11.1: Introduction to Alkynes
- Page ID
- 28230
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\)
The simplest alkyne—a hydrocarbon with carbon-to-carbon triple bond—has the molecular formula C2H2 and is known by its common name—acetylene. Its structure is H–C≡C–H.
Terminal Alkyne: Internal Alkyne:
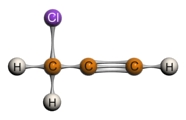
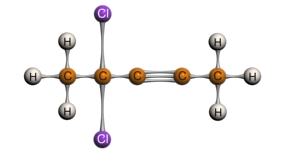
3-chloro-1-propyne 4,4-dichloro-2-pentyne
Bonding and Hybridization
| Bond | Name | Location | Overlap |
| Bond 1 | s (? bond) bond | Formed between 2 sp orbitals of carbon and hydrogen atoms | End-on overlap |
| Bond 2 | S (? bond) bond | Formed between the 2 sp orbital of 2 unsaturated Carbon atoms. | End-on overlap |
| Bond 3 | p-bonds (? bonds) | Formed between the 2 p-orbitals among the carbon atoms | Side-on overlap |

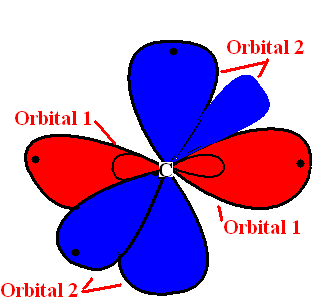
| Orbital | Name | Location |
| Orbital 1 | sp hybrid orbitals | Formed in the linear structure model of carbon atom |
| Orbital 2 | p-orbitals | Formed on each carbon |
Contributors
Jim Clark (Chemguide.co.uk)


