Osmosis a Type of Passive Transport
- Page ID
- 184233
Osmosis is the second type of passive transport and refers specifically to the diffusion of water across a semipermeable membrane.
If you have an area that has a high concentration of dissolved particles in a solution, the lower the concentration of water molecules in that solution.
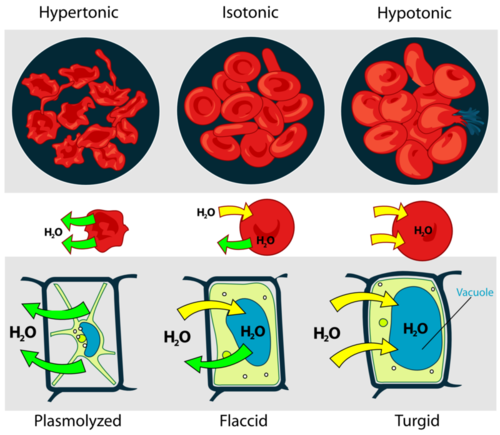
Osmosis affects cells when water diffuses across cell membranes. Look at the image here, as you read about the different types of solutions.
In hypertonic solutions, there is a low concentration of water outside of the cell, so water leaves the cell, and the cells tend to shrivel.
In isotonic solutions things are pretty much at equilibrium, water moves in and water moves out.
In hypotonic solutions there is a low concentration of water inside of the cell, so water moves into the cell making the cell turgid or causing the cell to burst.

