7.8: Standard Enthalpies of Formation
- Page ID
- 41250
- To understand Enthalpies of Formation and be able to use them to calculate Enthalpies of Reaction
One way to report the heat absorbed or released by chemical reactions would be to compile a massive set of reference tables that list the enthalpy changes for all possible chemical reactions, which would require an incredible amount of effort. Fortunately, Hess’s law allows us to calculate the enthalpy change for virtually any conceivable chemical reaction using a relatively small set of tabulated data, starting from the elemental forms of each atom at 25 oC and 1 atm pressure.
Enthalpy of formation (\(ΔH_f\)) is the enthalpy change for the formation of 1 mol of a compound from its component elements, such as the formation of carbon dioxide from carbon and oxygen. The formation of any chemical can be as a reaction from the corresponding elements:
\[ \text{elements} \rightarrow \text{compound} \nonumber\]
which in terms of the the Enthalpy of formation becomes
\[\Delta H_{rxn} = \Delta H_{f} \label{7.8.1} \]
For example, consider the combustion of carbon:
\[ \ce{ C(s) + O2 (g) -> CO2 (g)} \nonumber\]
then
\[ \Delta H_{rxn} = \Delta H_{f}\left [CO_{2}\left ( g \right ) \right ] \nonumber \]
The sign convention for ΔHf is the same as for any enthalpy change: \(ΔH_f < 0\) if heat is released when elements combine to form a compound and \(ΔH_f > 0\) if heat is absorbed.
The sign convention is the same for all enthalpy changes: negative if heat is released by the system and positive if heat is absorbed by the system.
Standard Enthalpies of Formation
The magnitude of ΔH for a reaction depends on the physical states of the reactants and the products (gas, liquid, solid, or solution), the pressure of any gases present, and the temperature at which the reaction is carried out. To avoid confusion caused by differences in reaction conditions and ensure uniformity of data, the scientific community has selected a specific set of conditions under which enthalpy changes are measured. These standard conditions serve as a reference point for measuring differences in enthalpy, much as sea level is the reference point for measuring the height of a mountain or for reporting the altitude of an airplane.
The standard conditions for which most thermochemical data are tabulated are a pressure of 1 atmosphere (atm) for all gases and a concentration of 1 M for all species in solution (1 mol/L). In addition, each pure substance must be in its standard state, which is usually its most stable form at a pressure of 1 atm at a specified temperature. We assume a temperature of 25°C (298 K) for all enthalpy changes given in this text, unless otherwise indicated. Enthalpies of formation measured under these conditions are called standard enthalpies of formation (\(ΔH^o_f\)) The enthalpy change for the formation of 1 mol of a compound from its component elements when the component elements are each in their standard states. The standard enthalpy of formation of any element in its most stable form is zero by definition.
The standard enthalpy of formation of any element in its standard state is zero by definition.
For example, although oxygen can exist as ozone (O3), atomic oxygen (O), and molecular oxygen (O2), O2 is the most stable form at 1 atm pressure and 25°C. Similarly, hydrogen is H2(g), not atomic hydrogen (H). Graphite and diamond are both forms of elemental carbon, but because graphite is more stable at 1 atm pressure and 25°C, the standard state of carbon is graphite (Figure \(\PageIndex{1}\)). Therefore, \(\ce{O2(g)}\), \(\ce{H2(g)}\), and graphite have \(ΔH^o_f\) values of zero.

The standard enthalpy of formation of glucose from the elements at 25°C is the enthalpy change for the following reaction:
\[ 6C\left (s, graphite \right ) + 6H_{2}\left (g \right ) + 3O_{2}\left (g \right ) \rightarrow C_{6}H_{12}O_{6}\left (s \right )\; \; \; \Delta H_{f}^{o} = - 1273.3 \; kJ \label{7.8.2} \]
It is not possible to measure the value of \(ΔH^oo_f \) for glucose, −1273.3 kJ/mol, by simply mixing appropriate amounts of graphite, \(\ce{O2}\), and \(\ce{H2}\) and measuring the heat evolved as glucose is formed since the reaction shown in Equation \(\ref{7.8.2}\) does not occur at a measurable rate under any known conditions. Glucose is not unique; most compounds cannot be prepared by the chemical equations that define their standard enthalpies of formation. Instead, values of \(ΔH^oo_f \) are obtained using Hess’s law and standard enthalpy changes that have been measured for other reactions, such as combustion reactions. Values of \(ΔH^o_f\) for an extensive list of compounds are given in Table T1. Note that \(ΔH^o_f\) values are always reported in kilojoules per mole of the substance of interest. Also notice in Table T1 that the standard enthalpy of formation of O2(g) is zero because it is the most stable form of oxygen in its standard state.
For the formation of each compound, write a balanced chemical equation corresponding to the standard enthalpy of formation of each compound.
- \(\ce{HCl(g)}\)
- \(\ce{MgCO3(s)}\)
- \(\ce{CH3(CH2)14CO2H(s)}\) (palmitic acid)
- Given:
-
compound formula and phase.
- Asked for:
-
balanced chemical equation for its formation from elements in standard states
- Strategy:
-
Use Table T1 to identify the standard state for each element. Write a chemical equation that describes the formation of the compound from the elements in their standard states and then balance it so that 1 mol of product is made.
- Solution:
-
To calculate the standard enthalpy of formation of a compound, we must start with the elements in their standard states. The standard state of an element can be identified in Table T1: by a \(ΔH^o_f\) value of 0 kJ/mol.
Hydrogen chloride contains one atom of hydrogen and one atom of chlorine. Because the standard states of elemental hydrogen and elemental chlorine are \(\ce{H2(g)}\) and \(\ce{Cl2(g)}\), respectively, the unbalanced chemical equation is
\[\ce{H2(g) + Cl2(g) \rightarrow HCl(g)} \nonumber\]
Fractional coefficients are required in this case because ΔHof values are reported for 1 mol of the product, \(\ce{HCl}\). Multiplying both \(\ce{H2(g)}\) and \(\ce{Cl2(g)}\) by 1/2 balances the equation:
\[ \ce{1/2 H_{2} (g) + 1/2 Cl_{2} (g) \rightarrow HCl (g)} \nonumber\]
The standard states of the elements in this compound are \(\ce{Mg(s)}\), \(\ce{C(s, graphite)}\), and \(\ce{O2(g)}\). The unbalanced chemical equation is thus
\[\ce{Mg(s) + C (s, graphite) + O2 (g) \rightarrow MgCO3 (s)} \nonumber\]
This equation can be balanced by inspection to give
\[ \ce{Mg (s) + C (s, graphite ) + 3/2 O2 (g)\rightarrow MgCO3 (s)} \nonumber\]
Palmitic acid, the major fat in meat and dairy products, contains hydrogen, carbon, and oxygen, so the unbalanced chemical equation for its formation from the elements in their standard states is as follows:
\[\ce{C(s, graphite) + H2(g) + O2(g) \rightarrow CH3(CH2)14CO2H(s)} \nonumber\]
There are 16 carbon atoms and 32 hydrogen atoms in 1 mol of palmitic acid, so the balanced chemical equation is
\[\ce{16C (s, graphite) + 16 H2(g) + O2(g) -> CH3(CH2)14CO2H(s) } \nonumber\]
For the formation of each compound, write a balanced chemical equation corresponding to the standard enthalpy of formation of each compound.
- \(\ce{NaCl(s)}\)
- \(\ce{H2SO4(l)}\)
- \(\ce{CH3CO2H(l)}\) (acetic acid)
- Answer a
-
\[ \ce{ Na (s) + 1/2 Cl2 (g) \rightarrow NaCl (s)} \nonumber \]
- Answer b
-
\[ \ce{H_{2} (g) + 1/8 S8 (s) + 2O2 ( g) \rightarrow H2 SO4( l) } \nonumber\]
- Answer c
-
\[\ce{2C(s) + O2(g) + 2H2(g) -> CH3CO2H(l)} \nonumber \]
Definition of Heat of Formation Reactions: https://youtu.be/A20k0CK4doI
Standard Enthalpies of Reaction
Tabulated values of standard enthalpies of formation can be used to calculate enthalpy changes for any reaction involving substances whose \(\Delta{H_f^o}\) values are known. The standard enthalpy of reaction \(\Delta{H_{rxn}^o}\) is the enthalpy change that occurs when a reaction is carried out with all reactants and products in their standard states. Consider the general reaction
\[ aA + bB \rightarrow cC + dD \label{7.8.3}\]
where \(A\), \(B\), \(C\), and \(D\) are chemical substances and \(a\), \(b\), \(c\), and \(d\) are their stoichiometric coefficients. The magnitude of \(ΔH^ο\) is the sum of the standard enthalpies of formation of the products, each multiplied by its appropriate coefficient, minus the sum of the standard enthalpies of formation of the reactants, also multiplied by their coefficients:
\[ \Delta H_{rxn}^{o} = \underbrace{ \left [c\Delta H_{f}^{o}\left ( C \right ) + d\Delta H_{f}^{o}\left ( D \right ) \right ] }_{\text{products} } - \underbrace{ \left [a\Delta H_{f}^{o}\left ( A \right ) + b\Delta H_{f}^{o}\left ( B \right ) \right ]}_{\text{reactants }} \label{7.8.4} \]
More generally, we can write
\[ \Delta H_{rxn}^{o} = \sum m\Delta H_{f}^{o}\left ( products \right ) - \sum n\Delta H_{f}^{o}\left ( reactants \right ) \label{7.8.5} \]
where the symbol \(\sum\) means “sum of” and \(m\) and \(n\) are the stoichiometric coefficients of each of the products and the reactants, respectively. “Products minus reactants” summations such as Equation \(\ref{7.8.5}\) arise from the fact that enthalpy is a state function. Because many other thermochemical quantities are also state functions, “products minus reactants” summations are very common in chemistry; we will encounter many others in subsequent chapters.
"Products minus reactants" summations are typical of state functions.
To demonstrate the use of tabulated ΔHο values, we will use them to calculate \(ΔH_{rxn}\) for the combustion of glucose, the reaction that provides energy for your brain:
\[ \ce{ C6H12O6 (s) + 6O2 (g) \rightarrow 6CO2 (g) + 6H2O (l)} \label{7.8.6} \]
Using Equation \(\ref{7.8.5}\), we write
\[ \Delta H_{f}^{o} =\left \{ 6\Delta H_{f}^{o}\left [ CO_{2}\left ( g \right ) \right ] + 6\Delta H_{f}^{o}\left [ H_{2}O\left ( g \right ) \right ] \right \} - \left \{ \Delta H_{f}^{o}\left [ C_{6}H_{12}O_{6}\left ( s \right ) \right ] + 6\Delta H_{f}^{o}\left [ O_{2}\left ( g \right ) \right ] \right \} \label{7.8.7} \]
From Table T1, the relevant ΔHοf values are ΔHοf [CO2(g)] = -393.5 kJ/mol, ΔHοf [H2O(l)] = -285.8 kJ/mol, and ΔHοf [C6H12O6(s)] = -1273.3 kJ/mol. Because O2(g) is a pure element in its standard state, ΔHοf [O2(g)] = 0 kJ/mol. Inserting these values into Equation \(\ref{7.8.7}\) and changing the subscript to indicate that this is a combustion reaction, we obtain
\[ \begin{align} \Delta H_{comb}^{o} &= \left [ 6\left ( -393.5 \; kJ/mol \right ) + 6 \left ( -285.8 \; kJ/mol \right ) \right ] - \left [-1273.3 + 6\left ( 0 \; kJ\;mol \right ) \right ] \label{7.8.8} \\[4pt] &= -2802.5 \; kJ/mol \end{align} \]
As illustrated in Figure \(\PageIndex{2}\), we can use Equation \(\ref{7.8.8}\) to calculate \(ΔH^ο_f\) for glucose because enthalpy is a state function. The figure shows two pathways from reactants (middle left) to products (bottom). The more direct pathway is the downward green arrow labeled \(ΔH^ο_{comb}\). The alternative hypothetical pathway consists of four separate reactions that convert the reactants to the elements in their standard states (upward purple arrow at left) and then convert the elements into the desired products (downward purple arrows at right). The reactions that convert the reactants to the elements are the reverse of the equations that define the \(ΔH^ο_f\) values of the reactants. Consequently, the enthalpy changes are
\[ \begin{align} \Delta H_{1}^{o} &= \Delta H_{f}^{o} \left [ glucose \left ( s \right ) \right ] \nonumber \\[4pt] &= -1 \; \cancel{mol \; glucose}\left ( \dfrac{1273.3 \; kJ}{1 \; \cancel{mol \; glucose}} \right ) \nonumber \\[4pt] &= +1273.3 \; kJ \nonumber \\[4pt] \Delta H_{2}^{o} &= 6 \Delta H_{f}^{o} \left [ O_{2} \left ( g \right ) \right ] \nonumber \\[4pt] & =6 \; \cancel{mol \; O_{2}}\left ( \dfrac{0 \; kJ}{1 \; \cancel{mol \; O_{2}}} \right ) \nonumber \\[4pt] &= 0 \; kJ \end{align} \label{7.8.9} \]
Recall that when we reverse a reaction, we must also reverse the sign of the accompanying enthalpy change (Equation \ref{7.8.4} since the products are now reactants and vice versa.
The overall enthalpy change for conversion of the reactants (1 mol of glucose and 6 mol of O2) to the elements is therefore +1273.3 kJ.
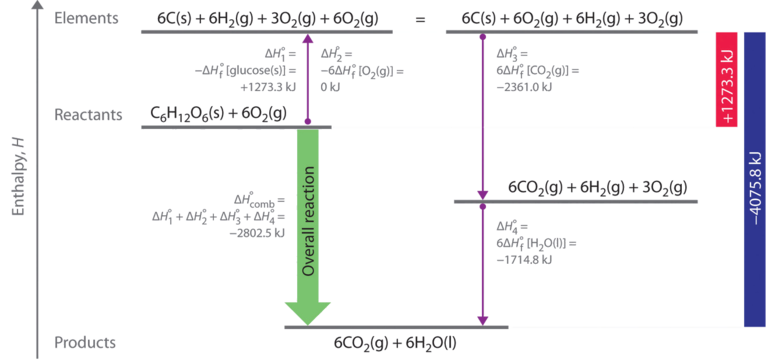
The reactions that convert the elements to final products (downward purple arrows in Figure \(\PageIndex{2}\)) are identical to those used to define the ΔHοf values of the products. Consequently, the enthalpy changes (from Table T1) are
\[ \begin{matrix} \Delta H_{3}^{o} = \Delta H_{f}^{o} \left [ CO_{2} \left ( g \right ) \right ] = 6 \; \cancel{mol \; CO_{2}}\left ( \dfrac{393.5 \; kJ}{1 \; \cancel{mol \; CO_{2}}} \right ) = -2361.0 \; kJ \\ \Delta H_{4}^{o} = 6 \Delta H_{f}^{o} \left [ H_{2}O \left ( l \right ) \right ] = 6 \; \cancel{mol \; H_{2}O}\left ( \dfrac{-285.8 \; kJ}{1 \; \cancel{mol \; H_{2}O}} \right ) = -1714.8 \; kJ \end{matrix} \]
The overall enthalpy change for the conversion of the elements to products (6 mol of carbon dioxide and 6 mol of liquid water) is therefore −4075.8 kJ. Because enthalpy is a state function, the difference in enthalpy between an initial state and a final state can be computed using any pathway that connects the two. Thus the enthalpy change for the combustion of glucose to carbon dioxide and water is the sum of the enthalpy changes for the conversion of glucose and oxygen to the elements (+1273.3 kJ) and for the conversion of the elements to carbon dioxide and water (−4075.8 kJ):
\[ \Delta H_{comb}^{o} = +1273.3 \; kJ +\left ( -4075.8 \; kJ \right ) = -2802.5 \; kJ \label{7.8.10} \]
This is the same result we obtained using the “products minus reactants” rule (Equation \(\ref{7.8.5}\)) and ΔHοf values. The two results must be the same because Equation \(\ref{7.8.10}\) is just a more compact way of describing the thermochemical cycle shown in Figure \(\PageIndex{1}\).
Long-chain fatty acids such as palmitic acid (\(\ce{CH3(CH2)14CO2H}\)) are one of the two major sources of energy in our diet (\(ΔH^o_f\) =−891.5 kJ/mol). Use the data in Table T1 to calculate ΔHοcomb for the combustion of palmitic acid. Based on the energy released in combustion per gram, which is the better fuel — glucose or palmitic acid?
Given: compound and \(ΔH^ο_{f}\) values
Asked for: \(ΔH^ο_{comb}\) per mole and per gram
Strategy:
- After writing the balanced chemical equation for the reaction, use Equation \(\ref{7.8.5}\) and the values from Table T1 to calculate \(ΔH^ο_{comb}\) the energy released by the combustion of 1 mol of palmitic acid.
- Divide this value by the molar mass of palmitic acid to find the energy released from the combustion of 1 g of palmitic acid. Compare this value with the value calculated in Equation \(\ref{7.8.8}\) for the combustion of glucose to determine which is the better fuel.
Solution:
A To determine the energy released by the combustion of palmitic acid, we need to calculate its \(ΔH^ο_f\). As always, the first requirement is a balanced chemical equation:
\[C_{16}H_{32}O_{2(s)} + 23O_{2(g)} \rightarrow 16CO_{2(g)} + 16H_2O_{(l)} \nonumber \]
Using Equation \(\ref{7.8.5}\) (“products minus reactants”) with ΔHοf values from Table T1 (and omitting the physical states of the reactants and products to save space) gives
\[ \begin{align*} \Delta H_{comb}^{o} &= \sum m \Delta H^o_f\left( {products} \right) - \sum n \Delta H^o_f \left( {reactants} \right) \\[4pt] &= \left [ 16\left ( -393.5 \; kJ/mol \; CO_{2} \right ) + 16\left ( -285.8 \; kJ/mol \; H_{2}O \; \right ) \right ] \\[4pt] & - \left [ -891.5 \; kJ/mol \; C_{16}H_{32}O_{2} + 23\left ( 0 \; kJ/mol \; O_{2} \; \right ) \right ] \\[4pt] &= -9977.3 \; kJ/mol \nonumber \end{align*} \]
This is the energy released by the combustion of 1 mol of palmitic acid.
B The energy released by the combustion of 1 g of palmitic acid is
\( \Delta H_{comb}^{o} \; per \; gram =\left ( \dfrac{9977.3 \; kJ}{\cancel{1 \; mol}} \right ) \left ( \dfrac{\cancel{1 \; mol}}{256.42 \; g} \right )= -38.910 \; kJ/g \nonumber \)
As calculated in Equation \(\ref{7.8.8}\), \(ΔH^o_f\) of glucose is −2802.5 kJ/mol. The energy released by the combustion of 1 g of glucose is therefore
\( \Delta H_{comb}^{o} \; per \; gram =\left ( \dfrac{-2802.5 \; kJ}{\cancel{1\; mol}} \right ) \left ( \dfrac{\cancel{1 \; mol}}{180.16\; g} \right ) = -15.556 \; kJ/g \nonumber \)
The combustion of fats such as palmitic acid releases more than twice as much energy per gram as the combustion of sugars such as glucose. This is one reason many people try to minimize the fat content in their diets to lose weight.
Use Table T1 to calculate \(ΔH^o_{rxn}\) for the water–gas shift reaction, which is used industrially on an enormous scale to obtain H2(g):
\[ \ce{ CO ( g ) + H2O (g ) -> CO2 (g) + H2 ( g )} \nonumber\]
- Answer
-
−41.2 kJ/mol
We can also measure the enthalpy change for another reaction, such as a combustion reaction, and then use it to calculate a compound’s \(ΔH^ο_f\) which we cannot obtain otherwise. This procedure is illustrated in Example \(\PageIndex{3}\).
Beginning in 1923, tetraethyllead [\(\ce{(C2H5)4Pb}\)] was used as an antiknock additive in gasoline in the United States. Its use was completely phased out in 1986 because of the health risks associated with chronic lead exposure. Tetraethyllead is a highly poisonous, colorless liquid that burns in air to give an orange flame with a green halo. The combustion products are \(\ce{CO2(g)}\), \(\ce{H2O(l)}\), and red \(\ce{PbO(s)}\). What is the standard enthalpy of formation of tetraethyllead, given that \(ΔH^ο_f\) is −19.29 kJ/g for the combustion of tetraethyllead and \(ΔH^ο_f\) of red PbO(s) is −219.0 kJ/mol?
Given: reactant, products, and \(ΔH^ο_{comb}\) values
Asked for: \(ΔH^ο_f\) of the reactants
Strategy:
- Write the balanced chemical equation for the combustion of tetraethyl lead. Then insert the appropriate quantities into Equation \(\ref{7.8.5}\) to get the equation for ΔHοf of tetraethyl lead.
- Convert \(ΔH^ο_{comb}\) per gram given in the problem to \(ΔH^ο_{comb}\) per mole by multiplying \(ΔH^ο_{comb}\) per gram by the molar mass of tetraethyllead.
- Use Table T1 to obtain values of \(ΔH^ο_f\) for the other reactants and products. Insert these values into the equation for \(ΔH^ο_f\) of tetraethyl lead and solve the equation.
Solution:
A The balanced chemical equation for the combustion reaction is as follows:
\[\ce{2(C2H5)4Pb(l) + 27O2(g) → 2PbO(s) + 16CO2(g) + 20H2O(l)} \nonumber\]
Using Equation \(\ref{7.8.5}\) gives
\[ \Delta H_{comb}^{o} = \left [ 2 \Delta H_{f}^{o}\left ( PbO \right ) + 16 \Delta H_{f}^{o}\left ( CO_{2} \right ) + 20 \Delta H_{f}^{o}\left ( H_{2}O \right )\right ] - \left [2 \Delta H_{f}^{o}\left ( \left ( C_{2}H_{5} \right ) _{4} Pb \right ) + 27 \Delta H_{f}^{o}\left ( O_{2} \right ) \right ] \nonumber \]
Solving for \(ΔH^o_f [\ce{(C2H5)4Pb}]\) gives
\[ \Delta H_{f}^{o}\left ( \left ( C_{2}H_{5} \right ) _{4} Pb \right ) = \Delta H_{f}^{o}\left ( PbO \right ) + 8 \Delta H_{f}^{o}\left ( CO_{2} \right ) + 10 \Delta H_{f}^{o}\left ( H_{2}O \right ) - \dfrac{27}{2} \Delta H_{f}^{o}\left ( O_{2} \right ) - \dfrac{\Delta H_{comb}^{o}}{2} \nonumber \]
The values of all terms other than \(ΔH^o_f [\ce{(C2H5)4Pb}]\) are given in Table T1.
B The magnitude of \(ΔH^o_{comb}\) is given in the problem in kilojoules per gram of tetraethyl lead. We must therefore multiply this value by the molar mass of tetraethyl lead (323.44 g/mol) to get \(ΔH^o_{comb}\) for 1 mol of tetraethyl lead:
\[\begin{align*} \Delta H_{comb}^{o} &= \left ( \dfrac{-19.29 \; kJ}{\cancel{g}} \right )\left ( \dfrac{323.44 \; \cancel{g}}{mol} \right ) \\[4pt] &= -6329 \; kJ/mol \end{align*} \]
Because the balanced chemical equation contains 2 mol of tetraethyllead, \(ΔH^o_{rxn}\) is
\[\begin{align*} \Delta H_{rxn}^{o} &= 2 \; \cancel{mol \; \left ( C_{2}H{5}\right )_4 Pb} \left ( \dfrac{-6329 \; kJ}{1 \; \cancel{mol \; \left ( C_{2}H{5}\right )_4 Pb }} \right ) \\[4pt] &= -12,480 \; kJ \end{align*}\]
C Inserting the appropriate values into the equation for \(ΔH^o_f [\ce{(C2H5)4Pb}]\) gives
\[ \begin{align*} \Delta H_{f}^{o} \left [ \left (C_{2}H_{4} \right )_{4}Pb \right ] & = \left [1 \; mol \;PbO \;\times 219.0 \;kJ/mol \right ]+\left [8 \; mol \;CO_{2} \times \left (-393.5 \; kJ/mol \right )\right ] +\left [10 \; mol \; H_{2}O \times \left ( -285.8 \; kJ/mol \right )\right ] + \left [-27/2 \; mol \; O_{2}) \times 0 \; kJ/mol \; O_{2}\right ] \left [12,480.2 \; kJ/mol \; \left ( C_{2}H_{5} \right )_{4}Pb \right ]\\[4pt]
&= -219.0 \; kJ -3148 \; kJ - 2858 kJ - 0 kJ + 6240 \; kJ = 15 kJ/mol \end{align*}\]
Ammonium sulfate, \(\ce{(NH4)2SO4}\), is used as a fire retardant and wood preservative; it is prepared industrially by the highly exothermic reaction of gaseous ammonia with sulfuric acid:
\[ \ce{2NH3(g) + H2SO4(aq) \rightarrow (NH4)2SO4(s)} \nonumber \]
The value of \(ΔH^o_{rxn}\) is -179.4 kJ/mole \(\ce{H2SO4}\). Use the data in Table T1 to calculate the standard enthalpy of formation of ammonium sulfate (in kilojoules per mole).
- Answer
-
−1181 kJ/mol
Calculating DH° using DHf°: https://youtu.be/Y3aJJno9W2c
Summary
- The standard state for measuring and reporting enthalpies of formation or reaction is 25 oC and 1 atm.
- The elemental form of each atom is that with the lowest enthalpy in the standard state.
- The standard state heat of formation for the elemental form of each atom is zero.
The enthalpy of formation (\(ΔH_{f}\)) is the enthalpy change that accompanies the formation of a compound from its elements. Standard enthalpies of formation (\(ΔH^o_{f}\)) are determined under standard conditions: a pressure of 1 atm for gases and a concentration of 1 M for species in solution, with all pure substances present in their standard states (their most stable forms at 1 atm pressure and the temperature of the measurement). The standard heat of formation of any element in its most stable form is defined to be zero. The standard enthalpy of reaction (\(ΔH^o_{rxn}\)) can be calculated from the sum of the standard enthalpies of formation of the products (each multiplied by its stoichiometric coefficient) minus the sum of the standard enthalpies of formation of the reactants (each multiplied by its stoichiometric coefficient)—the “products minus reactants” rule. The enthalpy of solution (\(ΔH_{soln}\)) is the heat released or absorbed when a specified amount of a solute dissolves in a certain quantity of solvent at constant pressure.
Contributors and Attributions
Modified by Joshua Halpern (Howard University)

