1.3C: Solutions of Liquid Solutes in Liquid Solvents
- Page ID
- 206907
Whereas all gases will mix to form solutions regardless of the proportions, liquids are much more fussy. Some liquids, such as ethyl alcohol and water, are miscible in all proportions. Others, like the proverbial oil and water, are not; each liquid has only a limited solubility in the other, and once either of these limits is exceeded, the mixture separates into two phases.
| solute → | liquid |
|---|---|
| energy to disperse solute | varies |
| energy to introduce into solvent | varies |
| increase in entropy | moderate |
| miscibility | "like dissolves like" |
The reason for this variability is apparent from the table. Mixing of two liquids can be exothermic, endothermic, or without thermal effect, depending on the particular substances. Whatever the case, the energy factors are not usually very large, but neither is the increase in randomness; the two factors are frequently sufficiently balanced to produce limited miscibility.
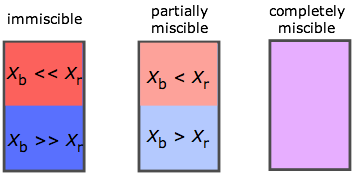
The range of possibilities is shown here in terms of the mole fractions X of two liquids A and B. If A and B are only slightly miscible, they separate into two layers according to their relative densities. Note that when one takes into account trace levels, no two liquids are totally immiscible.
Like Dissolves Like
A useful general rule is that liquids are completely miscible when their intermolecular forces are very similar in nature; “like dissolves like”. Thus water is miscible with other liquids that can engage in hydrogen bonding, whereas a hydrocarbon liquid in which London or dispersion forces are the only significant intermolecular effect will only be completely miscible with similar kinds of liquids.
Substances such as the alcohols, CH3(CH2)nOH, which are hydrogen-bonding (and thus hydrophilic) at one end and hydrophobic at the other, tend to be at least partially miscible with both kinds of solvents. If n is large, the hydrocarbon properties dominate and the alcohol has only a limited solubility in water. Very small values of n allow the –OH group to dominate, so miscibility in water increases and becomes unlimited in ethanol (n = 1) and methanol (n = 0), but miscibility with hydrocarbons decreases owing to the energy required to break alcohol-alcohol hydrogen bonds when the non polar liquid is added.
These considerations have become quite important in the development of alternative automotive fuels based on mixing these alcohols with gasoline. At ordinary temperatures the increased entropy of the mixture is great enough that the unfavorable energy factor is entirely overcome, and the mixture is completely miscible. At low temperatures, the entropy factor becomes less predominant, and the fuel mixture may separate into two phases, presenting severe problems to the fuel filter and carburetor.
Contributors
Stephen Lower, Professor Emeritus (Simon Fraser U.) Chem1 Virtual Textbook


