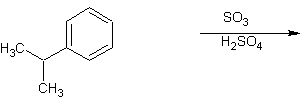18.9 Orientation Effects in Substituted Benzenes
- Page ID
- 28356
Substituted rings are divided into two groups based on the type of the substituent that the ring carries:
- Activated rings: the substituents on the ring are groups that donate electrons.
- Deactivated rings: the substituents on the ring are groups that withdraw electrons.
Introduction
Examples of activating groups in the relative order from the most activating group to the least activating:
-NH2, -NR2 > -OH, -OR> -NHCOR> -CH3 and other alkyl groups
with R as alkyl groups (CnH2n+1)
Examples of deactivating groups in the relative order from the most deactivating to the least deactivating:
-NO2, -CF3> -COR, -CN, -CO2R, -SO3H > Halogens
with R as alkyl groups (CnH2n+1)
The order of reactivity among Halogens from the more reactive (least deactivating substituent) to the least reactive (most deactivating substituent) halogen is:
F> Cl > Br > I
The order of reactivity of the benzene rings toward the electrophilic substitution when it is substituted with a halogen groups, follows the order of electronegativity. The ring that is substituted with the most electronegative halogen is the most reactive ring ( less deactivating substituent ) and the ring that is substituted with the least electronegatvie halogen is the least reactive ring ( more deactivating substituent ), when we compare rings with halogen substituents. Also the size of the halogen effects the reactivity of the benzene ring that the halogen is attached to. As the size of the halogen increase, the reactivity of the ring decreases.
The direction of the reaction
The activating group directs the reaction to the ortho or para position, which means the electrophile substitute the hydrogen that is on carbon 2 or carbon 4. The deactivating group directs the reaction to the meta position, which means the electrophile substitute the hydrogen that is on carbon 3 with the exception of the halogens that is a deactivating group but directs the ortho or para substitution.
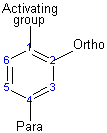
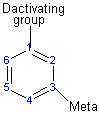
Substituents determine the reaction direction by resonance or inductive effect
Resonance effect is the conjugation between the ring and the substituent, which means the delocalizing of the \(\pi\) electrons between the ring and the substituent. Inductive effect is the withdraw of the sigma ( the single bond ) electrons away from the ring toward the substituent, due to the higher electronegativity of the substituent compared to the carbon of the ring.
Activating groups (ortho or para directors)
When the substituents like -OH have an unshared pair of electrons, the resonance effect is stronger than the inductive effect which make these substituents stronger activators, since this resonance effect direct the electron toward the ring. In cases where the subtituents is esters or amides, they are less activating because they form resonance structure that pull the electron density away from the ring.
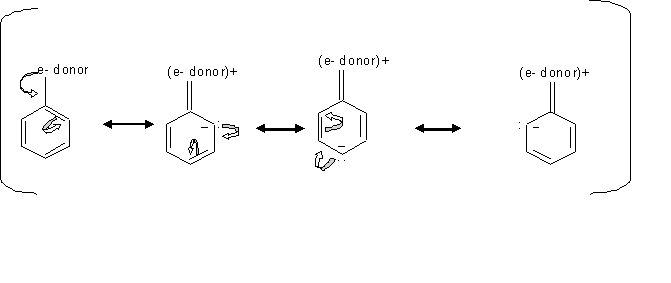
By looking at the mechanism above, we can see how groups donating electron direct the ortho, para electrophilic substition. Since the electrons locatinn transfer between the ortho and para carbons, then the electrophile prefer attacking the carbon that has the free electron.
Inductive effect of alkyl groups activates the direction of the ortho or para substitution, which is when s electrons gets pushed toward the ring.
Deactivating group (meta directors)
The deactivating groups deactivate the ring by the inductive effect in the presence of an electronegative atom that withdraws the electrons away from the ring.
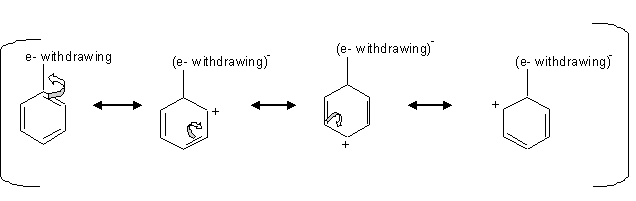
we can see from the mechanism above that when there is an electron withdraw from the ring, that leaves the carbons at the ortho, para positions with a positive charge which is unfavorable for the electrophile, so the electrophile attacks the carbon at the meta positions.
Halogens are an exception of the deactivating group that directs the ortho or para substitution. The halogens deactivate the ring by inductive effect not by the resonance even though they have an unpaired pair of electrons. The unpaired pair of electrons gets donated to the ring, but the inductive effect pulls away the s electrons from the ring by the electronegativity of the halogens.
Substituents determine the reactivity of rings
The reaction of a substituted ring with an activating group is faster than benzene. On the other hand, a substituted ring with a deactivated group is slower than benzene.
Activating groups speed up the reaction because of the resonance effect. The presence of the unpaired electrons that can be donated to the ring, stabilize the carbocation in the transition state. Thus; stabilizing the intermediate step, speeds up the reaction; and this is due to the decrease of the activating energy. On the other hand, the deactivating groups, withdraw the electrons away from the carbocation formed in the intermediate step, thus; the activation energy is increased which slows down the reaction.
The CH3Group is and ortho, para Director
Alkyl groups are Inductive activators
With o/p attack the form a tertiary arenium carbocation which speeds up the reaction
The O-CH3 Group is an ortho, para Director
Ortho and Para producst produces a resonance structure which stabilizes the arenium ion. This causes the ortho and para products for form faster than meta. Generally, the para product is preferred because of steric effects.
Acyl groups are meta Directors
Acyl groups are resonance deactivators. Ortho and para attack produces a resonance structure which places the arenium cation next to and additional cation. This destabilizes the arenium cation and slows down ortho and para reaction. By default the meta product forms faster because it lacks this destablizing resonance structure.
References
- Schore, N.E. and P.C. Vollhardt. 2007. Organic Chemistry, structure and function, 5th ed. New York,NY: W.H. Freeman and Company.
- Fryhle, C.B. and G. Solomons. 2008. Organic Chemistry, 9th ed.Danvers,MA: Wiley.
Outside Links
Problems
- Predict the direction of the electrophile substition on these rings:
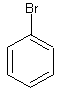
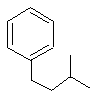
2. Which nitration product is going to form faster?
nitration of aniline or nitration of nitrobenzene?
3. Predict the product of the following two sulfonation reactions:
A.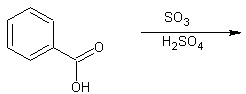
4. Classify these two groups as activating or deactivating groups:
A. alcohol
B. ester
5. By which effect does trichloride effect a monosubstituted ring?
Answers
1. The first substitution is going to be ortho and/or para substitution since we have a halogen subtituent. The second substition is going to be ortho and/or para substitution also since we have an alkyl substituent.
2. The nitration of aniline is going to be faster than the nitration of nitrobenzene, since the aniline is a ring with NH2 substituent and nitrobenzene is a ring with NO2 substiuent. As described above NH2is an activating group which speeds up the reaction and NO2 is deactivating group that slows down the reaction.
3. A. the product is 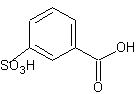
B. the product is 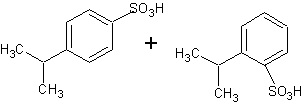
4. A. alcohol is an activating group.
B. ester is a deactivating group.
5. Trichloride deactivate a monosubstitued ring by inductive effect.
Contributors
- Lana Alawwad (UCD)