Solutions 20
- Page ID
- 47406
S20.1
\(F_2^+\)
For the neutral \(F_2\) molecule, the electron configuration (ignoring the \(\sigma_{1s}\) bonding and antibonding orbitals) is
\[ (\sigma_{2s})^2 (\sigma^*_{2s})^2 (\sigma_{2p_z})^2 (\pi_x)^2 (\pi_y)^2 (\pi^*_x)^2 (\pi^*_y)^2 \]
giving a bond order of 1. To form the \(F_2^+\) ion, the highest energy electron is removed, which is from either the \( \pi^*_x \) or \( \pi^*_y\) orbital (it does not matter). This generates bond order of 1.5. To form the \(F_2^-\) ion, an electron is added to the \(\sigma^*_{2p_z}\) antibonding orbital, giving a bond order of 0.5 also. \(F_2^+\) has the shortest bond since it is the strongest.
S20.2
4O2- + H2 ---> O2 + 3H2O2
S20.3
S20.4
Pyrene has a larger conjugated system with two more rings than naphthalene, so its \(\pi\) electrons are more delocalized.
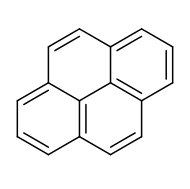
Pyrene (left) vs. naphthalene (right).
S20.5
"Law of conservation of orbitals" means that the number of hybrid orbitals formed must equal the number of orbitals that are used in the formation of the hybrid orbitals.
S20.6
Bond order=(# of bonding electrons - # of antibonding electrons)/2
Therefore, if bond order is 2, # of bonding electrons is 6, we get # of antibonding electrons to be 2.
The lewis structure will be a double bond between the two atoms with two more electrons on either of the two atoms.
One electron reduction means gaining one more electron, since the bonding orbitals have already had 4 electrons, the extra one will probably go to the antibonding orbital and lead to 3/2 bond order:
Bond order=[6-(2+1)]/2=3/2.

