1.2: Structure of Amino Acids
- Page ID
- 165253
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\)
Key structural features
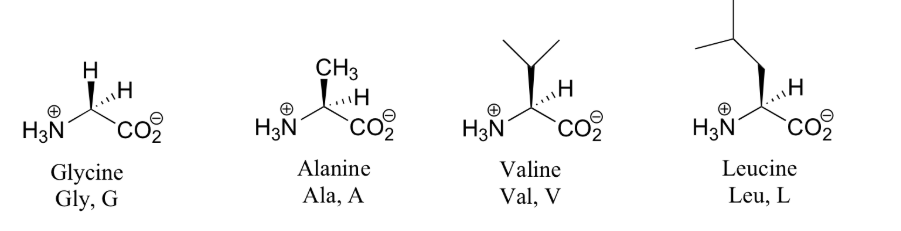
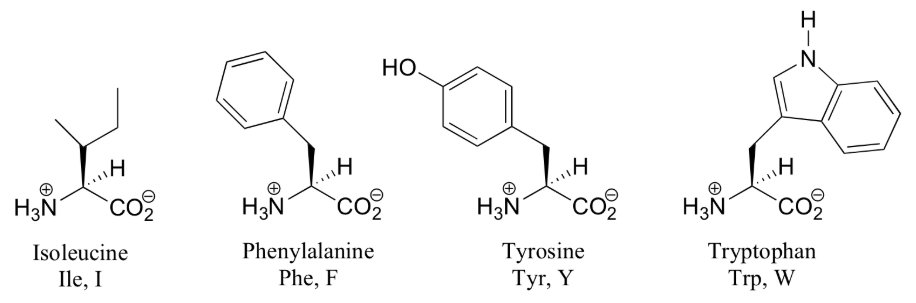
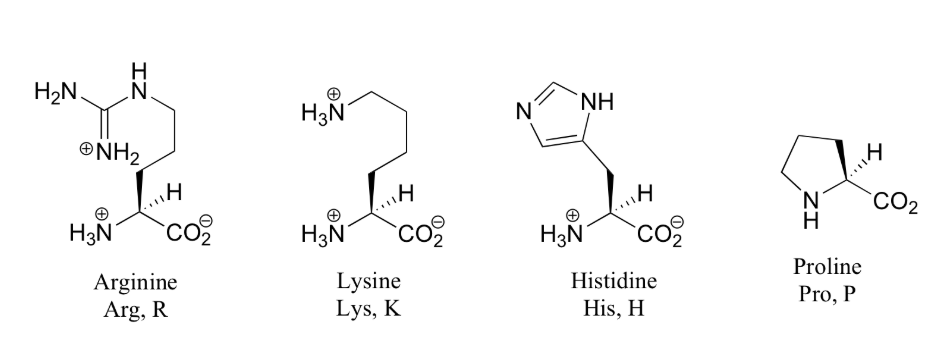
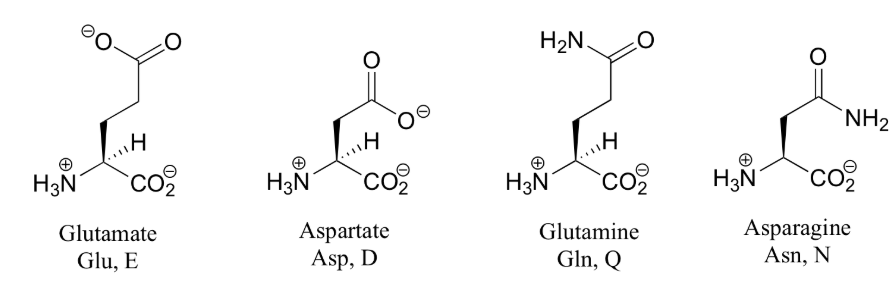
Amino acids are the building blocks (monomers) of proteins. 20 different amino acids are used to synthesize proteins. The shape and other properties of each protein is dictated by the precise sequence of amino acids in it.
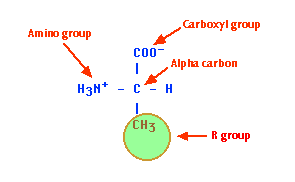
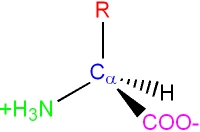
Each amino acid consists of an "alpha carbon atom" is tetrahedral and chiral (i.e. each of the four functional groups are different) to which is attached
- a hydrogen atom
- an amino group (hence "amino" acid)
- a carboxyl group (-COOH). This gives up a proton and is thus an acid (hence amino "acid")
- one of 20 different "R" groups. It is the structure of the R group that determines which of the 20 it is and its special properties. Amino acids are also known as "residues". The amino acid shown here is Alanine.

Amino acid monomers are chemically linked to form linear polymers known as proteins.
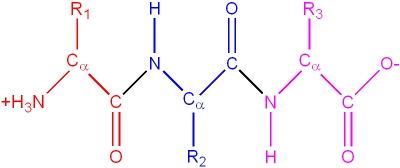
Note: this is drawn "flat" for clarity, but the Ca are still tetrahedral (the H atom on the Ca is also not shown in this diagram)
Key structural features:
- A "peptide bond" is formed by a condensation reaction between the carboxylic acid of one amino acid with the amino group of the next amino acid
- The amino acid R1, at the "amino terminus" of the polymer is the "first" amino acid. The residue (R3 in the above diagram) at the carboxyl terminal is known as the "last" amino acid. These termini define the directionality of the protein.
Contributors
- Mike Blaber (Florida State University)
- Organic Chemistry With a Biological Emphasis by Tim Soderberg (University of Minnesota, Morris)

