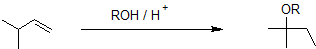Introduction
Acid-catalyzed ether synthesis from alkenes is limited by carbocation stability. Carbocation rearrangement can occur to form a more stable ion as shown in the example below.
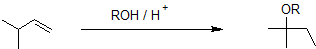
Acid-catalyzed ether synthesis using the alkoxymercuration-demercuration reaction pathway reliably produces the Markovnikov product without carbocation rearrangment as shown in the example below.
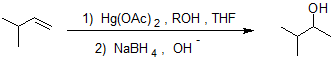
Alkoxymercuration-demercuration is a two step pathway used to produce ethers that proceeds in a Markovnikov manner and is stereospecific (anti addition). The two steps of alkoxymerecuration-demercuraton take place on opposite faces of the double bond creating trans stereochemistry.
Alkoxymercuration-Demercuration Mechanism
This reaction follows electrophilic addition mechanism we have learned. The major difference is that a mercurium ion bridge stabilizes the carbocation intermediate so that it cannot rearrange. Metals are electropositive. Mercury carries a partial positive charge in the acetate complex and is the electrophile. During the first step of this mechanism, the pi electrons form a bond to mercury while the lone pair on the mercury simultaneously bonds to the other vinyl carbon creating a mercurium ion bridge. The mercurium ion forms in conjunction with the loss of an acetate ion. The mercurium ion stabilizes the carbocation so that it does not rearrange. In the second step of this mechanism, an alcohol molecule reacts with the most substituted carbon to open the mercurium ion bridge. The third step of this mechanism is a proton transfer to a solvent alcohol molecule to neutralize the addition product. The fourth step of the reaction pathway is the reduction of the organomercury intermediate with sodium borohydride under basic conditions. The mechanism of the fourth step is beyond the scope of first year organic chemistry.
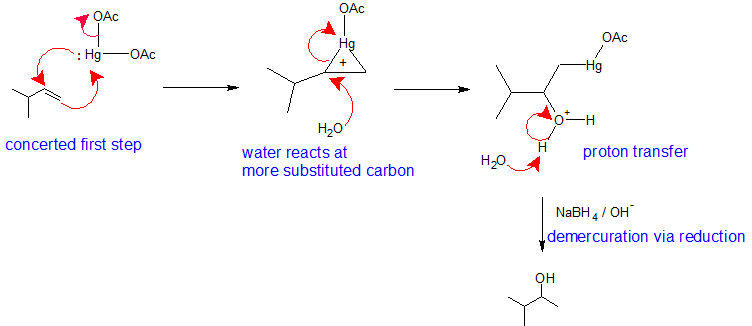
Notice that overall, the alkoxymercuration - demercuration mechanism follows Markovnikov's regioselectivity with the OR group attached to the most substituted carbon and the H attached to the least substituted carbon. The reaction is useful, because strong acids are not required and carbocation rearrangements are avoided because no discreet carbocation intermediate forms.
Exercise
6. Show how 3-methyl-2-isopropoxypentane may be synthesized by
a) Williamson ether synthesis
b) Alkoxymercuration-demercuration
c) Which synthesis is better? Why?
- Answer
-
6.
a)

b)

c) The alkoxymercuration-demercuration is more effective because the Williamson Synthesis would use a bulky base and secondary alkyl halide. These reactants would favor the elimination mechanism. Additionally, the alkene can under go carbocation rearrangement, so the mercurium ion stabilization of the reactive intermediate is needed.