16.10 The Diels–Alder Reaction
- Page ID
- 28333
After completing this section, you should be able to
- write an equation to represent a typical Diels-Alder reaction.
- draw the structure of the product formed when a given conjugated diene reacts with a given dienophile in a Diels-Alder reaction.
- identify the diene and dienophile that must be used to prepare a given compound by a Diels-Alder reaction.
- explain the general mechanism of the Diels-Alder reaction, without necessarily being able to describe it in detail.
Make certain that you can define, and use in context, the key terms below.
- Diels-Alder cycloaddition
- pericyclic reaction
The Diels-Alder reaction is an example of an organic chemical reaction which does not proceed by either a polar or a free radical pathway, but rather a pericyclic reaction.
Although we do not expect you to be able to provide a detailed account of the mechanism of this reaction, you should learn enough about the Diels-Alder reaction to fulfil the objectives stated above. You will find it useful to contrast the mechanism of the Diels-Alder reaction with the polar and radical mechanisms studied earlier.
The unique character of conjugated dienes manifests itself dramatically in the Diels-Alder Cycloaddition Reaction. The Diels-Alder reaction is an important and widely used synthetic method for making six-membered rings. In the Diels-Alder reaction, a conjugated diene, simply referred to as the diene, reacts with a double or triple bond co-reactant called the dienophile, because it combines with (has an affinity for) the diene. During the reaction, two pi-bonds are converted to two sigma-bonds. The Diels-Alder cycloaddition is classified as a pericyclic process. Pericyclic reactions involve the redistribution of bonding electrons in a single step mechanism and will be discussed in greater detail in Chapter 30. In particular, the Diels-alder reaction is called a [4+2] process because the diene has four pi-electrons that shift position in the reaction and the dienophile has two.
General Reaction
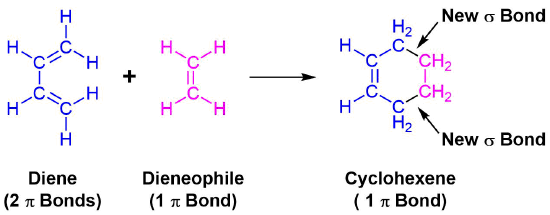
An example of the Diels-Alder reaction is the cycloaddition of 1,3-butadiene to cyanoethene (acrylonitrile) to form 4-cyanocyclohexene.
Example \(\PageIndex{1}\)
Mechanism
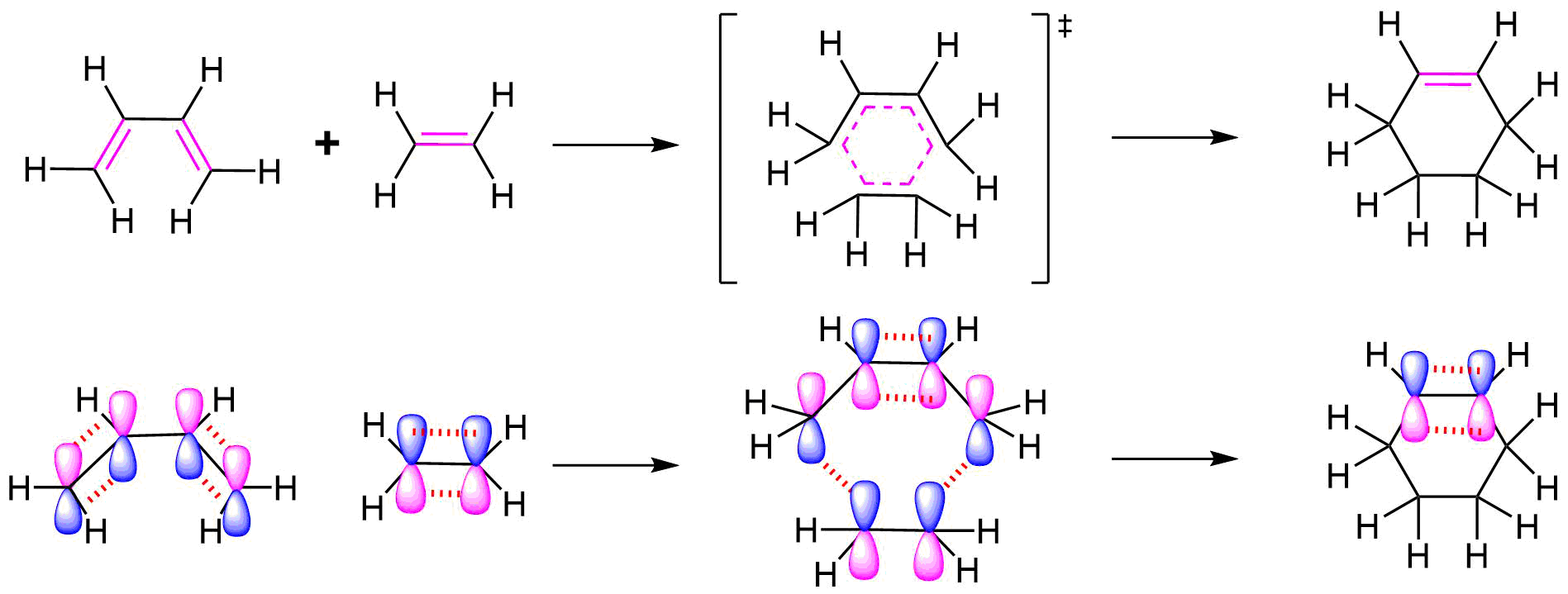
All of the electron rearrangements of the Diels-Alder reaction take place once in a single mechanistic step. During this step carbons 1 and 4 of the diene and both alkene carbons of the dienophile, rehybridize from sp2 to sp3 and electrons rearrange to create two new sigma bonds in the cyclic product. Carbons 2 and 3 of the diene remain sp2 hybridized and form a new pi bond in the product.
The mechanism occurs through a cyclic transition state in which there is head-on overlap of two p orbitals on carbons 1 and 4 of the diene with the two p orbitals from the alkene of the dienophile to form two new sigma bonds in the cyclohexene product. The remaining two p orbitals from the diene overlap to form the new pi bond.
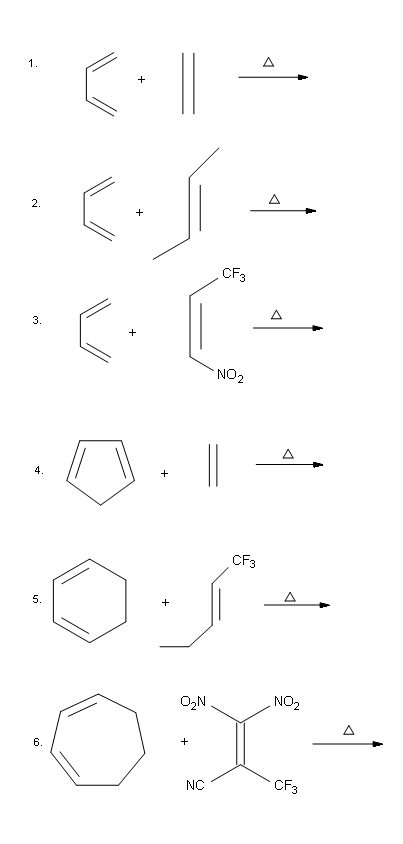
Problems
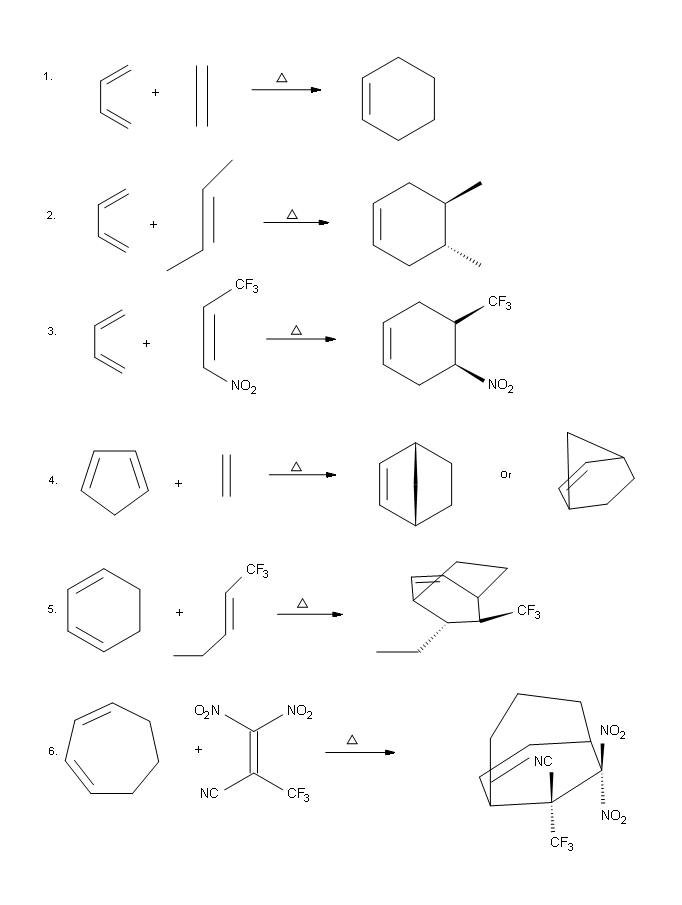
Answers
.png?revision=1&size=bestfit&width=590&height=782)
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
Organic Chemistry With a Biological Emphasis by Tim Soderberg (University of Minnesota, Morris)
- Amar Patel (UCD)
- Layne A. Morsch (University of Illinois Springfield)


