22.3: Reaction of acyl derivatives with weak nucleophiles
- Page ID
- 225904
Introduction
As we saw with aldehydes and ketones in section 21.2, weak nucleophiles such as alcohols and water do not directly add to an unactivated carbonyl group; instead, the C=O must first be activated using acid. The same applies with carboxylic acid derivatives, so there are several acyl substitution reactions that involve acid catalysis. This type of mechanism is best exemplified by Fischer esterification, which involves the acid catalyzed reaction between an activated (protonated) form of a carboxylic acid and and alcohol (as weak nucleophile) to form an ester.
Preparation of esters by Fischer esterification
Learning Objectives
- To identify and describe the substances from which most esters are prepared.
The reaction is reversible. As a specific example of an esterification reaction, butyl acetate can be made from acetic acid and 1-butanol.
A Closer Look: Condensation Polymers
A commercially important esterification reaction is condensation polymerization, in which a reaction occurs between a dicarboxylic acid and a dihydric alcohol (diol), with the elimination of water. Such a reaction yields an ester that contains a free (unreacted) carboxyl group at one end and a free alcohol group at the other end. Further condensation reactions then occur, producing polyester polymers.
The most important polyester, polyethylene terephthalate (PET), is made from terephthalic acid and ethylene glycol monomers:
Polyester molecules make excellent fibers and are used in many fabrics. A knitted polyester tube, which is biologically inert, can be used in surgery to repair or replace diseased sections of blood vessels. PET is used to make bottles for soda pop and other beverages. It is also formed into films called Mylar. When magnetically coated, Mylar tape is used in audio- and videocassettes. Synthetic arteries can be made from PET, polytetrafluoroethylene, and other polymers.
Summary
Esters are made by the reaction of a carboxylic acid with an alcohol, a process that is called esterification.
Synthesis of ‘banana oil’
Many esters can be synthesized simply by mixing together a carboxylic acid and an alcohol, along with a catalytic amount of sulfuric acid. A popular reaction in undergraduate organic lab courses is the preparation of isopentyl acetate (also called ‘banana oil’ because it is a flavor component in bananas) from acetic acid and isopentyl alcohol.
Notice the double arrows in the above figure: because esters and carboxylic acids are of approximately the same stability, this esterification reaction is highly reversible.
The main role of the acid catalyst in this reaction is to protonate the carboxylic acid, thus making the carbonyl carbon more electrophilic.
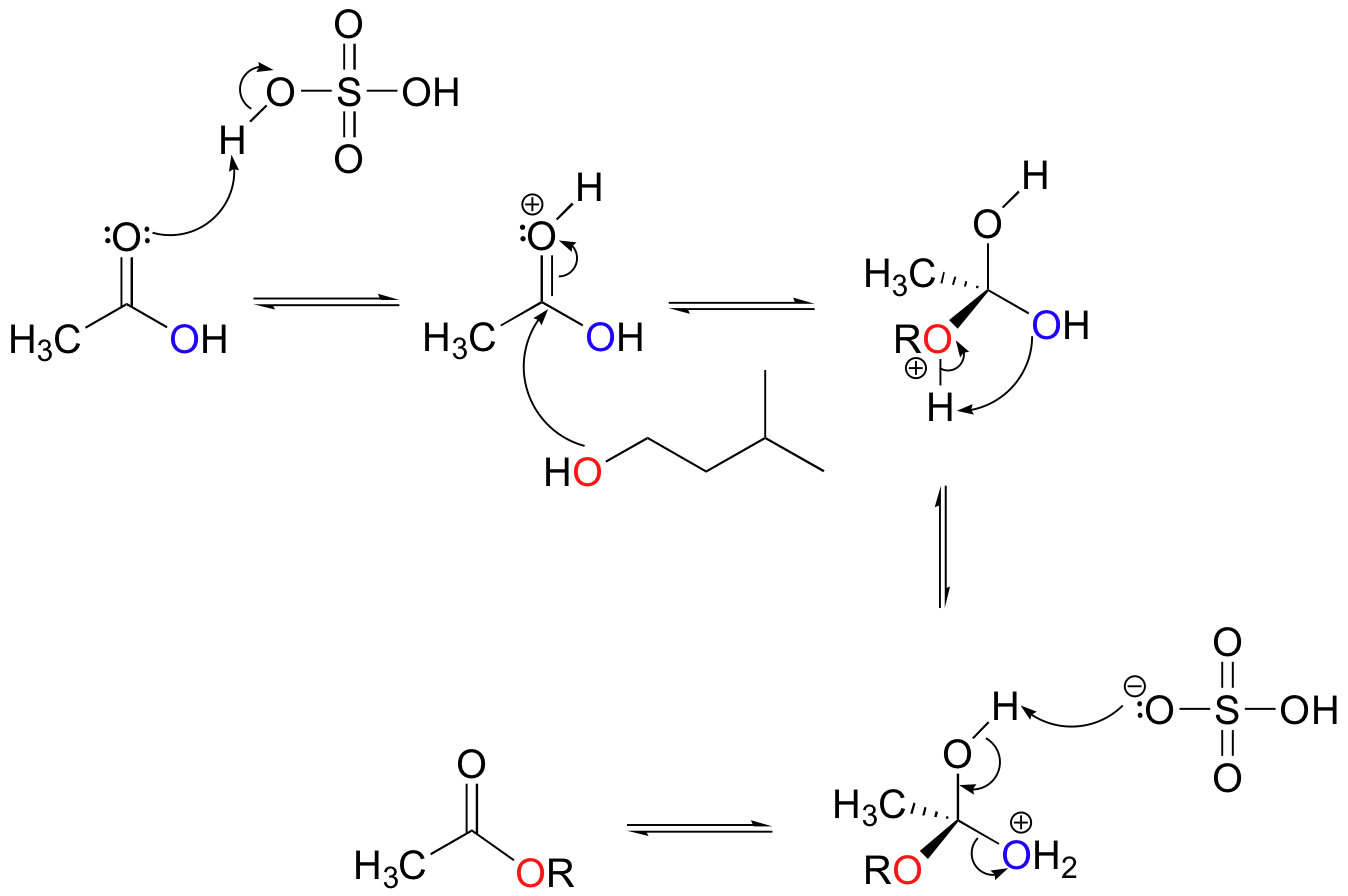
Exercises
Exercise 12.4: In the synthesis of isopentyl acetate, an excess of acetic acid is used, usually about three molar equivalents relative to the amount of isopentyl alcohol. What is the reason for this?
Exercise 12.5: Could a similar esterification reaction occur with the addition of a small amount of NaOH instead of H2SO4? Explain.
Exercise 12.6: Draw a complete mechanism for the exact reverse of the acid-catalyzed esterification shown above. What would you call this reaction in organic chemistry terms?
Many other fragrant esters can be synthesized in similar reactions. A few examples of esters found in foods are given below.
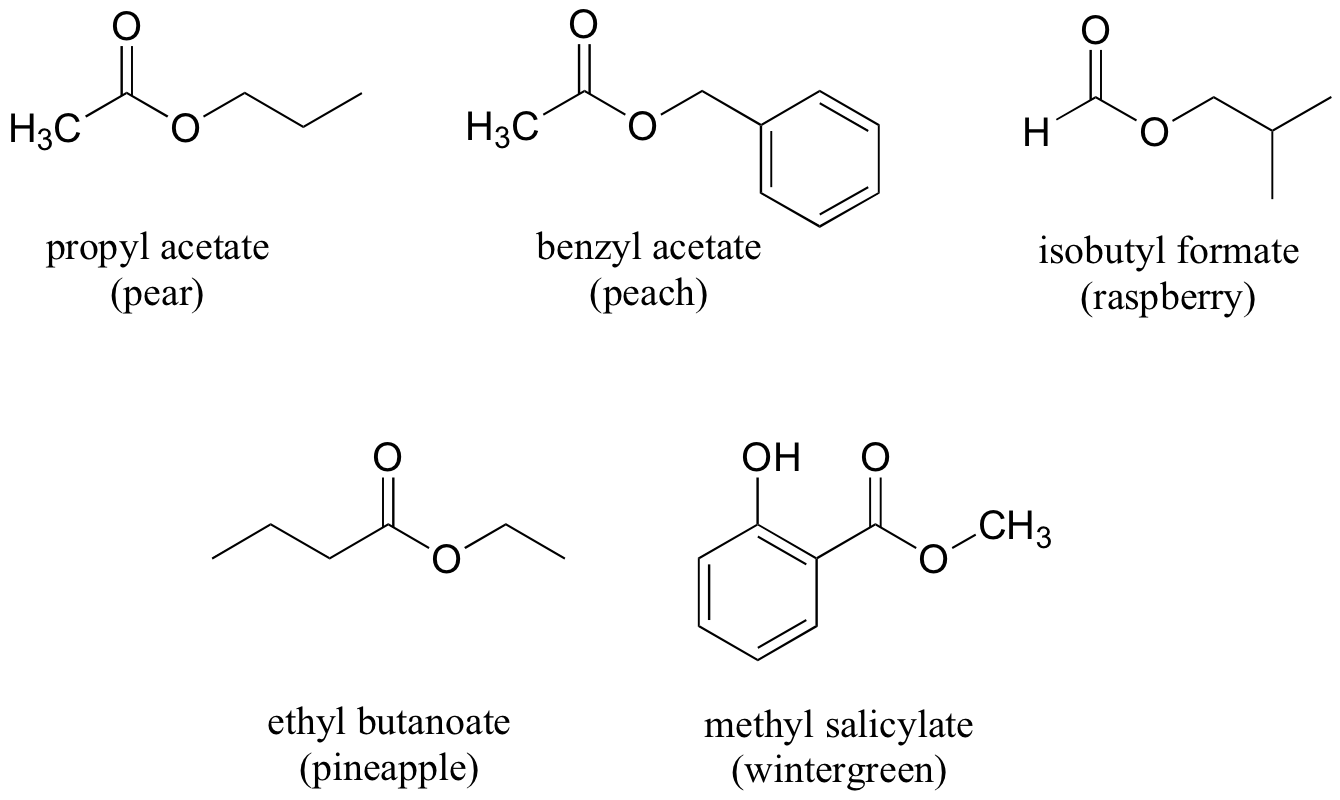
Exercises
Exercise 12.7: For each fragrant ester shown above, provide the carboxylic acid and alcohol starting materials needed.
Concept Review Exercises
- From what carboxylic acid and what alcohol can the ester isopropyl nonanoate be made?
- From what carboxylic acid and what alcohol can the ester cyclobutyl butyrate be made?
Answers
[reveal-answer q=”544827″]Show Answer[/reveal-answer]
[hidden-answer a=”544827″]
- nonanoic acid and isopropyl alcohol
- butyric acid and cyclobutyl alcohol[/hidden-answer]
Exercises
Write the equation for the reaction of acetic acid with each compound.
- ethanol
- 1-butanol in the presence of a mineral acid catalyst
Write the equation for the reaction of benzoic acid with each compound.
- methanol
- 1-propanol in the presence of a mineral acid catalyst
Answer
[reveal-answer q=”460266″]Show Answer[/reveal-answer]
[hidden-answer a=”460266″]
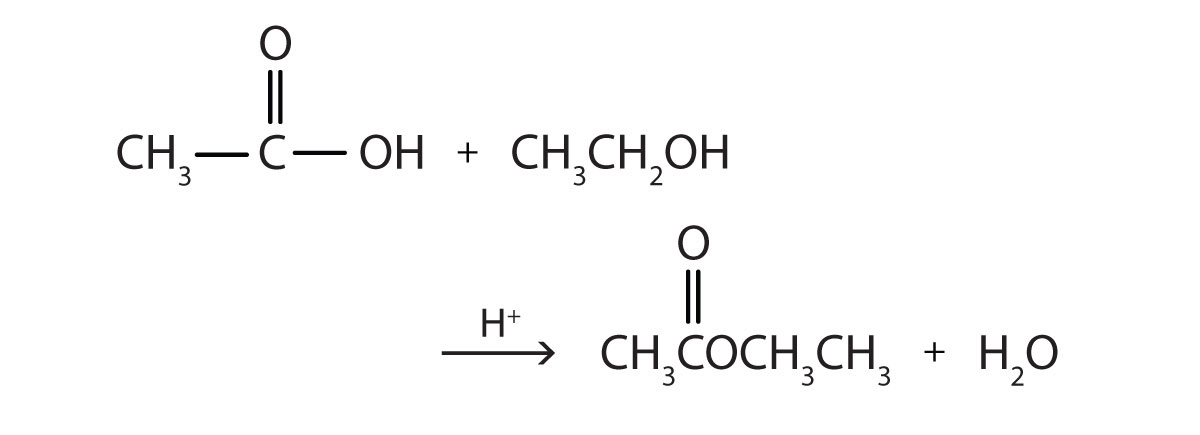
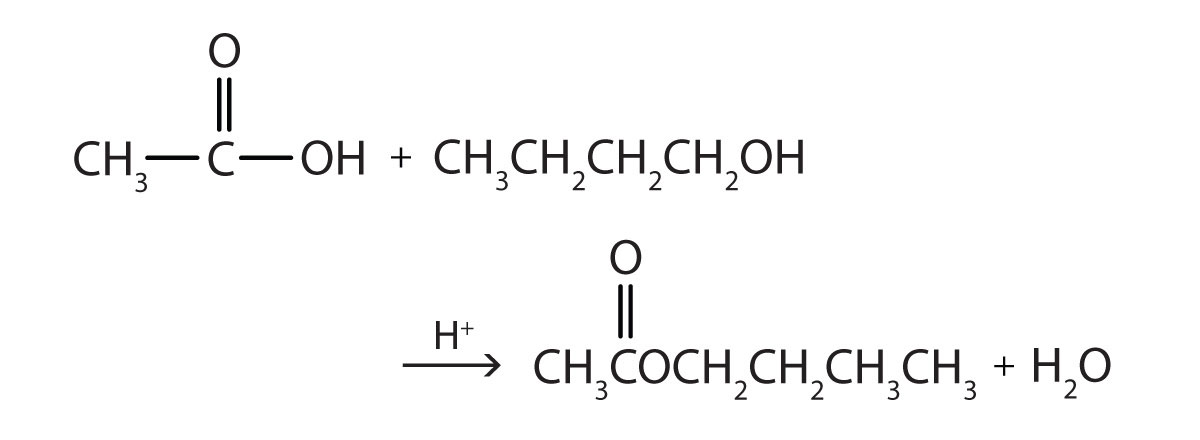 [/hidden-answer]
[/hidden-answer]
Preparation of acyl chlorides
Thionyl chloride is a liquid at room temperature and has the formula $$SOCl_2$$. It reacts with carboxylic acids to produce an acyl chloride, giving off sulfur dioxide and hydrogen chloride gases. For example:
\[ CH_3COOH + SOCl_2 \rightarrow CH_3COCl +SO_2 + HCl\]
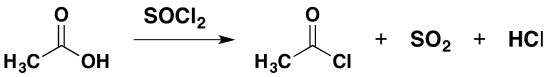
The separation is simplified to an extent because the by-products are both gases. Fractional distillation is still required to separate the acyl chloride from any excess acid or thionyl chloride.
Contributors
Jim Clark (Chemguide.co.uk)
Video

Hydrolysis of Amides
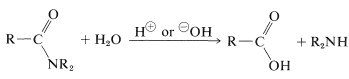 Amide hydrolysis can be an important route to amines. Hydrolysis under acidic conditions requires strong acids such as sulfuric or hydrochloric, and temperatures of about $$100^\text{o}$$ for several hours. The mechanism involves protonation of the amide on oxygen followed by attack of water on the carbonyl carbon. The tetrahedral intermediate formed dissociates ultimately to the carboxylic acid and the ammonium salt:
Amide hydrolysis can be an important route to amines. Hydrolysis under acidic conditions requires strong acids such as sulfuric or hydrochloric, and temperatures of about $$100^\text{o}$$ for several hours. The mechanism involves protonation of the amide on oxygen followed by attack of water on the carbonyl carbon. The tetrahedral intermediate formed dissociates ultimately to the carboxylic acid and the ammonium salt: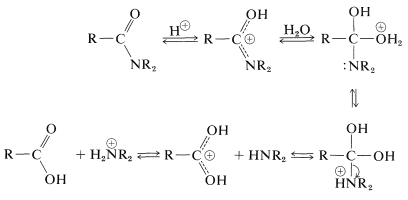 A brief summary of important amide reactions follows:
A brief summary of important amide reactions follows: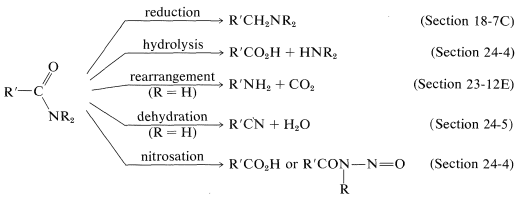
Contributors
- John D. Robert and Marjorie C.Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, “You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format.”
- 24.3: Synthesis of Amides. Authored by: John D. Robert and Marjorie C. Caserio . Located at: https://chem.libretexts.org/Textbook_Maps/Organic_Chemistry/Book%3A_Basic_Principles_of_Organic_Chemistry_(Roberts_and_Caserio)/24%3A_Organonitrogen_Compounds_II%3A_Amides%2C_Nitriles%2C_and_Nitro_Compounds/24.3%3A_Synthesis_of_Amides. Project: Chemistry LibreTexts. License: CC BY-NC-SA: Attribution-NonCommercial-ShareAlike
- 15.7: Preparation of Esters. Located at: https://chem.libretexts.org/Textbook_Maps/Introductory_Chemistry/Book%3A_The_Basics_of_GOB_Chemistry_(Ball_et_al.)/15%3A_Organic_Acids_and_Bases_and_Some_of_Their_Derivatives/15.07_Preparation_of_Esters. Project: Chemistry LibreTexts. License: CC BY-NC-SA: Attribution-NonCommercial-ShareAlike
- Preparation of Acyl Chlorides. Authored by: Jim Clark. Located at: https://chem.libretexts.org/Textbook_Maps/Organic_Chemistry/Supplemental_Modules_(Organic_Chemistry)/Acid_Halides/Synthesis_of_Acid_Halides/Preparation_of_Acyl_Chlorides. Project: Chemistry LibreText. License: CC BY-NC-SA: Attribution-NonCommercial-ShareAlike
- Organic Chemistry with a Biological Emphasis. Authored by: Tim Soderberg. Located at: https://chem.libretexts.org/Textbook_Maps/Organic_Chemistry/Book%3A_Organic_Chemistry_with_a_Biological_Emphasis_(Soderberg)/12%3A_Acyl_substitution_reactions/12.4%3A_Esters. Project: Chemistry Libretexts. License: CC BY-NC-SA: Attribution-NonCommercial-ShareAlike
- 24.4: Hydrolysis of Amides. Authored by: John D. Robert and Marjorie C. Caserio. Located at: https://chem.libretexts.org/Textbook_Maps/Organic_Chemistry/Book%3A_Basic_Principles_of_Organic_Chemistry_(Roberts_and_Caserio)/24%3A_Organonitrogen_Compounds_II%3A_Amides%2C_Nitriles%2C_and_Nitro_Compounds/24.4%3A_Hydrolysis_of_Amides. Project: Chemistry Libretexts. License: CC BY-NC-SA: Attribution-NonCommercial-ShareAlike


