5.4: Hydration of Alkenes
- Page ID
- 205341
This page looks at the production of alcohols by the direct hydration of alkenes - adding water directly to the carbon-carbon double bond.
Manufacturing ethanol
Ethanol is manufactured by reacting ethene with steam. The reaction is reversible.
![]()
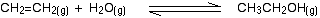
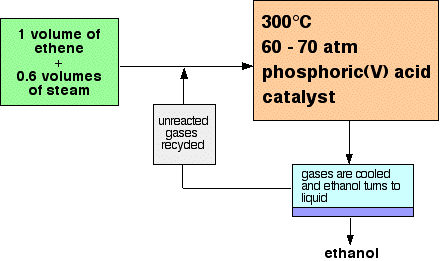
Only 5% of the ethene is converted into ethanol at each pass through the reactor. By removing the ethanol from the equilibrium mixture and recycling the ethene, it is possible to achieve an overall 95% conversion. A flow scheme for the reaction looks like this:
Manufacturing other alcohols
If you start from an unsymmetrical alkene like propene, you have to be careful to think about which way around the water adds across the carbon-carbon double bond. Markovnikov's Rule says that when you add a molecule HX across a carbon-carbon double bond, the hydrogen joins to the carbon atom which already has the more hydrogen atoms attached to it.
Thinking of water as H-OH, the hydrogen will add to the carbon with the more hydrogens already attached. That means that in the propene case, you will get propan-2-ol rather than propan-1-ol.
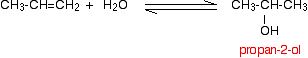
The conditions used during manufacture vary from alcohol to alcohol.
Contributors
Jim Clark (Chemguide.co.uk)


