Sulfa Drug Synthesis
Sulfa drugs are an important group of synthetic antimicrobial agents (pharmaceuticals) that contain the sulfonamide group. The synthesis of sulfanilamide (a sulfa drug) illustrates how the reactivity of aniline can be modified to make possible an electrophilic aromatic substitution. The corresponding acetanilide undergoes chlorosulfonation. The resulting 4-acetamidobenzenesulfanyl chloride is treated with ammonia to replace the chlorine with an amino group and affords 4-acetamidobenzenesulfonamide. The subsequent hydrolysis of the sulfonamide produces the sulfanilamide.
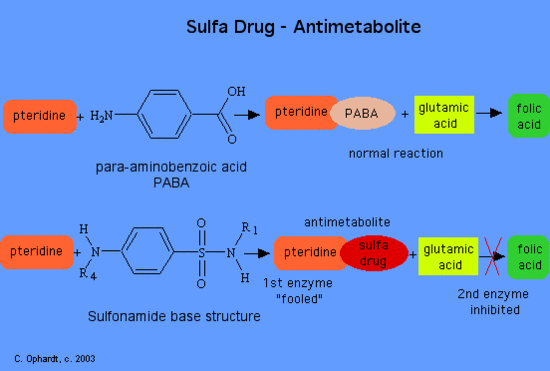
Their use introduced and substantiated the concept of metabolic antagonism. Sulfonamides, as antimetabolites, compete with para-aminobenzoic acid (PABA) for incorporation into folic acid. The action of sulfonamides illustrates the principle of selective toxicity where some difference between mammal cells and bacterial cells is exploited. All cells require folic acid for growth. Folic acid (as a vitamin is in food) diffuses or is transported into human cells. However, folic acid cannot cross bacterial cell walls by diffusion or active transport. For this reason bacteria must synthesize folic acid from p-aminobenzoic acid. Sulfonamides or sulfa drugs have the following general structures as shown below.
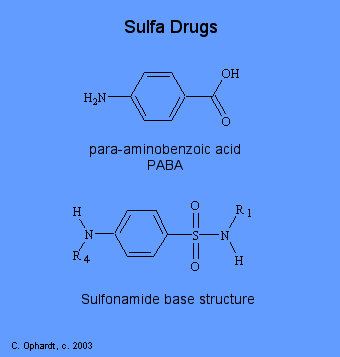
Sulfanilamide which was the first compound used of this type has H's at R1 and R4. To date about 15,000 sulfonamide derivatives, analogues, and related compounds have been synthesized. This has lead to the discovery of many useful drugs which are effective for diuretics, antimalerial and leprosy agents, and antithyroid agents. The basic structure of sulfonamide cannot be modified if it is to be an effective competitive "mimic" for p-aminobenzoic acid. Essential structural features are the benzene ring with two substituents para to each other; an amino group in the fourth position; and the singly substituted 1-sulfonamido group.

Mechanism for Action
Normally folic acid is synthesized in two steps in bacteria by the top reaction on the left. If A sulfa drug is used, the first enzyme is not to specific and can use the sulfonamide in the first reaction. This reaction produces the product containing pteridine and the sulfa drug. The next and final step is the reaction PABA + with glutamic acid to make folic acid. If the sulfa drug has been substituted for the PABA, then the final enzyme is inhibited and no folic acid is produced.
Recent studies indicate that substituents on the N(1) nitrogen may play the role of competing for a site on the enzyme surface reserved for the glutamate residue in p-aminobenzoic acid-glutamate through one of the following two ways:
- Direct competition in the linking of PABA-glutamate with the pteridine derivative.
- Indirect interference with the coupling of glutamate to dihydropteroic acid.