Chapter 2.4: Electronic Structure of the Transition Metals
- Page ID
- 19878
 |
Prince George's Community College |
 |
| Unit I: Atoms Unit II: Molecules Unit III: States of Matter Unit IV: Reactions Unit V: Kinetics & Equilibrium Unit VI: Thermo & Electrochemistry Unit VII: Nuclear Chemistry |
||
Learning Objective
- To write the electron configuration of the transition metals and understand the basis for the exceptions to the normal order of filling.
Now you can use the information you learned in Section 2.3 to determine the electronic structure of every element in the periodic table. The process of describing each atom’s electronic structure consists, essentially, of beginning with hydrogen and adding one proton and one electron at a time to create the next heavier element in the table. Well almost, but the exceptions are instructional.
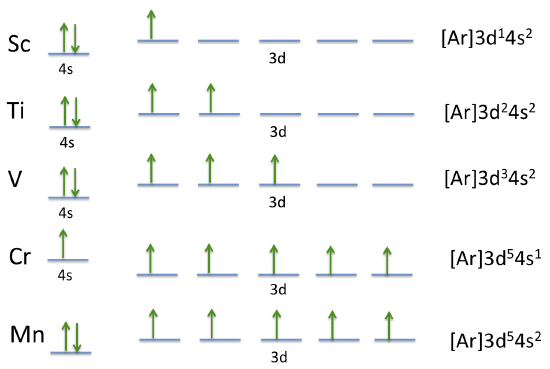
Adding a proton and an electron to form the next atom results in small changes in the energy levels relative to each other but the order remains the same, at least until we get to the d levels, where in some atoms the relative energies of the ns and the (n-1)d orbitals shifts. In the fourth period, this happens for Cr and Cu, which, instead of having two electrons in the 4s orbital have only one. The first three elements in the d block of the fourth period sequentially have one more d electron than the last. In agreement with Hund's rules each of these is added to a different d orbital with parallel spins.
For Cr, something different happens, of the six electrons, five are found in the 4d level and only one in the 4s. Electron repulsion and favoring parallel spins moves the 3d level below the 4s when there are 6 electrons. The normal pattern resumes with Mn (manganese).
Figure 2.4.1 The electronic configurations of the first five fourth period transition metals Notice how the relative positions of the 4s and 3d orbitals move relative to each other as more electrons are added.
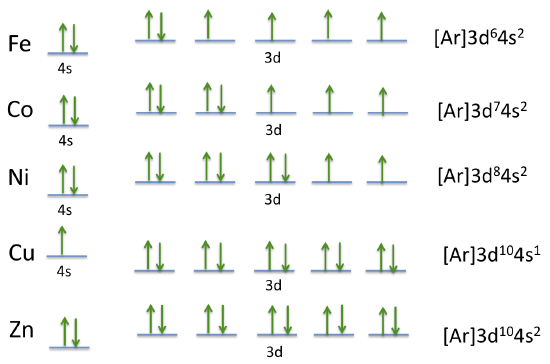
If we continue on to Zn, the "exception" repeats itself with Cu, where there are now 10 electrons in the 3d level and only one in the 4s.
We can look at the electron configurations of the rest of the d block elements.
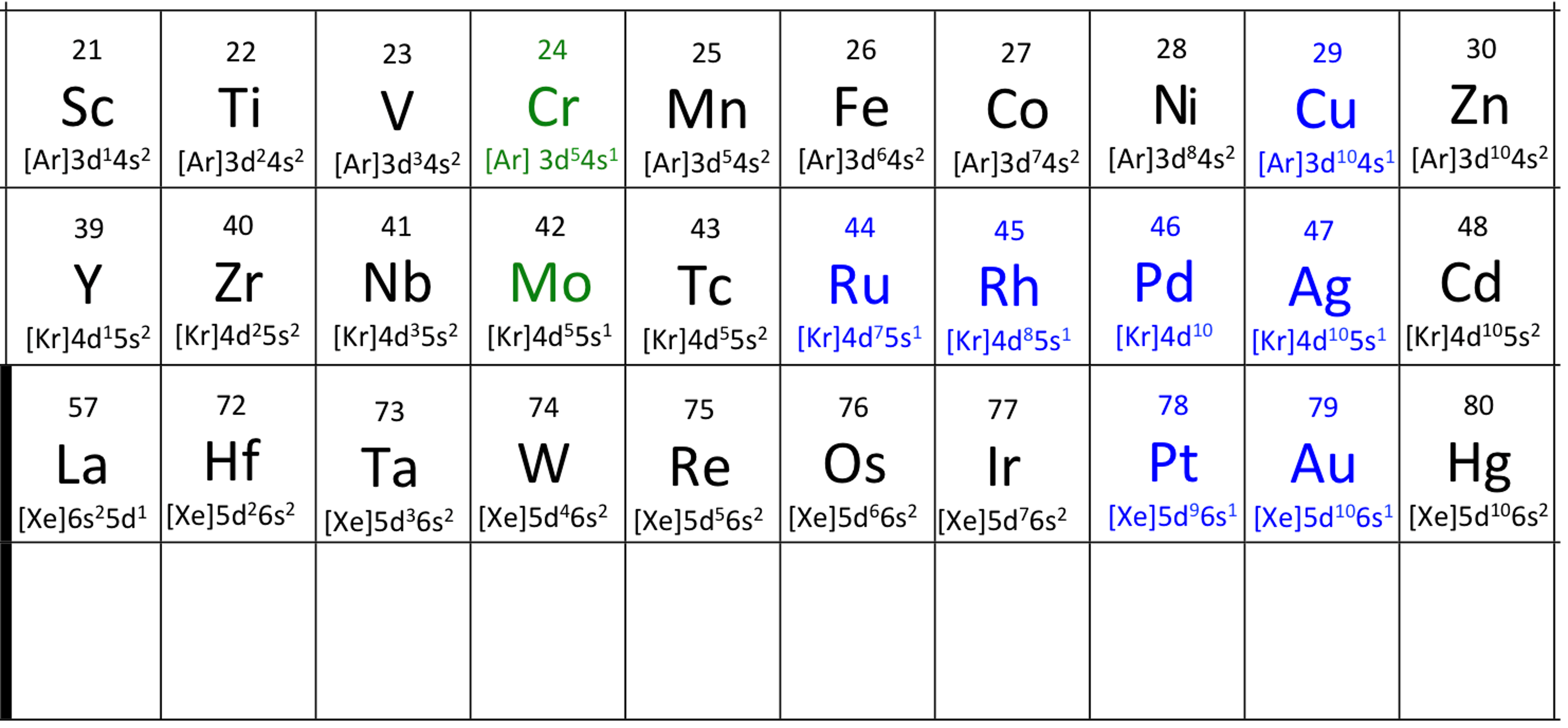
Figure 2.4.3 The electronic configurations of the transition metals. Only the ns and (n-1)d electrons have been shown to save space. The 14 electrons in the atoms from Hf to Hg have not been written in. The electron configuration of Mo (molybdenum) is similar to that of Cr, and both are shown in green. Those metals shown in blue, with the exception of Pd, paladium, have only one ns electron. Pd has none. Cu, Ag, Au and Pd have ten electrons in the (n-1)d orbitals. These metals are all very soft, fairly unreactive, rare, and with the exception of Cu used in jewelry.
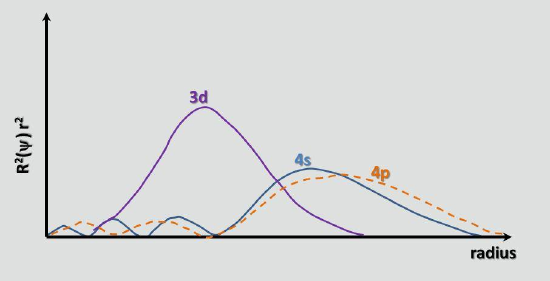
The transition metals, as a general rule, have similar properties. The reason for this is that the extent of the orbitals from the nucleus depends on the principal quantum numbers. Thus, the orbitals of the ns electrons extend further out than those of the (n-1) d electrons in the same periods, and therefore are more available for bonding and reactions. As other atoms and molecules approach the metal atoms, the ns electrons are the ones that are first affected.
Contributors
- Anonymous
Modified by Joshua Halpern, Scott Sinex and Scott Johnson