8.1: Introduction
- Page ID
- 236767
A Crash Course of Kinetics, Equilbrum and Thermodynamics
This unit was custom built for Oregon Tech PDX - Metro students. These three topics are central to general chemistry and are used extensively in many other fields, including engineering. Before the OT engineering students go off into their programs, it is important for them to have basic concepts of these three topics. Usually, these subjects are presented in their own unit and take a week to cover each! This crash course will cover the general concepts of kinetics, equilibrium, and thermodynamics, so that students have a basic understanding when they enter their upper division courses. Students that take General Chemistry III will learn this content in much more detail and for those students that plan to take organic chemistry next year, a strong understanding of these three subjects is imperative.
While these three topics are related, they are independent concepts and models. It is very important to distinguish between the three topics.
Kinetics (rates)
From baking a cake to determining the useful lifespan of a bridge, rates of chemical reactions play important roles in our understanding of processes that involve chemical changes. When planning to run a chemical reaction, we should ask at least two questions. The first is: “Will the reaction produce the desired products in useful quantities?” The second question is: “How rapidly will the reaction occur?” A reaction that takes 50 years to produce a product is about as useful as one that never gives a product at all. A third question is often asked when investigating reactions in greater detail: “What specific molecular-level processes take place as the reaction occurs?” Knowing the answer to this question is of practical importance when the yield or rate of a reaction needs to be controlled.
The study of chemical kinetics concerns the second and third questions—that is, the rate at which a reaction yields products and the molecular-scale means by which a reaction occurs. In this chapter, we will examine the factors that influence the rates of chemical reactions, the mechanisms by which reactions proceed, and the quantitative techniques used to determine and describe the rate at which reactions occur.
Equilibrium
Imagine a beach populated with sunbathers and swimmers. As those basking in the sun get too hot and want to cool off, they head into the surf to swim. As the swimmers tire, they head to the beach to rest. If these two rates of transfer (sunbathers entering the water, swimmers leaving the water) are equal, the number of sunbathers and swimmers would be constant, or at equilibrium, although the identities of the people are constantly changing from sunbather to swimmer and back. An analogous situation occurs in chemical reactions. Reactions can occur in both directions simultaneously (reactants to products and products to reactants) and eventually reach a state of balance.
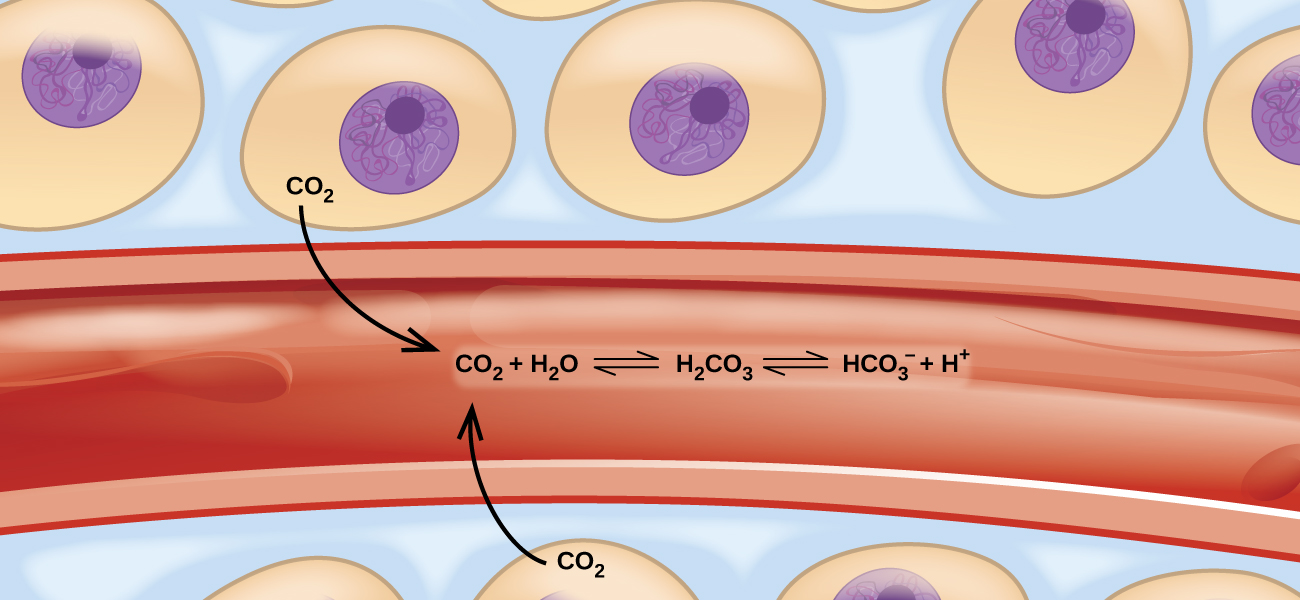
These balanced two-way reactions occur all around and even in us. For example, they occur in our blood, where the reaction between carbon dioxide and water forms carbonic acid (HCO−3)(HCO3−) (Figure 13.0.113.0.1). Human physiology is adapted to the amount of ionized products produced by this reaction (HCO−3HCO3− and H+). In this chapter, you will learn how to predict the position of the balance and the yield of a product of a reaction under specific conditions, how to change a reaction's conditions to increase or reduce yield, and how to evaluate an equilibrium system's reaction to disturbances
Thermodynamics
Among the many capabilities of chemistry is its ability to predict if a process will occur under specified conditions. Thermodynamics, the study of relationships between the energy and work associated with chemical and physical processes, provides this predictive ability. Previous chapters in this text have described various applications of thermochemistry, an important aspect of thermodynamics concerned with the heat flow accompanying chemical reactions and phase transitions. This chapter will introduce additional thermodynamic concepts, including those that enable the prediction of any chemical or physical changes under a given set of conditions.
Contributors and Attributions
Paul Flowers (University of North Carolina - Pembroke), Klaus Theopold (University of Delaware) and Richard Langley (Stephen F. Austin State University) with contributing authors. Textbook content produced by OpenStax College is licensed under a Creative Commons Attribution License 4.0 license. Download for free at http://cnx.org/contents/85abf193-2bd...a7ac8df6@9.110).


