13.8 Examples of Aromatic Compounds
- Page ID
- 28344
Aromatic Compound with a single ring
Aromatic Compounds with more than one ring
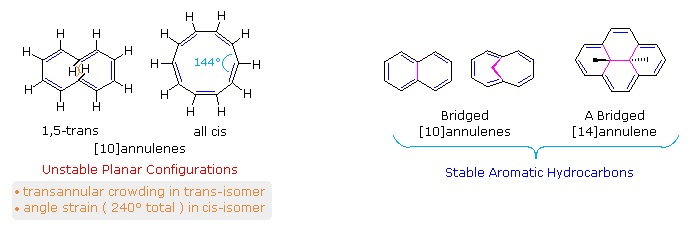
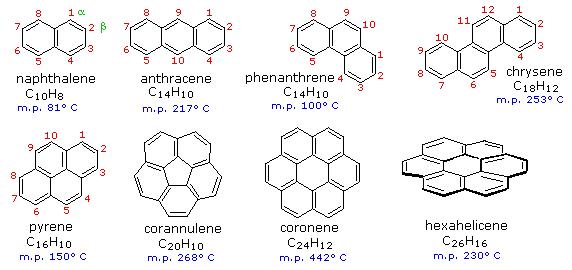
Benzene rings may be joined together (fused) to give larger polycyclic aromatic compounds. A few examples are drawn below, together with the approved numbering scheme for substituted derivatives. The peripheral carbon atoms (numbered in all but the last three examples) are all bonded to hydrogen atoms. Unlike benzene, all the C-C bond lengths in these fused ring aromatics are not the same, and there is some localization of the pi-electrons.
The six benzene rings in coronene are fused in a planar ring; whereas the six rings in hexahelicene are not joined in a larger ring, but assume a helical turn, due to the crowding together of the terminal ring atoms. This helical configuration renders the hexahelicene molecule chiral, and it has been resolved into stable enantiomers.
Aromatic Heterocycles
Many unsaturated cyclic compounds have exceptional properties that we now consider characteristic of "aromatic" systems. The following cases are illustrative:
| Compound | Structural | Reaction | Thermodynamic | |||
| 1,3-Cyclopentadiene | | Addition ( 0 ºC ) | Slight | |||
| 1,3,5-Cycloheptatriene | Addition ( 0 ºC ) | Slight | ||||
| 1,3,5,7-Cyclooctatetraene | Addition ( 0 ºC ) | Slight | ||||
| Benzene | Substitution | Large | ||||
| Pyridine | Substitution | Large | ||||
| Furan | Substitution ( 0 ºC ) | Moderate | ||||
| Pyrrole | Substitution | Moderate | ||||
Benzene is the archetypical aromatic compound. It is planar, bond angles=120º, all carbon atoms in the ring are sp2 hybridized, and the pi-orbitals are occupied by 6 electrons. The aromatic heterocycle pyridine is similar to benzene, and is often used as a weak base for scavenging protons. Furan and pyrrole have heterocyclic five-membered rings, in which the heteroatom has at least one pair of non-bonding valence shell electrons. By hybridizing this heteroatom to a sp2 state, a p-orbital occupied by a pair of electrons and oriented parallel to the carbon p-orbitals is created. The resulting planar ring meets the first requirement for aromaticity, and the π-system is occupied by 6 electrons, 4 from the two double bonds and 2 from the heteroatom, thus satisfying the Hückel Rule.
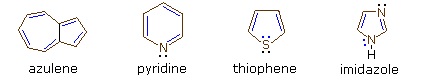
Four illustrative examples of aromatic compounds are shown above. The sp2 hybridized ring atoms are connected by brown bonds, the π-electron pairs and bonds that constitute the aromatic ring are colored blue. Electron pairs that are not part of the aromatic π-electron system are black. The first example is azulene, a blue-colored 10 π-electron aromatic hydrocarbon isomeric with naphthalene. The second and third compounds are heterocycles having aromatic properties. Pyridine has a benzene-like six-membered ring incorporating one nitrogen atom. The non-bonding electron pair on the nitrogen is not part of the aromatic π-electron sextet, and may bond to a proton or other electrophile without disrupting the aromatic system. In the case of thiophene, a sulfur analog of furan, one of the sulfur electron pairs (colored blue) participates in the aromatic ring π-electron conjugation. The last compound is imidazole, a heterocycle having two nitrogen atoms. Note that only one of the nitrogen non-bonding electron pairs is used for the aromatic π-electron sextet. The other electron pair (colored black) behaves similarly to the electron pair in pyridine.
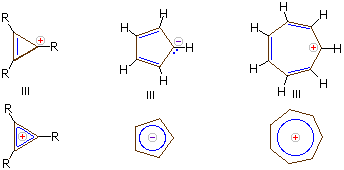
Charged Aromatic Compounds
Carbanions and carbocations may also show aromatic stabilization. Some examples are:
The three-membered ring cation has 2 π-electrons and is surprisingly stable, considering its ring strain. Cyclopentadiene is as acidic as ethanol, reflecting the stability of its 6 π-electron conjugate base. Salts of cycloheptatrienyl cation (tropylium ion) are stable in water solution, again reflecting the stability of this 6 π-electron cation.
Contributors
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry