acts as an acid in aqueous solution, the pH of the oceans is also dependent on the concentration of CO2 in the atmosphere; it has been estimated that if only 1% of the carbonate presently in sediments were still in the atmosphere, the pH of the oceans would be 5.9, instead of the present 8.2.
Energy balance of the atmosphere and earth
The amount of energy (the solar flux) impinging on the outer part of the atmosphere is 1367 watts m–2 . About 30% of this is reflected or scattered back into space by clouds, dust, and the atmospheric gas molecules themselves, and by the earth’s surface. About 19% of the radiation is absorbed by clouds or the atmosphere (mainly by and O3 , but not CO2), leaving 51% of the incident energy available for absorption by the earth’s surface. If one takes into account the uneven illumination of the earth’s surface and the small flux of internal heat to the surface, the assumption of thermal equilibrium requires that the earth emit about 240 watts m–2. This corresponds to the power that would be emitted by a black body at 255 K, or –18°C, which is the average temperature of the atmosphere at an altitude of 5 km. The observed mean global surface temperature of the earth is 13°C, and is presumably the temperature required to maintain thermal equilibrium between the earth and the atmosphere.
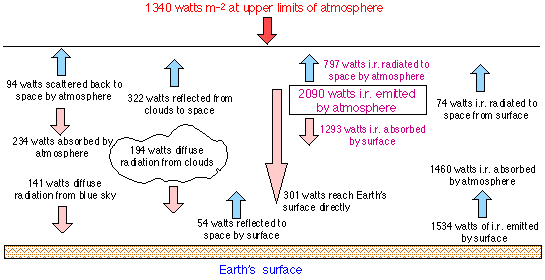
The greenhouse effect
The energy radiated by the earth has a longer wavelength (maximum 12m) than the incident radiation. Most gases absorb radiation in this range quite efficiently, including those gases such as CO2 and N2O that do not absorb the incident radiation. The energy absorbed by atmospheric gases is re-radiated in all directions; some of it therefore escapes into space, but a portion returns to the earth and is reabsorbed, thus raising its temperature.This is commonly called the greenhouse effect. If the amount of an infrared-absorbing gas such as carbon dioxide increases, a larger fraction of the incident solar radiation is trapped, and the mean temperature of the earth will increase.
Any significant increase in the temperature of the oceans would increase the atmospheric concentrations of both water and CO2, producing the possibility of a runaway process that would be catastrophic from a human perspective. Fossil fuel combustion and deforestation during the last two hundred years have increased the atmospheric CO2 concentration by 25%, and this increase is continuing. The same combustion processes responsible for the increasing atmospheric CO2 concentration also introduce considerable quantities of particulate materials into the upper atmosphere. The effect of these would be to scatter more of the incoming solar radiation, reducing the amount that reaches and heats the earth’s surface. The extent to which this process counteracts the greenhouse effect is still a matter of controversy; all that is known for sure is that the average temperature of the Earth is increasing.
Nitrous oxide
Carbon dioxide is not the only atmospheric gas of anthropogenic origin that can affect the heat balance of the earth; other examples are SO2 and N2O. Nitrous oxide is of particular interest, since its abundance is fairly high, and is increasing at a rate of about 0.5% per year. It is produced mainly by bacteria, and much of the increase is probably connected with introduction of increased nitrate into the environment through agricultural fertilization and sewage disposal. Besides being a strong infrared absorber, N2O is photochemically active, and can react with ozone. Any significant depletion of the ozone content of the upper atmosphere would permit more ultraviolet radiation to reach the earth. This would have numerous deleterious effects on present life forms, as well as contributing to a temperature increase. The warming effect attributed to anthropogenic additions of greenhouse gases to the atmosphere is estimated to be about 2 watts per m2 , or about 1.5% of the 150 watts per m2trapped by clouds and atmospheric gases. This is a relatively large perturbation compared to the maximum variation in solar output of 0.5 watts per m2 that has been observed during the past century. Continuation of greenhouse gas emission at present levels for another century could increase the atmospheric warming effect by 6-8 watts per m2.
A less-appreciated side effect of the increase in atmospheric carbon dioxide (and of other plant nutrients such as nitrates) may be reduction in plant species diversity by selectively encouraging the growth of species which are ordinarly held in check by other species that are able to grow well with fewer nutrients. This effect, for which there is already some evidence, could be especially pronounced when the competing species utilize the C3 and C4 photosynthetic pathways that differ in their sensitivity to CO2.
Contributors and Attributions
Stephen Lower, Professor Emeritus (Simon Fraser U.) Chem1 Virtual Textbook


