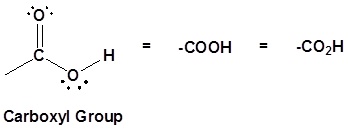Structure of the carboxyl acid group
Carboxylic acids are organic compounds which incorporate a carboxyl functional group, CO2H. The name carboxyl comes from the fact that a carbonyl and a hydroxyl group are attached to the same carbon.
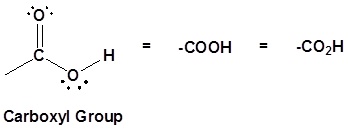
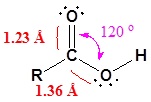
The carbon and oxygen in the carbonyl are both sp2 hybridized which give a carbonyl group a basic trigonal shape. The hydroxyl oxygen is also sp2 hybridized which allows one of its lone pair electrons to conjugate with the pi system of the carbonyl group. This make the carboxyl group planar an can represented with the following resonance structure.
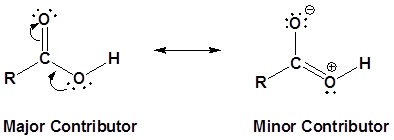
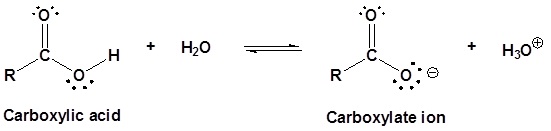
Carboxylic acids are named such because they can donate a hydrogen to produce a carboxylate ion. The factors which affect the acidity of carboxylic acids are discussed in the next section of this chapter.
Physical Properties of Some Carboxylic Acids
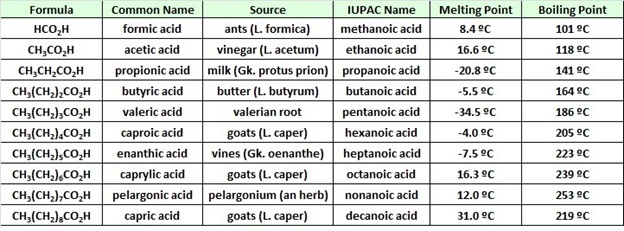
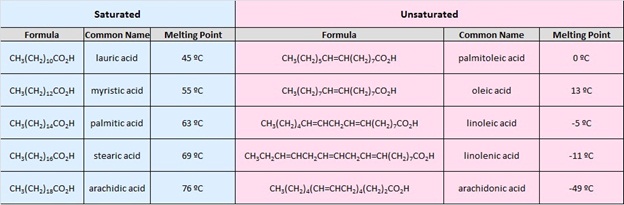
The table at the beginning of this page gave the melting and boiling points for a homologous group of carboxylic acids having from one to ten carbon atoms. The boiling points increased with size in a regular manner, but the melting points did not. Unbranched acids made up of an even number of carbon atoms have melting points higher than the odd numbered homologs having one more or one less carbon. This reflects differences in intermolecular attractive forces in the crystalline state. In the table of fatty acids we see that the presence of a cis-double bond significantly lowers the melting point of a compound. Thus, palmitoleic acid melts over 60º lower than palmitic acid, and similar decreases occur for the C18 and C20 compounds. Again, changes in crystal packing and intermolecular forces are responsible.
The factors that influence the relative boiling points and water solubilities of various types of compounds were discussed earlier. In general, dipolar attractive forces between molecules act to increase the boiling point of a given compound, with hydrogen bonds being an extreme example. Hydrogen bonding is also a major factor in the water solubility of covalent compounds To refresh your understanding of these principles Click Here. The following table lists a few examples of these properties for some similar sized polar compounds (the non-polar hydrocarbon hexane is provided for comparison).
The first five entries all have oxygen functional groups, and the relatively high boiling points of the first two is clearly due to hydrogen bonding. Carboxylic acids have exceptionally high boiling points, due in large part to dimeric associations involving two hydrogen bonds. A structural formula for the dimer of acetic acid is shown here. When the mouse pointer passes over the drawing, an electron cloud diagram will appear. The high boiling points of the amides and nitriles are due in large part to strong dipole attractions, supplemented in some cases by hydrogen bonding.

|
Formula
|
IUPAC Name
|
Molecular Weight
|
Boiling Point
|
Water Solubility
|
Physical Properties of Some Organic Compounds
| CH3(CH2)2CO2H |
butanoic acid |
88 |
164 ºC |
very soluble |
| CH3(CH2)4OH |
1-pentanol |
88 |
138 ºC |
slightly soluble |
| CH3(CH2)3CHO |
pentanal |
86 |
103 ºC |
slightly soluble |
| CH3CO2C2H5 |
ethyl ethanoate |
88 |
77 ºC |
moderately soluble |
| CH3CH2CO2CH3 |
methyl propanoate |
88 |
80 ºC |
slightly soluble |
| CH3(CH2)2CONH2 |
butanamide |
87 |
216 ºC |
soluble |
| CH3CON(CH3)2 |
N,N-dimethylethanamide |
87 |
165 ºC |
very soluble |
| CH3(CH2)4NH2 |
1-aminobutane |
87 |
103 ºC |
very soluble |
| CH3(CH2)3CN |
pentanenitrile |
83 |
140 ºC |
slightly soluble |
| CH3(CH2)4CH3 |
hexane |
86 |
69 ºC |
insoluble |
Carboxylate Salts
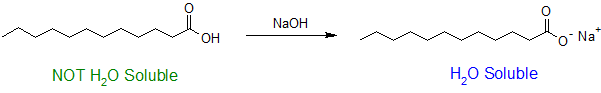
The water solubility of carboxylic acids is determined by the ratio of carboxyl groups to the the number of carbon atoms in the molecule following the "4 to 6 Rule". As seen with amines, water solubility of carboxylic acids can be increased when they are ionized. Typically a strong base is used to deprotonate the carboxylic acid and drive the reaction to completion as shown below.
Exercise
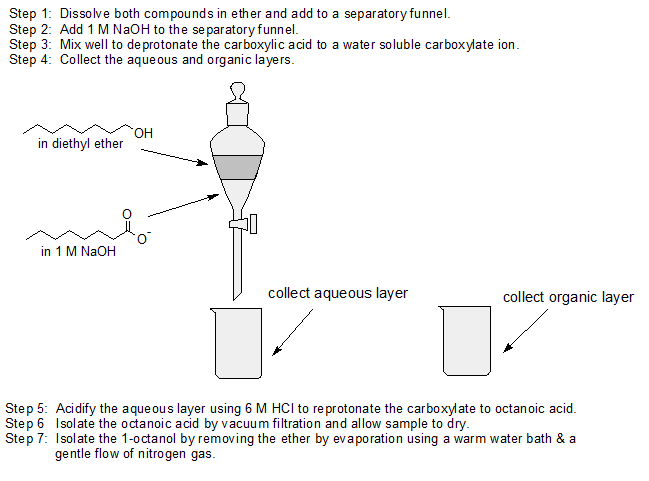
1. Use acid-base chemistry and differences in water solubility to separate 1-octanol from octanoic acid using the following solutions: 1 M NaOH, ether, and 6 M HCl and any lab equipment.
- Answer
-
1.