12.8: Water - A Remarkable Molecule
- Page ID
- 48664
- Interpret the unique properties of water in terms of a phase diagram.
Earth is the only known body in our solar system that has liquid water existing freely on its surface; life on Earth would not be possible without the presence of liquid water. Water has several properties that make it a unique substance among substances. It is an excellent solvent; it dissolves many other substances and allows those substances to react when in solution. In fact, water is sometimes called the universal solvent because of this ability. Water has unusually high melting and boiling points (0°C and 100°C, respectively) for such a small molecule. The boiling points for similar-sized molecules, such as methane (BP = −162°C) and ammonia (BP = −33°C), are more than 100° lower. Though a liquid at normal temperatures, water molecules experience a relatively strong intermolecular interaction that allows them to maintain the liquid phase at higher temperatures than expected.
Unlike most substances, the solid form of water is less dense than its liquid form, which allows ice to float on water. In colder weather, lakes and rivers freeze from the top, allowing animals and plants to continue to live underneath. Water also requires an unusually large amount of energy to change temperature. While 100 J of energy will change the temperature of 1 g of Fe by 230°C, this same amount of energy will change the temperature of 1 g of H2O by only 100°C. Thus, water changes its temperature slowly as heat is added or removed. This has a major impact on weather, as storm systems like hurricanes can be impacted by the amount of heat that ocean water can store. Water’s influence on the world around us is affected by these properties. Isn’t it fascinating that such a small molecule can have such a big impact?
Phase Diagram for Water
Water is a unique substance in many ways. One of these special properties is the fact that solid water (ice) is less dense than liquid water just above the freezing point. The phase diagram for water is shown in the figure below.
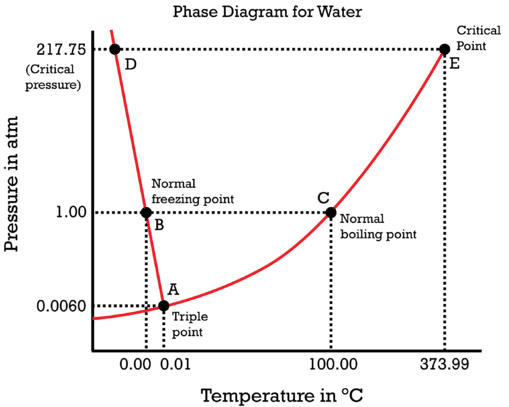
Notice one key difference between the general phase diagram and the phase diagram for water. In water's diagram, the slope of the line between the solid and liquid states is negative rather than positive. The reason for this is that water is an unusual substance, as its solid state is less dense than the liquid state. Ice floats in liquid water. Therefore, a pressure change has the opposite effect on those two phases. If ice is relatively near its melting point, it can be changed into liquid water by the application of pressure. The water molecules are actually closer together in the liquid phase than they are in the solid phase.
Refer again to water's phase diagram (figure above). Notice point \(E\), labeled the critical point. What does that mean? At \(373.99^\text{o} \text{C}\), particles of water in the gas phase are moving very, very rapidly. At any temperature higher than that, the gas phase cannot be made to liquefy, no matter how much pressure is applied to the gas. The critical pressure \(\left( P_\text{C} \right)\) is the pressure that must be applied to the gas at the critical temperature in order to turn it into a liquid. For water, the critical pressure is very high, \(217.75 \: \text{atm}\). The critical point is the intersection point of the critical temperature and the critical pressure.
Summary
- Solid water is less dense than liquid water just above the freezing point.
- The critical temperature \(\left( T_\text{C} \right)\) of a substance is the highest temperature at which the substance can possibly exist as a liquid.
- The critical pressure \(\left( P_\text{C} \right)\) is the pressure that must be applied to the gas at the critical temperature in order to turn it into a liquid.
- The critical point is the intersection point of the critical temperature and the critical pressure.

