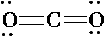Homework 26
- Page ID
- 28941
Q2.40
A children's cough syrup contains 25.0mg of Benzocaine (a numbing agent) per 5.00 milliliters of syrup. The bottle instructs a parent to give a max of 5.00mg per kilogram of body weight per day. How many mL of the cough syrup can a 32.0 lb toddler take in one day?
Solution
Strategy:
What do we know?
A The cough syrup is concentrated at 25.0mg Benzocaine per 5.0 mL► 25.0mg/5mL. We can divide by 5 to simplify this ratio so we know how many mL contain 5.0mg, the recommended per kg dose. ► 5.0mg/1mL
B The recommended maximum dose is 5.0mg per kilogram of the child's weight. ► 5.0mg/kg
What are we trying to find?
- The amount of mL needed to supply 5mg per kilogram of body weight.
- Child's weight in kg
Since the ratio of Benzocaine to kilogram of weight is 5:1, we can find the weight in kilograms and use dimensional analysis to find the correct ratio.
Solving:
A To convert, we can use dimensional analysis. Remember that each "fraction" represents a ratio of two things to each other. In order for dimensional analysis to work, units for the given information have to match the units in the denominator of the ratio to the right of it. (Think of it this way, we cant divide apples by oranges!)
\[32lb \cdot \frac{1lb}{2.204kg}= 14.519kg\]
B When calculating sigfigs, we round our answer at the end. We are going to use this number in our dimensional analysis question.
C Using dimensional analysis, we can multiply the weight by the ratio of Benzocaine per kg. The kg unit cancels because it is present both as a numerator and denominator. Cancelling them is equivalent to simplifying them to an implied 1. The units on the far right should be the answer we are looking for.
\[14.519kg \cdot \frac{5mL}{1kg}= 72.595mL\]
Answer:
The child can take a maximum of 72.6mL of the cough syrup.
Q6.29
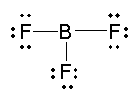
Draw Lewis Structures for the following molecular compounds: NH3, CO2, BF3
Solution
Strategy:
What do we know?
A We know to draw a Lewis structure, we will be using valence electrons. We know how to find these from looking at the periodic table. The group number-10 gives us valence electrons, or we can use the atomic number-core electrons.
| Element | Valence Electrons |
| N | 5 |
| H | 1 |
| C | 4 |
| O | 6 |
| B | 3 |
| F | 7 |
B The next step involves the steps to draw a Lewis structure.
- Draw the correct skeleton structure (arrange the elements without any bonds yet). Hydrogens should always be on the outside, and the least electronegative atom should be in the center.
- Calculate the total number of valence electrons. If you are presented with a + ion, subtract one from the total for each positive charge. If you are presented with a - ion, add one to the total for each negative charge. Remember to multiply the valence electrons by subscripts given to the compounds. For example, when calculating CO2, we would add 4 for carbon + (6)(2) for oxygen because there are two of them.
- NH3 has 8 valence electrons total
- CO2 has 16 valence electrons total
- BF3 has 24 valence electrons total
- Place a pair of bonding electrons (2) between each element. If you have extra electrons, begin to fill the octet of the other atoms with LONE PAIRS, starting with the terminal atoms.
- If you have used all of your valence electrons and some structures are not "happy", do not have 8 valence electrons, use lone pairs to form double or triple bonds.
- Check your work! Make sure the number of electrons in the structure match the amount in step 2.
Draw the structures.
Answer: