Comprehensive HW 5 Key
- Page ID
- 207147
Q1
This questions takes a lot of steps. Start with an isothermal expansion to get the pressure down to 1 atm.
\[ \begin{align} Q_{iso} &= nRT \ln \left(\dfrac{P_i}{P_f}\right) \\[5pt] &= 0.56 \times 8.314 \times 573 \times \ln \left(\dfrac{21.9}{1}\right) \\[5pt] &= 8.234 \,kJ \end{align}\]
Check should have to input heat to keep the temperature up while the system is doing work expanding.
Then cool to 100 C
\[Q_{vap} = mC \Delta T = 0.56 \times 18 \times 1.996 \times -100 = -2.011 kJ \]
Then condense
\[Q_{condense} = m \Delta H_v = 10.08 \times 2257 = -22.75 kJ \]
Then cool to 0 C
\[Q_{liq} = mC \Delta T = 10.08 \times 4.1 \times -100 = -4.132 kJ \]
Then freeze
\[Q_{freeze} = m \Delta H_f = 10.08 \times 333.55 = -3.36 kJ \]
Then cool the ice to -6
\[Q_{ice} = mC \Delta T = 10.08 \times 2.108 \times -6 = -0.12 kJ \]
Add them all up and you have:
\[Q_{total} = -2.011 + -22.75 + -4.132 + -3.36 + -0.12 = -32.373 kJ \]
Is released by the water.
Q2
Use the Clausius-Clapeyron Eq.
\[\ln \left( \dfrac{P_1}{P_2} \right) = \dfrac{\Delta H_{vap}}{R} \left( \dfrac{1}{T_2}- \dfrac{1}{T_1} \right) \]
Solve for \(\Delta H_{vap}\)
\[\ln \left( \dfrac{0.85}{0.897} \right) = \dfrac{\Delta H_{vap}}{R} \left( \dfrac{1}{298}- \dfrac{1}{293} \right) \]
\[\ln \left( \dfrac{0.85}{0.897} \right) = \dfrac{\Delta H_{vap}}{R} \left( \dfrac{1}{298}- \dfrac{1}{293} \right) \]
\[ -0.0538 = \dfrac{\Delta H_{vap}}{R} (-5.726 \times 10^{-5}) \]
\[\Delta H_{vap} = -0.0538 \div -5.726 \times 10^{-5} \times 8.314 = 7.811 kJ \]
Clausius-Clapeyron Eq number 2
\[\ln \left( \dfrac{1}{0.850} \right) = \dfrac{7811}{8.314} \left( \dfrac{1}{293}- \dfrac{1}{T_1} \right) \]
\[0.162 = 939 \times (0.0034 - \dfrac{1}{T_1}) \]
\[T_1 = 309 K \]
Q3
a. More Clausius-Clapeyron Eq Yay!
\[\ln \left( \dfrac{P_1}{P_2} \right) = \dfrac{\Delta H_{vap}}{R} \left( \dfrac{1}{T_2}- \dfrac{1}{T_1} \right) \]
\[\ln \left( \dfrac{33.4}{100} \right) = \dfrac{\Delta H_{vap}}{R} \left( \dfrac{1}{225}- \dfrac{1}{210} \right) \]
\[-1.096 = \dfrac{\Delta H_{vap}}{R} (-3.17\times 10^{-4}) \]
\[\Delta H_{vap} = 28.7 kJ/molK \]
b. Clausius-Clapeyron Eq number 4 I think
\[\ln \left( \dfrac{?}{100} \right) = \dfrac{28700}{8.314} \left( \dfrac{1}{225}- \dfrac{1}{249} \right) \]
\[\ln \left( \dfrac{?}{100} \right) = 1.47 \]
\[P_1 = 438 torr \]
c. 5 times.............
\[\ln \left( \dfrac{200}{100} \right) = \dfrac{28700}{8.314} \left( \dfrac{1}{225}- \dfrac{1}{T_1} \right) \]
\[2.007\times 10^{-4} = (0.0044 - \dfrac{1}{T_1}\]
\[T_1 = 238 K = -35 C \]
Q4
The atoms gain enough kinetic energy to overcome the attractive forces and spread out.
Q5
Ethylene glycol has two locations that can hydrogne bond so it has a much bigger intermolecular forces.
Q6
a. The surface tension decreases as a function of temperture because the molecules get more kinetic energy and the relative magnitude of the intermolecular forces decreases.
b. Same reason.
Q7
Use the Ideal Gas Law
\[PV = nRT \]
a. 0.974 mols
b. 0.101
Always had to be smaller because we have increased the temperature and decreased the pressure.
Q8
a. The air pressure is lower at altitude so the boiling point temperature is lower and the egg cooks slower.
b. Sweating cools the body because fo the water to evaporate it receives energy from the surroundings lowering the temperature.
Q9
X. Metallic
Y. Molecular
Z. Ionic
Q10
Numbers are slightly off. Pay attention to the slope of the solid-liquid interface
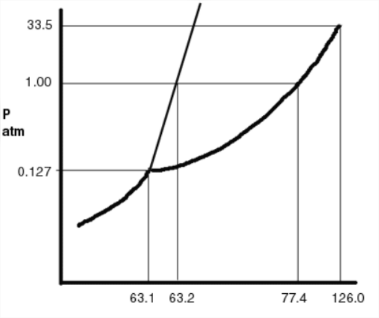
Q11
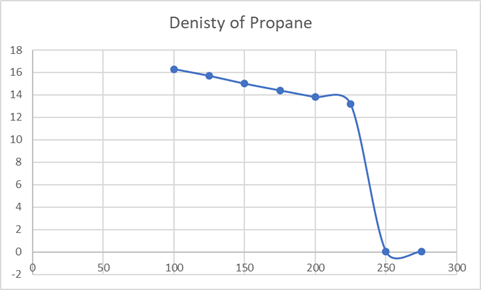
Boils at 250 K
Q12
a. See the first question
b.
\[Q_total = mC\Delta T +m\Delta H_{fus} + mC\Delta T + m\Delta H_{vap} + mC\Delta T \]
\[Q_total = 1 \times 2.1 \times 10 +1 \times 333 + 1 \times 4.1 \times 100 + 1 \times 2257 + 1 \times 1.9 \times 20 = 3.059 kJ \]
c.
Need enough energy to melt the ice and bring up the temperature.
\[Q =mC\Delta T +m\Delta H_{fus} + mC\Delta T = 25 \times 2.1 \times 3 + 25 \times 333 + 25 \times 4.1 \times 45 = 13095 J\]
\[Q_{boil} = mC\Delta T = m \times 4.1 \times 55 = 13095 \]
\[m_{boil} = 57.51 \]
d.
Need to cool by
\[Q = 150 \times 4.1 \times 7 = 4305 \]
\[Q_{ice} = m ( 2.1 \times 5 + 333 + 4.1 \times 15) = m \times 405 \]
\[m_{ice} = 4305/405 = 10.6 g \]
Q13
The normal boiling point is the temperature at which the vapor pressure equals the atm.
Q14
Volatile liquids have large vapor pressures. If they have the same mass that doesn't mean the atoms or the arrangement of the atoms in the molecules are the same. Volatile liquids have much lower intermolecular forces than nonvolatile liquids.
Q15
a. Gas to Liquid to Solid
b. Phase transitions are movements inside the same state.
So all the transistions between the different types (allotropes) of ice or solid sulfur.

