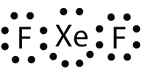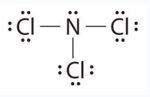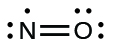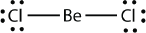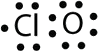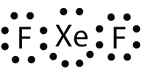As important and useful as the octet rule is in chemical bonding, there are many covalent molecules with central atoms that do not have eight electrons in their Lewis structures. This does not mean that the octet rule is useless—quite the contrary. As with many rules, there are exceptions, or violations. These molecules fall into three categories:
Odd-electron molecules
Although they are few, some stable compounds, often called free radicals, have an odd number of electrons in their valence shells. With an odd number of electrons, at least one atom in the molecule will have to violate the octet rule. Examples of stable, odd-electron molecules are \(\ce{NO}\), \(\ce{NO2}\), and \(\ce{ClO2}\). The Lewis electron dot diagram for \(\ce{NO}\), a compound produced in internal combustion engines when oxygen and nitrogen react at high temperatures, is as follows:
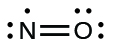
As you can see, the nitrogen and oxygen share four electrons between them. The oxygen atom has an octet of electrons but the nitrogen atom has only seven valence electrons, two electrons in the double bond, one lone pair, and one additional lone electron. Although \(\ce{NO}\) is a stable compound, it is very chemically reactive, as are most other odd-electron compounds.
Electron-deficient molecules
These stable compounds have less than eight electrons around an atom in the molecule, i.e. they have less than an octet. The most common examples are the covalent compounds of beryllium and boron. For example, beryllium can form two covalent bonds, resulting in only four electrons in its valence shell:
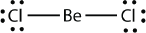
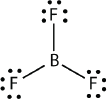
Boron commonly makes only three covalent bonds, resulting in only six valence electrons around the \(\ce{B}\) atom. A well-known example is \(\ce{BF3}\):
Expanded Octet Molecules
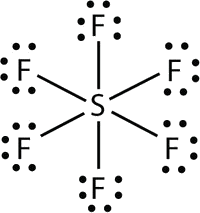
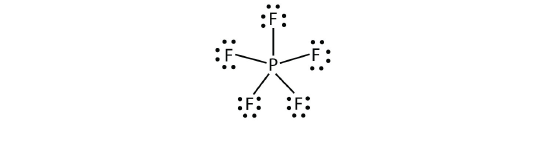
Elements in the second period of the periodic table (n = 2) can accommodate only eight electrons in their valence shell orbitals because they have only four valence orbitals (one 2s and three 2p orbitals). Elements in the third and higher periods (n ≥ 3) have more than four valence orbitals and can share more than four pairs of electrons with other atoms because they have empty d orbitals in the same shell. Molecules formed from these elements have expanded octets and are sometimes called hypervalent molecules. Phosphorous pentachloride shares five pairs of electrons for a total of ten electrons in the valence shell.
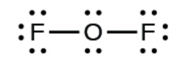
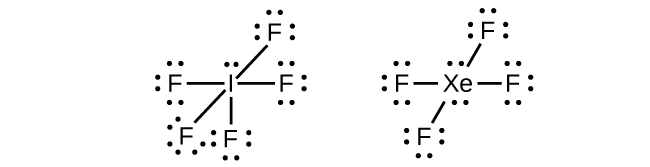
In some expanded octet molecules, such as IF5 and XeF4, some of the electrons in the outer shell of the central atom are lone pairs:
Example \(\PageIndex{2}\)
Identify each violation to the octet rule by drawing a Lewis electron dot diagram.
- \(\ce{ClO}\)
- \(\ce{SF6}\)
Solution
- With one Cl atom and one O atom, this molecule has 6 + 7 = 13 valence electrons, so it is an odd-electron molecule. A Lewis electron dot diagram for this molecule is as follows:
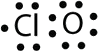
- In \(\ce{SF6}\), the central \(\ce{S}\) atom makes six covalent bonds to the six surrounding F atoms, so it is an expanded valence shell molecule. Its Lewis electron dot diagram is as follows:
Exercise \(\PageIndex{2}\): Xenon Difluoride
Identify the violation to the octet rule in \(\ce{XeF2}\) by drawing a Lewis electron dot diagram.
- Answer
-
The Xe atom has an expanded valence shell with more than eight electrons around it.