7.10: Nucleophilic Substitution Reactions (Problems)
- Page ID
- 234567
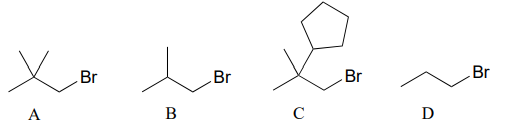
P7.1: Rank the following molecules in order of how fast they would be expected to react with \(CH_3SNa\) in acetone. (\(CH_3SNa\) is simply the sodium salt of \(CH_3S^-\). \(Na^+\) is a spectator ion.)
P7.2: Draw line structures representing the most stable cation with the given molecular formula:
- \(C_3H_7^+\)
- \(C_4H_9^+\)
- \(C_3H_8N^+\)
- \(C_4H_7^+\)
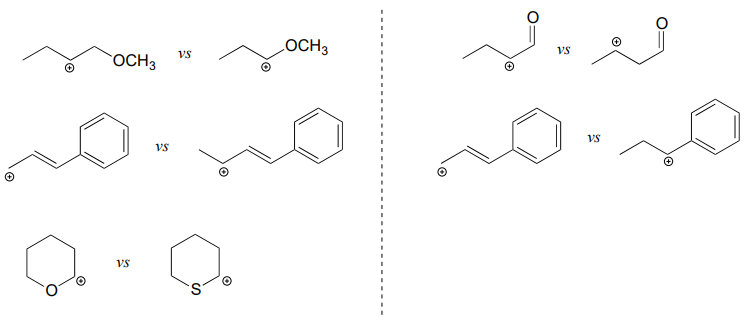
P7.3: For each pair of carbocations below, choose the one that is more stable, and explain your reasoning.
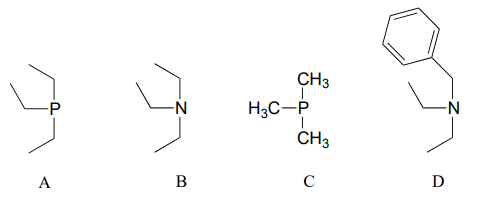
P7.4: Arrange the following species in order of increasing nucleophilicity in protic solvent:
P7.5: Predict the organic products of the following nucleophilic substitution reactions, all of which are carried out in polar aprotic solvent. Show stereochemistry at chiral carbons. Hints: \(Na_2CO_3\), sodium carbonate, is a weak base. For part (f): What is the conjugate acid of \(NH_2^-\)? What is the \(pK_a\) of this conjugate acid, and what is the \(pK_a\) of a terminal alkyne?
a.
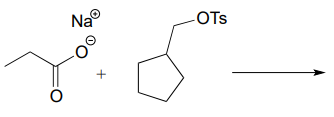
b.
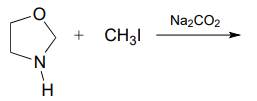
c.
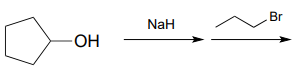
d.
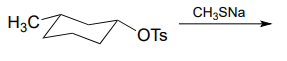
e.
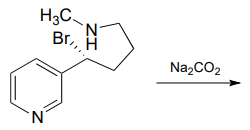
f.

P7.6: Which of the reactions in the previous problem has a unimolecular rate determining step? Explain.
P7.7: From the following pairs, select the compound that would react more rapidly with bromomethane in acetone solvent.
- water or hydroxide ion
- \(CH_3S^-\) or \(CH_3OH\)
- \(CH_2S^-\) or \(CH_3SH\)
- acetate ion or hydroxide ion
- diethyl sulfide or diethyl ether
- dimethylamine or diethylether
- trimethylamine or 2,2-dimethylpropane
P7.8: Methyl iodide (0.10 mole) is added to a solution that contains 0.10 mole \(NaOCH_3\) and 0.10 mole \(NaSCH_3\).
- Predict the most abundant neutral organic product that would form, and explain your reasoning.
- Assume that you isolate a mixture the major product (which you predicted in part) along with a smaller amount of a different nucleophilic substitution product. Explain briefly but specifically how you could use \(^1H-NMR\) to determine the ratio of the two products in the mixture.
P7.9: For each pair of compounds, predict which will more rapidly undergo solvolysis in methanol solution.
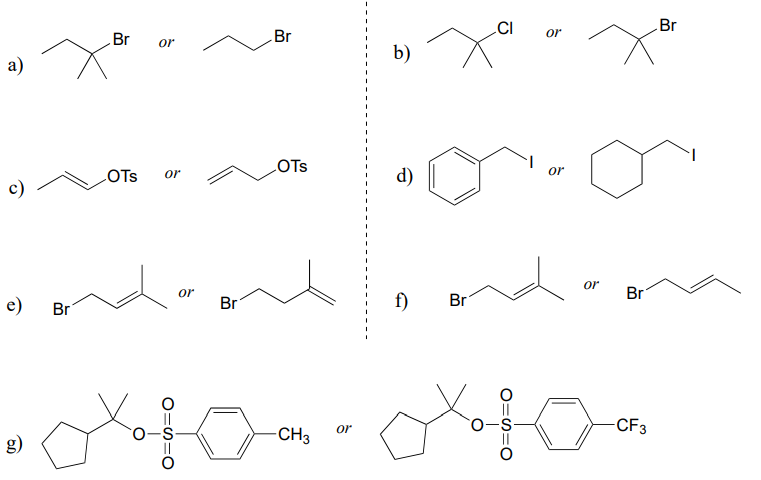
P7.10: Predict the solvolysis product(s) of each of the reactions below. Consider both regiochemistry and stereochemistry.
a.
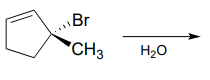
b.
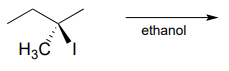
c.
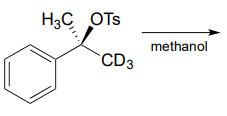
e) Draw a complete curved-arrow mechanism for the formation of the secondary allylilc alcohol product in part (a).
P7.11: Show starting compounds that would lead to the following products through nucleophilic substitution reactions.
a.
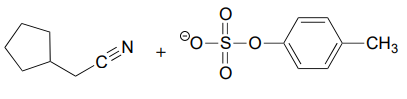
b.

c.
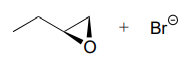
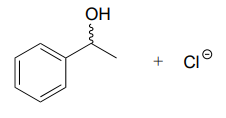
d.
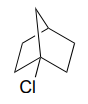
P7.12: The fused ring compound shown below is very unreactive to nucleophilic substitution, even with a powerful nucleophile.. Explain. (Hint – consider bond geometry - a model will be very helpful!)
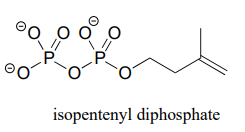
P7.13 Laboratory synthesis of isopentenyl diphosphate - the 'building block' molecule used by nature for the construction of isoprenoid molecules (section 1.3A) - was accomplished by first converting isopentenyl alcohol into an alkyl tosylate then displacing the tosylate group with an inorganic pyrophosphate nucleophile. Based on this verbal description, draw a mechanism for the second (nucleophilic substitution) step, showing starting and ending compounds for the step and curved arrows for electron movement.
P7.14: Choline, an important neutotransmitter in the nervous system, is formed from 2-(N,N-dimethylamino)ethanol:

- Besides the enzyme and the starting compound, what other important biomolecule do you expect plays a part in the reaction?
- Draw a mechanism for the reaction.
- Briefly explain how \(^1H-NMR\) could be used to distinguish between the substrate and the product of this reaction.
P7.15 The following is a reaction in the biosynthesis of morphine in opium poppies. (Science 1967, 155, 170; J. Biol. Chem 1995, 270, 31091).
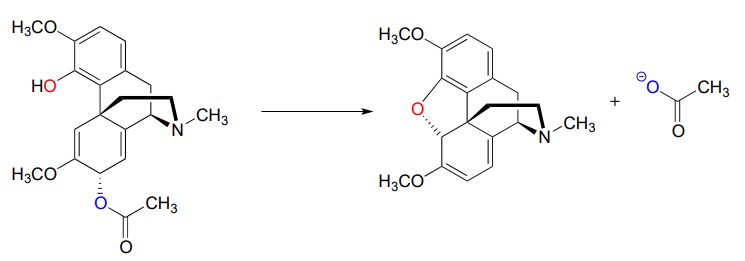
- Draw a complete mechanism, assuming an \(S_N1\) pathway.
- What would you expect to be the most noticeable difference between the IR spectrum of the product and that of the substrate?
- This reaction is an example of the regiospecificity of enzymatic nucleophilic substitution reactions noted earlier in the chapter. Draw two alternate nucleophilic, ring-closing steps for this reaction (leading to different products from what is shown above), and explain why these alternate pathways are both less favorable than the actual reaction catalyzed by the enzyme.
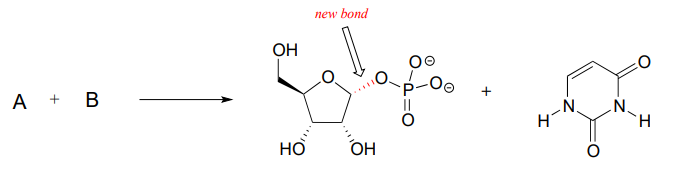
P7.16: The enzymatic reaction below, which is part of the metabolism of nucleic acids, proceeds by an \(S_N1\) mechanism. The new bond formed in the substitution is indicated.
- Predict the structures of the two substrates A and B.
- Draw a complete mechanism, and use resonance drawings to illustrate how both the carbocation intermediate and the leaving group are stabilized.
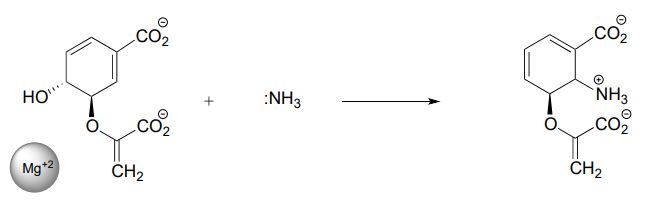
P7.17: Below is the first step of the reaction catalyzed by anthranilate synthase, an enzyme involved in biosynthesis of the amino acid tryptophan.
- This reaction is somewhat unusual in that the leaving group is a hydroxide anion, which is of course is normally thought to be a very poor leaving group. However, studies show that an \(Mg^{+2}\) ion is bound in the active site close to the hydroxide. Explain how the presence of the magnesium ion contributes to the viability of hydroxide as a leaving group.
- Draw a complete mechanism for the reaction, assuming an \(S_N1\) pathway.
P7.18: The reaction below is part of the biosynthesis of peptidoglycan, a major component of bacterial cell walls. Is it likely to proceed by a nucleophilic substitution mechanism? Explain.
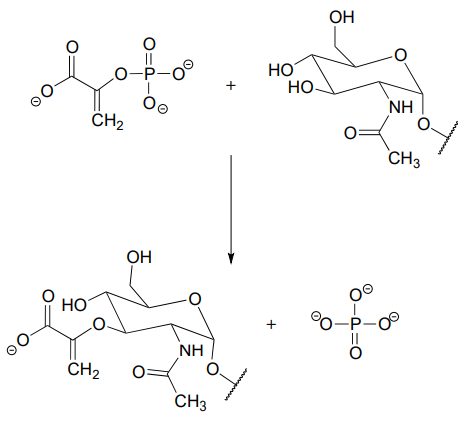
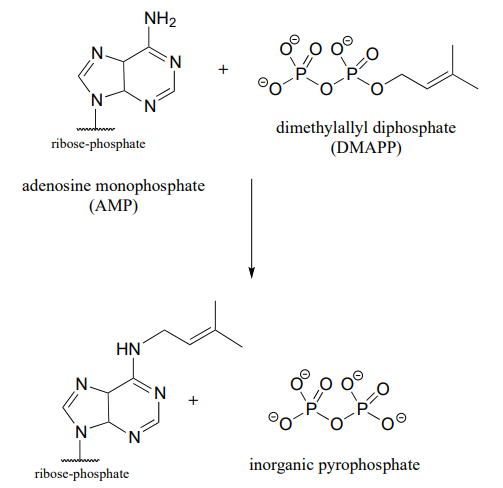
P7.19: Compare the reaction below, catalyzed by the enzyme AMP-DMAPP transferase, to the protein prenyltransferase reaction we learned about in section 8.8, the mechanism of which, as we discussed, is thought to be mostly \(S_N2\)-like with some \(S_N1\)-like character.
- Is the AMP-DMAPP transferase reaction below likely to have more or less \(S_N1\)-like character compared to the protein prenyltransferase reaction? Explain.
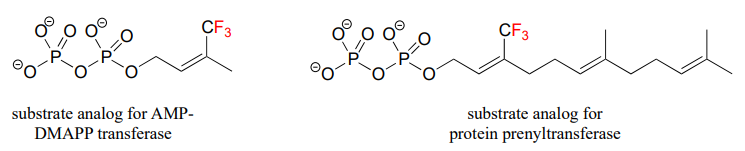
- Given your answer to part (a), which reaction is likely to be more dramatically slowed down when a fluorinated isoprenoid substrate analog is substituted for the natural substrate? Explain.
P7.20: In a classic experiment in physical organic chemistry, (\(R\))-2-iodooctane was allowed to react (non-enzymatically) with a radioactive isotope of iodide ion, and the researchers monitored how fast the radioactive iodide was incorporated into the alkane (the rate constant of incorporation, \(k_i\)) and also how fast optical activity was lost (the rate constant of racemization, \(k_r\)). They found that the rate of racemization was, within experimental error, equal to twice the rate of incorporation. Discuss the significance of this result - what does it say about the actual mechanism of the reaction?
Contributors
Organic Chemistry With a Biological Emphasis by Tim Soderberg (University of Minnesota, Morris)


