9.6: Preparation of Alcohols, Ethers, and Epoxides
- Page ID
- 28200
Alcohols are prepared by SN2 reaction
Alkyl halides can be converted to alcohols by using SN2 reactions with OH- as a nucleophile. Substrates that undergo substitution by SN1 reaction can be converted to alcohols using water as the nucleophile (and it can even be the solvent). Recall that SN1 reactions are promoted in polar, protic solvents.
Ethers are prepared by SN2 reaction
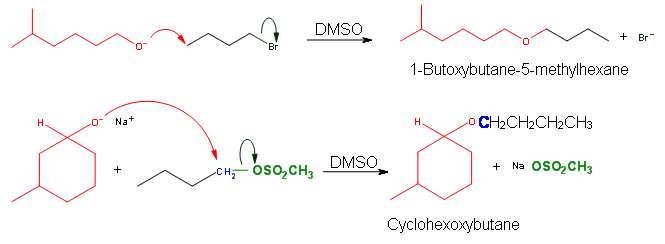
Williamson Ether Reactions involve an alkoxide that reacts with a primary haloalkane or a sulfonate ester. Alkoxides consist of the conjugate base of an alcohol and are comprised of an R group bonded to an oxygen atom. They are written as RO–, where R is the organic substituent.

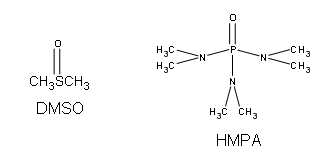
Ethers can be synthesized in standard SN2 conditions by coupling an alkoxide with a haloalkane/sulfonate ester. The alcohol that supplies the electron rich alkoxide can be used as the solvent, as well as dimethyl sulfoxide (DMSO) or hexamethylphosphoric triamide (HMPA).
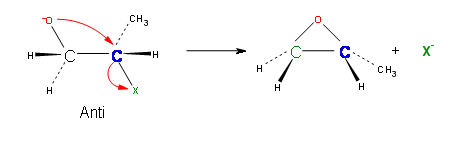
Epoxides are prepared by an SN2
Sn2 reactions are characterized by the inversion of stereochemistry at the site of the leaving group. Williamson Ether synthesis is not an exception to this rule and the reaction is set in motion by the backside attack of the nucleophile. This requires that the nucleophile and the electrophile are in anti-configuration.