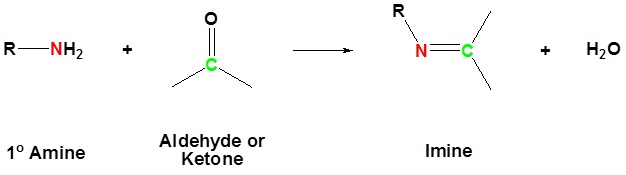
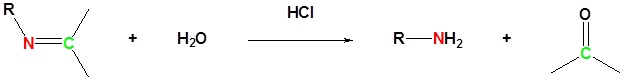
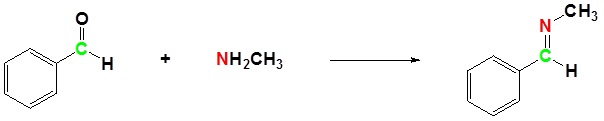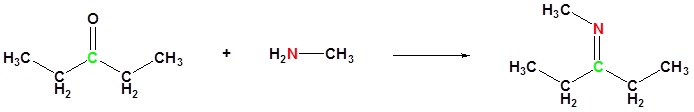The reaction of aldehydes and ketones with ammonia or 1º-amines forms imine derivatives, also known as Schiff bases (compounds having a C=N function). Water is eliminated in the reaction, which is acid-catalyzed and reversible in the same sense as acetal formation. The pH for reactions which form imine compounds must be carefully controlled. The rate at which these imine compounds are formed is generally greatest near a pH of 5, and drops at higher and lower pH's. At high pH there will not be enough acid to protonate the OH in the intermediate to allow for removal as H2O. At low pH most of the amine reactant will be tied up as its ammonium conjugate acid and will become non-nucleophilic.
Converting reactants to products simply
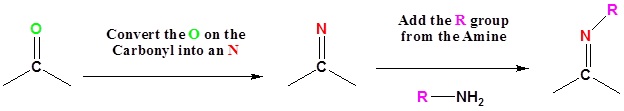
1) Nucleophilic addition reaction

2) Proton transfer
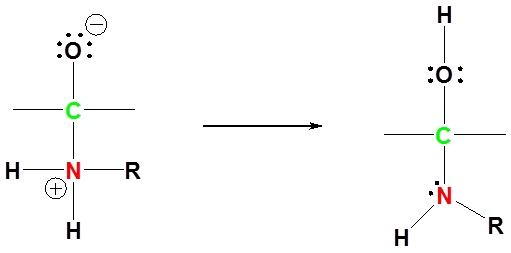
3) Protonation of OH
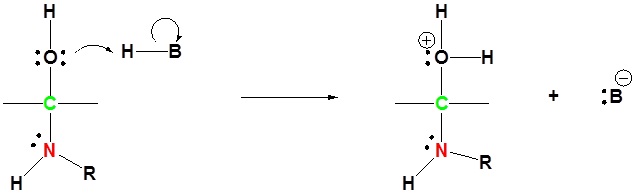
4) Removal of water
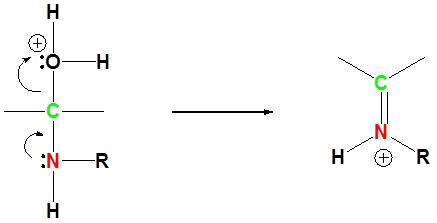
5) Deprotonation to form neutral final product
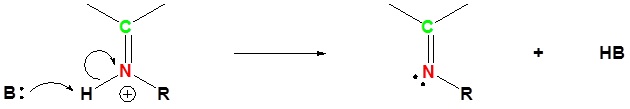
Imines can be hydrolyzed back to the corresponding primary amine under acidic aqueous conditons.
Reactions involving other reagents of the type Y-NH2
Imines are sometimes difficult to isolate and purify due to their sensitivity to hydrolysis. Consequently, other reagents of the type Y–NH2 have been studied, and found to give stable products (R2C=N–Y) useful in characterizing the aldehydes and ketones from which they are prepared. Some of these reagents are listed in the following table, together with the structures and names of their carbonyl reaction products. Hydrazones are used as part of the Wolff-Kishner reduction and will be discussed in more detail in another module.
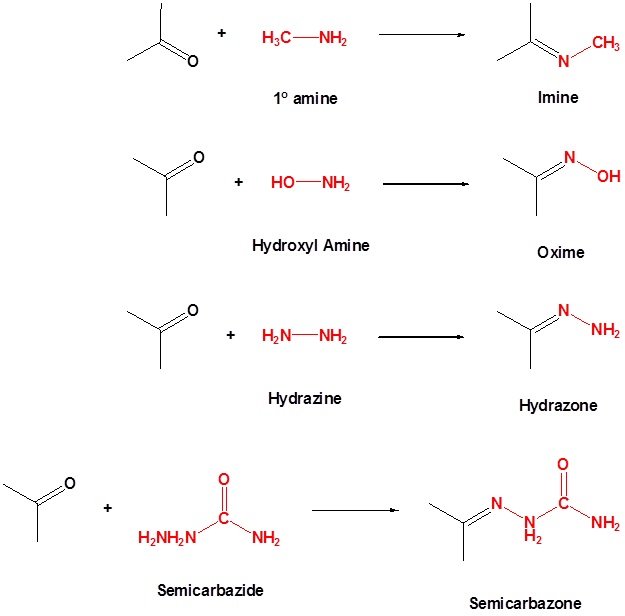
With the exception of unsubstituted hydrazones, these derivatives are easily prepared and are often crystalline solids - even when the parent aldehyde or ketone is a liquid. Since melting points can be determined more quickly and precisely than boiling points, derivatives such as these are useful for comparison and identification of carbonyl compounds. It should be noted that although semicarbazide has two amino groups (–NH2) only one of them is a reactive amine. The other is amide-like and is deactivated by the adjacent carbonyl group.
Exercise
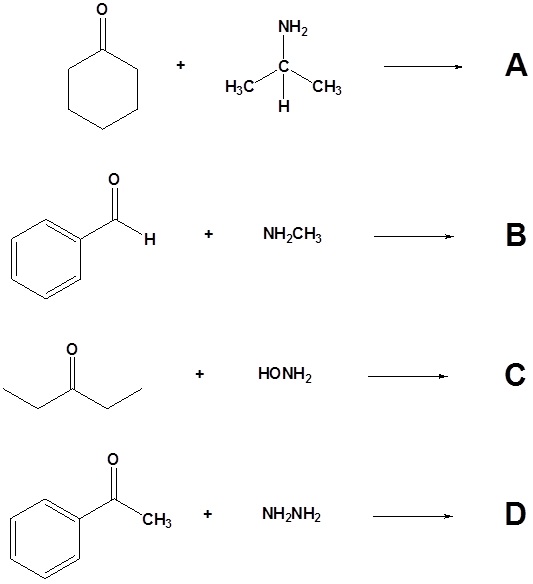
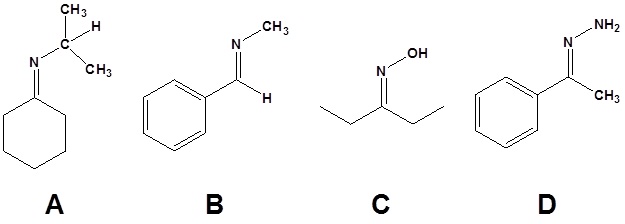
13. Draw the products of the following reactions.
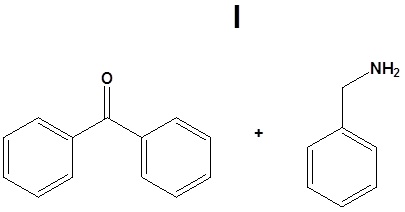
14. Draw the structure of the reactant needed to produce the indicated product.
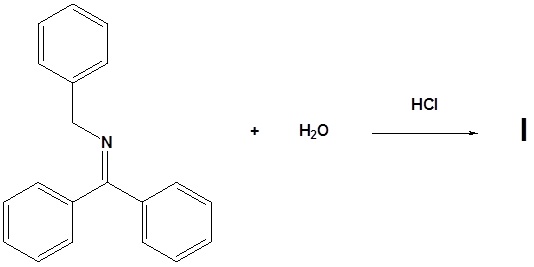
- Answer
-
13.
14.
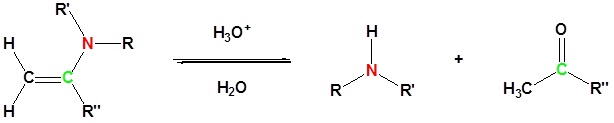
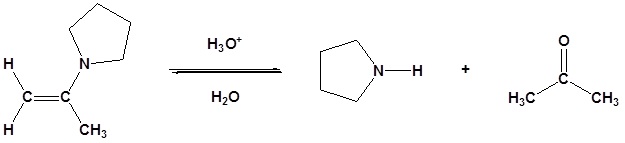
Most aldehydes and ketones react with 2º-amines to give products known as enamines. It should be noted that, like acetal formation, these are acid-catalyzed reversible reactions in which water is lost. Consequently, enamines are easily converted back to their carbonyl precursors by acid-catalyzed hydrolysis.
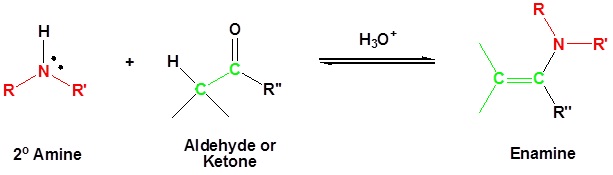

Mechanism
1) Nuleophilic addition reaction
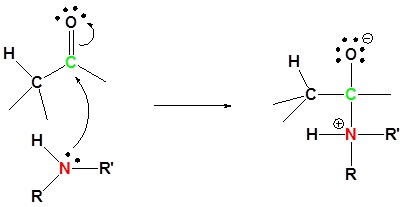
2) Proton transfer
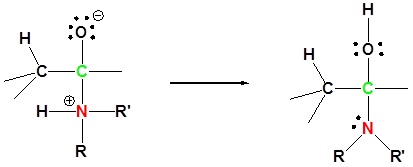
3) Protonation of OH
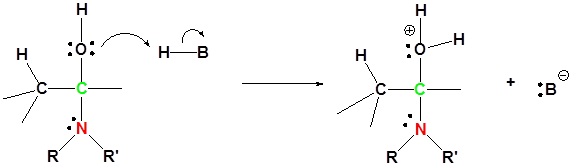
4) Removal of water
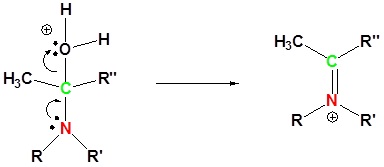
5) Deprotonation to neutralize final product
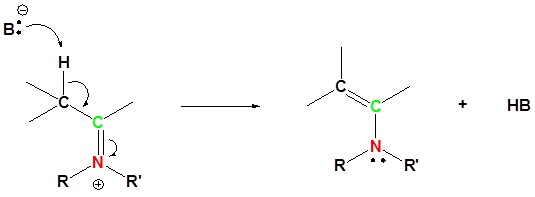
Reversibility of Enamines
Exercise
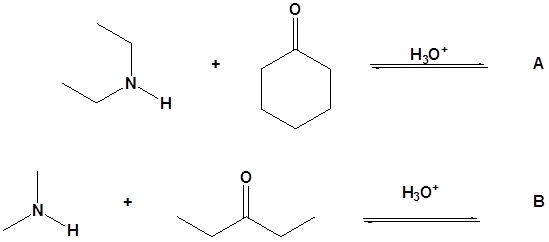
15. Draw the products for the following reactions.
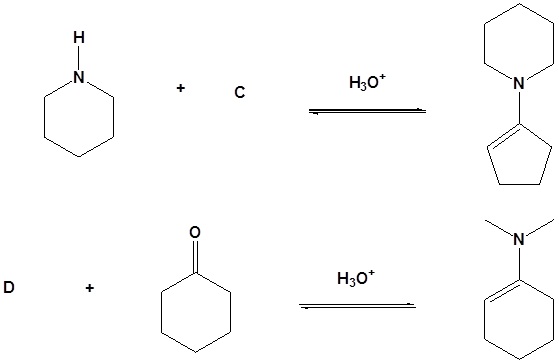
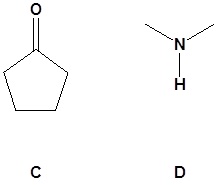
16. Draw the missing reactant to complete each reaction below.
- Answer
-
15.
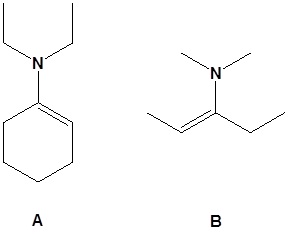
16.
Contributors and Attributions