7.10: Consequences of Climate Change
- Page ID
- 189480
As part of your assigned reading for this section, read the article “The Coming Storm” by Don Belt published in National Geographic: http://ngm.nationalgeographic.com/pr...desh/belt-text In this section, we will discuss the effects of climate change, both those that have already been observed, as well as future predictions based on scientific climate models (see section 7.5 for a discussion of scientific models). Here, the differences between the terms global warming and climate change become apparent. Global warming refers to the increase in the average temperature of the Earth’s atmosphere due to elevated greenhouse gas concentrations, heightening the greenhouse effect. We have already observed this increase occurring, as you saw in Figure 7.5.2 from section 7.5. We have also seen, and expect to continue to see, other changes occurring in the climate of the Earth. Furthermore, changes have been observed, and we expect to continue to observe, changes in other chemical, physical, and biological aspects of the Earth’s environment. We will only discuss some of the consequences of climate change in this section, including changes in temperature, precipitation, ocean level, and ocean acidity. There are many more changes that have been seen, and are projected to continue in the future. These include: changes in the amount and distribution of ice and snow, changes in seasonality, ecosystem shift, and habitat changes of plant and animal populations, in addition to others. For more information about these consequence of climate change, visit this site: http://www.epa.gov/climatechange/sci...ors/index.html.
Temperature and precipitation
Temperature and precipitation are the two most direct impacts on the Earth’s climate due to climate change. By now, you should already understand why an increase in greenhouse gas levels in the atmosphere causes an increase in temperature. But why does it also impact precipitation patterns? As you already know, water vapor is an important component of the Earth’s atmosphere (see Chapter 6). As the air in the troposphere warms and cools, the amount of water vapor that it holds changes dramatically. Here in Georgia, we have very hot and humid summers. The high summer humidity in this region is possible due to the increased capability warm air has to hold water vapor. Simply put, warmer air can hold more water than cooler air. As air cools, its ability to hold water vapor decreases, and any excess water will leave the air as liquid water. A great example of this is the formation of dew on surfaces overnight. During the day, the temperature is warmer than it is at night, and the air has a relatively high holding capacity for water vapor. When the sun sets, the air cools, decreasing its capacity to hold water vapor. That extra water must go somewhere, and it does that by accumulating on surfaces. Similarly, when warm and cool air fronts collide, the chances for rain and thunderstorms increase. Furthermore, an increase in temperature enhances evaporation occurring at the Earth’s surface. This increased evaporation leads to greater concentrations of water vapor in the atmosphere which can lead to increased precipitation.
The change in temperature that we have already seen in the Earth’s average atmospheric temperature is relatively small (about 0.6 °C, according to Figure 7.5.2 from section 7.5). However, as with many of the aspects of climate change, the potential for greater changes increases dramatically as time progresses in the future. This can be seen in Figure \(\PageIndex{1}\), which displays a model of the predicted temperature increase. Notice that these changes occur relatively rapidly, and are not uniform across the globe. What might be some of the reasons for this?
.png?revision=1&size=bestfit&width=818&height=482) Figure \(\PageIndex{1}\): Projected changes in global average temperatures under three emissions scenarios (rows) for three different time periods (columns). Changes in temperatures are relative to 1961- 1990 averages. The scenarios come from the IPCC Special Report on Emissions Scenarios: B1 is a low emissions scenario, A1B is a medium-high emissions scenario, and A2 is a high emissions scenario. Source: IPCC Working Group I: The Physical Science Basis, 2007.
Figure \(\PageIndex{1}\): Projected changes in global average temperatures under three emissions scenarios (rows) for three different time periods (columns). Changes in temperatures are relative to 1961- 1990 averages. The scenarios come from the IPCC Special Report on Emissions Scenarios: B1 is a low emissions scenario, A1B is a medium-high emissions scenario, and A2 is a high emissions scenario. Source: IPCC Working Group I: The Physical Science Basis, 2007.
Changes in precipitation occur due to a variety of factors, including changes in atmospheric water vapor content due to changing temperature, as discussed above. Also at play is the heightened evaporation rate of water on Earth’s surface under warmer temperatures. More evaporation leads to more precipitation. Finally, shifts in wind patterns impact the distribution of precipitation events. As you can see in Figure \(\PageIndex{2}\), there are some areas of the globe that are expected to have an increase in precipitation, while others are expected to have a dramatic decrease. Some major population centers projected to have a moderate to severe precipitation increase include (population estimates of the metropolitan area given in parentheses): New York, United States (20.1 million); Bogotá, Colombia (12.1 m.); and Manila, Philippines (11.9 m.). What sort of challenges might these cities face in the future as they deal with this change in their climate?
.png?revision=1&size=bestfit&width=655&height=410) Figure 7.6.1). The plotted precipitation differences were computed as the difference between the 2081 to 2100 20-year averages minus the 1951 to 2000 50-year average. Blue areas project increases in precipitation; brown areas project decreases.
Figure 7.6.1). The plotted precipitation differences were computed as the difference between the 2081 to 2100 20-year averages minus the 1951 to 2000 50-year average. Blue areas project increases in precipitation; brown areas project decreases.
In contrast, many more major metropolitan areas are projected to have a moderate to severe precipitation decrease (droughts) by the end of the 21st century. These include Delhi, India (21.8 m.); Lagos, Nigeria (21 m.); São Paulo, Brazil (20.9 m.); Kolkata, India (14.6 m.); Istanbul, Turkey (14.4 m.); Los Angeles, United States (13.3 m.); Rio de Janeiro, Brazil (12 m.); Paris, France (12 m.); and Lahore, Pakistan (11.3 m.). The largest challenge that these areas are likely to face is a dwindling water supply for drinking and agriculture. See Chapter 8 for more detail on challenges faced by societies to supply clean, reliable water to their populations and farms.
Additional challenges may be felt by all areas of the world with regard to changes in the seasonality or timing of precipitation, as well the form in which precipitation falls (e.g., mist or downpour; rain, ice, or snow). All of these factors affect the availability of soil water for plants, the flow of rivers and streams, and the overall accessibility of water worldwide. Furthermore, scientists predict an increase in the number and severity of storms as climate change progresses. For a full discussion of the potential impacts of this, see the assigned article.
Sea level rise While we know that water continuously cycles around the world (see Chapter 8 for information on the water cycle), and that the overall quantity of water on Earth will not change due to global climate change, the distribution of this water is changing. In particular, oceans are increasing in volume while land ice stores (such as glaciers) are decreasing. This contributes to an increase in sea level worldwide (Figure \(\PageIndex{3}\)).
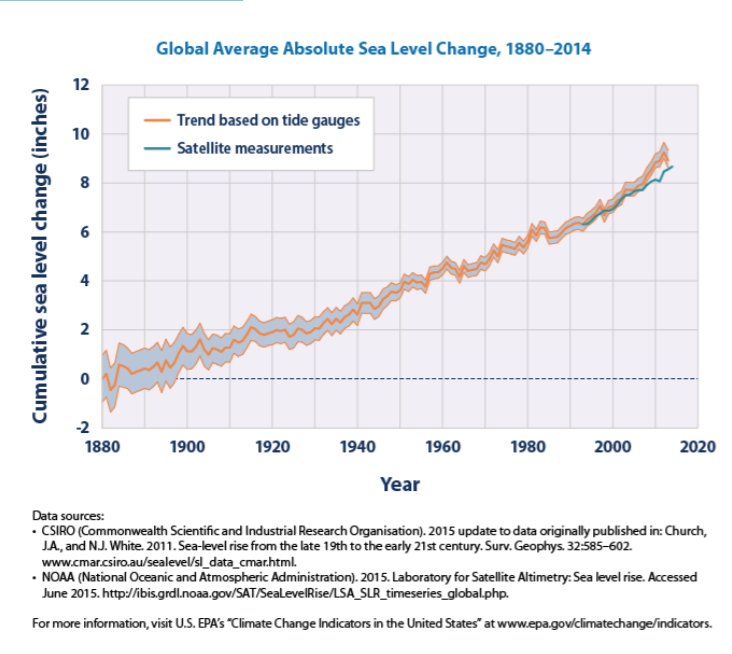.png?revision=1&size=bestfit&width=660&height=590) Figure \(\PageIndex{3}\): This graph shows average absolute cumulative changes in sea level for the world’s oceans since 1880, based on a combination of tide gauge measurements and recent satellite measurements. The shaded band shows the likely range of values, based on the number of measurements collected and the precision of the methods used.
Figure \(\PageIndex{3}\): This graph shows average absolute cumulative changes in sea level for the world’s oceans since 1880, based on a combination of tide gauge measurements and recent satellite measurements. The shaded band shows the likely range of values, based on the number of measurements collected and the precision of the methods used.
From the data in Figure \(\PageIndex{3}\), we see that sea level has increased at an average of 0.06 inches (0.15 cm) per year over the time period shown above. Most of this rise, however, has occurred within the most recent decades. The rate of increase has gone up to between 0.11 to 0.14 inches (0.28 to 0.36 cm) per year since 1993. There are two forces causing sea level to rise, both caused by climate change. First, the increased global temperature has caused increased ice melting in many regions of the globe. Melting land ice (such as the glacier shown in Figure \(\PageIndex{4}\)) contributes to sea level rise because water that used to be stored in ice sitting on top of land becomes running water which reaches the ocean through runoff. We also observe sea ice melting (see http://www.epa.gov/climatechange/sci ence/indicators/index.html for data and figures). Sea ice, such as the ice that covers the arctic regions of the Northern Hemisphere, has no land underneath it. When it melts, the water stays in the same locations, and the overall sea level does not change.
.png?revision=1) Figure \(\PageIndex{4}\): Glacier Retreat
Figure \(\PageIndex{4}\): Glacier Retreat
The second factor that influences sea level rise is a phenomenon called thermal expansion. Due to the physical properties of water, as water warms, its density decreases. A less dense substance will have fewer molecules in a given area than a more dense substance (see Chapter 1 supplemental material). This means that as the overall temperature of the oceans increases due to global climate change, the same amount of water molecules will now occupy a slightly larger volume. This may not seem significant, but considering the 1.3 billion trillion liters (264 billion gallons) of water in the ocean, even a small change in density can have large effects on sea level as a whole.
Scientists have already documented sea level rise in some areas of the world, including one familiar to most of us: the Southeastern United States. Figure \(\PageIndex{5}\) depicts the measured land area lost due to increasing sea level since 1996. Note that the Southeast (defined here as the Atlantic coast of North Carolina south to Florida) is particularly susceptible to land area loss due to the gently sloping nature of our coastline. Moving northward into the Mid-Atlantic States (defined here as Virginia north to Long Island, New York), coastal habitats tend to have a steeper geography, which protects against some losses.
.png?revision=1&size=bestfit&width=534&height=462) Figure 7.6.5: This graph shows the net amount of land converted to open water along the Atlantic coast during three time periods: 1996–2001, 1996–2006, and 1996–2011. The results are divided into two regions: the Southeast and the Mid-Atlantic. Negative numbers show where land loss is outpaced by the accumulation of new land.
Figure 7.6.5: This graph shows the net amount of land converted to open water along the Atlantic coast during three time periods: 1996–2001, 1996–2006, and 1996–2011. The results are divided into two regions: the Southeast and the Mid-Atlantic. Negative numbers show where land loss is outpaced by the accumulation of new land.
While the ecological effects of sea level rise remain in the United States, we don’t project any catastrophic loss of life, property, or livelihood for some time. This is, in part, due to large investments that we have made in infrastructure to protect our cities and farmlands. This is not the case in many areas of the world. For a discussion of the impacts of sea level rise on less industrialized nations of Bangladesh, Maldives, Kiribati, and Fiji, review the required article reading.
Ocean acidification
Dissolved CO2 is essential for many organisms, including shell-building animals and other organisms that form a hard coating on their exterior (e.g., shellfish, corals, Haptophyte algae). This hard coating is built out of aragonite, a mineral form of the molecule calcium carbonate, CaCO3. These organisms rely on the formation of carbonate ions (see Chapter 1 supplemental material for information on ions), CO3 2-, from dissolved CO2, through a natural, chemical reaction that occurs. This takes place through a chain-reaction equation, where bicarbonate (HCO3- ) is formed as an intermediate, and hydrogen ions (H+ ) are generated (equations \(\PageIndex{1}\) and \(\PageIndex{2}\)).
\[CO_{2} + H_{2}O \leftrightarrow H^{+} + HCO_{3}^{-}\]
\[HCO_{3}^{-} \leftrightarrow H^{+} + CO_{3}^{2-} \]
To have a better visualization of this process, follow along with the interactive graphic at: http://www.whoi.edu/home/oceanus_ima...ification.html.
As you can see, both equations \(\PageIndex{1}\) and \(\PageIndex{2}\) each produce one H+ . This is significant to water chemistry because an increase in H+ concentration means a decrease in the pH of the water. You can see in Figure \(\PageIndex{6}\) that a lower pH means that the liquid is more acidic. As shown in the interactive graphic, an increase in CO2 in the atmosphere causes additional CO2 to be dissolved in the ocean. This means that more CO2 in the atmosphere leads to more acidic ocean environments.
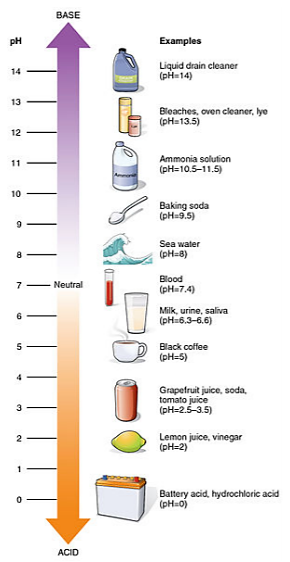.png?revision=1&size=bestfit&width=302&height=585) Figure 7.6.6: The pH scale and relative acidity. Illustration from Anatomy & Physiology, Connexions Web site. http://cnx.org/content/col11496/1.6/, Jun 19, 2013.
Figure 7.6.6: The pH scale and relative acidity. Illustration from Anatomy & Physiology, Connexions Web site. http://cnx.org/content/col11496/1.6/, Jun 19, 2013.
Unfortunately for shell-building animals, the buildup of H+ in the more acidic ocean environment blocks the absorption of calcium and CO32-, and makes the formation of aragonite more difficult. An aragonite deficit is already being documented in many of the world’s oceans, as shown in Figure \(\PageIndex{7}\).
The increasing acidity of the world’s oceans is resulting in habitat changes across the globe. This is only expected to worsen as atmospheric CO2 levels continue to increase. Many organisms, including the corals that are the foundation species of the beautiful coral reefs, are very sensitive to changes in ocean pH. Scientists have documented cases of ecosystem destruction through coral bleaching, caused by the effects of climate change including ocean acidification and increased temperature. For more information, visit the NOAA Coral Reef Conservation Program website: coralreef.noaa.gov/threats/climate/.
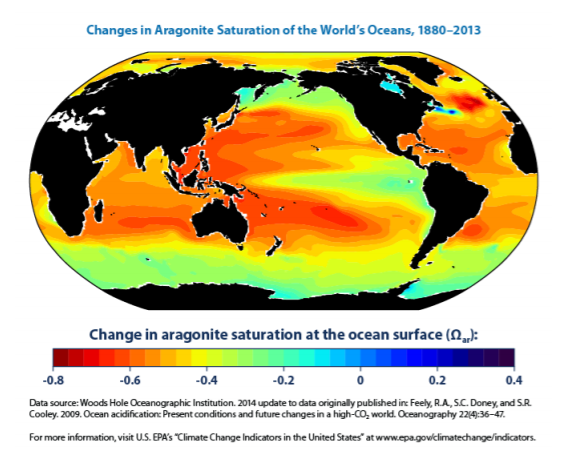.png?revision=1&size=bestfit&width=644&height=515) Figure \(\PageIndex{7}\): This map shows changes in the aragonite saturation level of ocean surface waters between the 1880s and the most recent decade (2004–2013). Aragonite is a form of calcium carbonate that many marine animals use to build their skeletons and shells. A negative change represents a decrease in saturation.
Figure \(\PageIndex{7}\): This map shows changes in the aragonite saturation level of ocean surface waters between the 1880s and the most recent decade (2004–2013). Aragonite is a form of calcium carbonate that many marine animals use to build their skeletons and shells. A negative change represents a decrease in saturation.

